Advanced Synthesis of Pyrrole Hydrazone Difluoroboron Fluorescent Dyes for Commercial OLED Applications
Advanced Synthesis of Pyrrole Hydrazone Difluoroboron Fluorescent Dyes for Commercial OLED Applications
The rapid evolution of the organic light-emitting diode (OLED) industry demands materials that offer superior photoelectric performance alongside manufacturability. Patent CN108530474B introduces a groundbreaking class of pyrrole hydrazone hydrazine difluoroboron fluorescent dyes that address these critical needs through a novel molecular architecture. These compounds, characterized by a large molecular plane and rigid structure, demonstrate exceptional molar absorption coefficients and high fluorescence quantum yields, positioning them as prime candidates for next-generation display technologies. The patent details a robust 'one-pot' preparation工艺 that streamlines production, moving away from complex multi-step sequences that often plague fine chemical manufacturing. By integrating condensation and coordination into a unified workflow, this technology offers a pathway to high-purity electronic chemicals with reduced processing overhead.
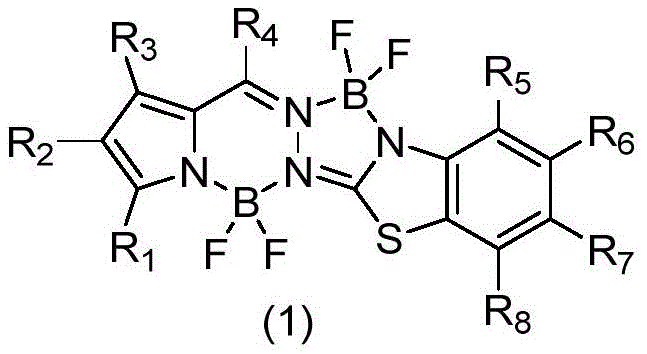
For R&D directors and procurement specialists seeking a reliable OLED material supplier, understanding the structural versatility of these dyes is paramount. The general formula allows for extensive derivatization at positions R1 through R8, enabling fine-tuning of emission wavelengths and solubility profiles without compromising the core fluorophore's stability. This adaptability is crucial for customizing materials for specific device architectures, whether for green, red, or blue emission layers. Furthermore, the inherent photostability of the difluoroboron core ensures long operational lifetimes for the final electronic devices, a key metric for consumer electronics manufacturers. The ability to modify the pyrrole and benzothiazole moieties independently provides a vast chemical space for optimization, ensuring that intellectual property portfolios can be expanded around this core scaffold.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis routes for organoboron fluorescent dyes often suffer from significant inefficiencies that hinder commercial scale-up. Conventional methods typically require the isolation of intermediate hydrazones prior to the boron coordination step, necessitating multiple purification stages such as column chromatography or recrystallization between each reaction. This multi-step approach not only increases the consumption of solvents and silica gel but also leads to cumulative yield losses that can render the process economically unviable for mass production. Additionally, many existing protocols rely on harsh reaction conditions or expