Advanced Synthetic Route for 2-Hydroxy Propanedinitrile: Industrial Scalability and Cost Efficiency
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable pathways for critical intermediates that bridge the gap between laboratory discovery and commercial manufacturing. A pivotal advancement in this domain is detailed in patent CN102936209A, which discloses a practical and highly efficient synthetic method for 2-hydroxy propanedinitrile (also known as hydroxymalononitrile). This compound serves as a vital precursor for generating protected derivatives, such as dimethyl tertiary butyl silicon-protected or acetyl-protected variants, which are essential reagents for the rapid and efficient one-step synthesis of hydroxy esters or oxyamides. Historically, the production of this key building block has been hindered by operational complexity and safety concerns, but this new methodology offers a transformative solution by leveraging in situ generated peroxy acids. By shifting the paradigm from harsh, traditional oxidation protocols to a controlled, mild acidic environment, this technology addresses the longstanding bottlenecks of yield and scalability that have plagued R&D teams globally.
For procurement specialists and supply chain managers, the implications of this patent extend far beyond mere chemical curiosity; it represents a tangible opportunity for cost reduction in pharma manufacturing. The process utilizes commodity chemicals such as urea hydrogen peroxide, acetic anhydride, and trifluoroacetic anhydride, all of which are readily available in the global market at competitive price points. Unlike legacy routes that might require exotic catalysts or extreme pressure conditions, this method operates effectively at temperatures ranging from 0°C to 35°C. This mild thermal profile not only enhances safety by minimizing the risk of runaway exotherms but also reduces the energy burden on production facilities. As a reliable pharmaceutical intermediate supplier, understanding these underlying process efficiencies allows us to offer clients a more stable supply chain, less susceptible to the volatility associated with complex, multi-step syntheses that rely on scarce reagents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to the innovations described in CN102936209A, the synthesis of 2-hydroxy propanedinitrile was fraught with significant technical and economic challenges that rendered it unsuitable for modern industrial demands. Existing bibliographical information indicates that conventional raw materials were often difficult to obtain, necessitating complex supply chains that increased lead times and costs. Furthermore, the reaction conditions employed in traditional methods were frequently severe, requiring high temperatures or aggressive reagents that posed safety hazards and complicated equipment requirements. These harsh environments often led to prolonged reaction times, sometimes extending over many hours or even days, which drastically reduced throughput in batch reactors. Additionally, the enforceability of these old methods was low, meaning that reproducing laboratory results on a pilot or commercial scale was inconsistent, leading to variable yields and impurity profiles that required expensive purification steps. Ultimately, these factors combined to make industrial production economically unviable, forcing many manufacturers to rely on small-scale, high-cost sources or to seek alternative, less optimal synthetic routes.
The Novel Approach
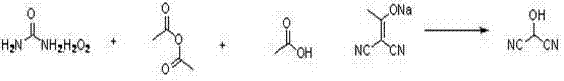
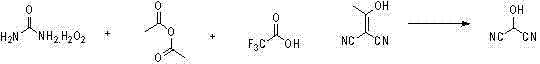
The novel approach presented in this patent fundamentally reengineers the synthetic pathway by utilizing 2-(1-hydroxy acetal) propanedinitrile as a stable, accessible starting material. This substrate undergoes a smooth transformation into the target 2-hydroxy propanedinitrile through reaction with a peroxy acid, which is conveniently generated in situ. The innovation lies in the specific combination of urea hydrogen peroxide with either acetic anhydride or trifluoroacetic anhydride to create the active oxidant directly within the reaction vessel. This eliminates the need to handle unstable, concentrated peroxy acids externally, thereby enhancing operational safety. The reaction proceeds efficiently under mild acidic catalysis, typically using acetic acid or trifluoroacetic acid, ensuring that the sensitive nitrile groups remain intact while the acetal functionality is selectively oxidized and hydrolyzed. This streamlined process not only shortens the overall reaction time significantly but also simplifies the post-reaction workup, allowing for straightforward extraction and isolation of the product as a stable oily liquid.
Mechanistic Insights into Peroxy Acid Mediated Oxidation
The core of this synthetic breakthrough relies on the mechanistic elegance of peroxy acid oxidation under acidic conditions. When urea hydrogen peroxide reacts with an acid anhydride (such as acetic anhydride), it generates a peroxy acid (like peracetic acid) and urea as a byproduct. This peroxy acid acts as a potent electrophilic oxidant. In the presence of the 2-(1-hydroxy acetal) propanedinitrile substrate, the peroxy acid attacks the electron-rich enol ether or acetal carbon center. The acidic environment, provided by the added carboxylic acid catalyst, protonates the oxygen atoms, facilitating the cleavage of the C-O bond and the subsequent formation of the hydroxyl group. This mechanism is highly selective; the mild acidity ensures that the nitrile groups (-CN), which are susceptible to hydrolysis under strong acidic or basic conditions, remain largely unaffected. This chemoselectivity is crucial for maintaining high purity, as it prevents the formation of amide or carboxylic acid byproducts that would otherwise contaminate the final intermediate. The use of urea hydrogen peroxide also provides a controlled release of active oxygen, preventing the localized hot spots that can occur with liquid hydrogen peroxide additions.
Impurity control is another critical aspect where this mechanism excels, directly addressing the concerns of R&D directors focused on quality. Traditional oxidation methods often generate a plethora of side products due to over-oxidation or radical pathways initiated by metal contaminants. In contrast, this metal-free organic oxidation system minimizes the introduction of heavy metal impurities, which is a significant advantage for pharmaceutical applications where residual metals must be strictly controlled. The reaction byproducts, primarily urea and the corresponding carboxylic acid, are water-soluble and easily removed during the aqueous workup phase involving sodium sulfite quenching and ethyl acetate extraction. The patent data indicates that yields can consistently reach above 74%, with some embodiments achieving up to 86%, demonstrating the robustness of this mechanistic pathway. The resulting product is stable when stored below 35°C, often in the form of an acetic acid solvate, which further simplifies logistics by reducing the need for immediate derivatization or cryogenic storage.
How to Synthesize 2-Hydroxy Propanedinitrile Efficiently
Implementing this synthesis in a production environment requires careful attention to the preparation of the oxidant and the control of reaction parameters to maximize yield and safety. The process begins with the in situ generation of the peroxy acid, followed by the controlled addition to the substrate solution. Detailed operational parameters, including specific molar ratios and temperature ramps, are critical for reproducibility. For a comprehensive understanding of the standardized operating procedures required to replicate these results safely and effectively, please refer to the technical guide below.
- Preparation of Peroxy Acid: React urea hydrogen peroxide with acetic anhydride or trifluoroacetic anhydride in dichloromethane at 0-35°C to generate the active oxidant in situ.
- Oxidative Reaction: Add the prepared peroxy acid solution to a mixture of 2-(1-hydroxy acetal) propanedinitrile and a catalytic acid (acetic or trifluoroacetic acid) while maintaining temperature between 0-35°C.
- Workup and Isolation: Quench the reaction with sodium sulfite, extract with ethyl acetate, dry over anhydrous sodium sulfate, and concentrate under vacuum to obtain the oily product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthetic route offers profound benefits that resonate deeply with procurement managers and supply chain heads tasked with optimizing margins and ensuring continuity. The shift towards using commodity-grade reagents like urea hydrogen peroxide and acetic anhydride decouples the production of this intermediate from the supply volatility of specialized, high-cost catalysts. This strategic sourcing advantage means that production schedules are less likely to be disrupted by shortages of niche chemicals, providing a more predictable manufacturing timeline. Furthermore, the simplified workup procedure, which avoids complex chromatography or distillation steps, translates directly into reduced processing time and lower utility consumption per kilogram of product. These operational efficiencies accumulate to create a leaner cost structure, allowing for more competitive pricing in the final API or intermediate market without sacrificing quality standards.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the use of inexpensive, bulk-available oxidants significantly lowers the raw material cost base. Additionally, the mild reaction conditions reduce energy consumption for heating and cooling, while the high selectivity minimizes waste generation and the associated costs of disposal and purification. This holistic reduction in operational expenditure allows for substantial cost savings that can be passed down the supply chain.
- Enhanced Supply Chain Reliability: By relying on a synthetic route that uses stable, solid oxidants (urea hydrogen peroxide) rather than hazardous liquid peroxides, the logistical barriers to entry are lowered. Storage and transport of the key reagents are safer and more compliant with international regulations, reducing the risk of shipping delays. The robustness of the reaction also means that batch-to-batch variability is minimized, ensuring a consistent supply of high-quality intermediate to downstream customers.
- Scalability and Environmental Compliance: The process is inherently scalable due to its exotherm management and lack of high-pressure requirements. The aqueous workup and the nature of the byproducts (urea, acetic acid) facilitate easier wastewater treatment compared to processes generating heavy metal sludge or halogenated organic waste. This alignment with green chemistry principles simplifies environmental permitting and reduces the long-term liability associated with hazardous waste management.
Frequently Asked Questions (FAQ)
To further clarify the technical and commercial implications of this technology, we have compiled a set of frequently asked questions based on the patent specifications and our internal process analysis. These answers address common concerns regarding safety, scalability, and product stability, providing a transparent view of what partners can expect when integrating this intermediate into their supply chain. Understanding these details is essential for making informed decisions about vendor selection and process adoption.
Q: What are the primary advantages of using urea hydrogen peroxide in this synthesis?
A: Urea hydrogen peroxide serves as a stable, solid source of hydrogen peroxide, which simplifies handling and storage compared to liquid peroxide solutions. It allows for the safe in situ generation of peroxy acids (like peracetic acid) under mild conditions (0-35°C), significantly reducing safety risks associated with exothermic reactions and enabling easier industrial scale-up.
Q: How does this method improve upon conventional synthetic routes for 2-hydroxy propanedinitrile?
A: Conventional methods often suffer from complex operational steps, severe reaction conditions, and prolonged reaction times, leading to low yields and poor enforceability for industrial production. This novel approach utilizes readily available starting materials and mild oxidative conditions to achieve yields exceeding 74%, with a simplified workup process that involves standard extraction and concentration, making it highly suitable for large-scale manufacturing.
Q: What are the critical storage conditions for the synthesized 2-hydroxy propanedinitrile?
A: The product, 2-hydroxy propanedinitrile, exists stably in an acetic acid form. To maintain stability and prevent decomposition, the storage temperature should be kept below 35°C. The patent suggests preserving the product at a substance concentration between 1 to 15 mol/L to ensure optimal shelf-life and chemical integrity for downstream applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Hydroxy Propanedinitrile Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from a patented laboratory method to a commercial reality requires more than just chemical knowledge; it demands engineering excellence and rigorous quality assurance. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising yields reported in CN102936209A are not just theoretical but are consistently achieved in our manufacturing suites. We adhere to stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to verify the identity and purity of every batch, guaranteeing that the 2-hydroxy propanedinitrile supplied meets the exacting standards required for pharmaceutical synthesis.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthetic route can benefit your specific projects. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our implementation of this technology compares to your current supply options. We encourage you to contact us today to索取 specific COA data and route feasibility assessments, allowing us to demonstrate our commitment to being a reliable partner in your drug development and manufacturing journey.
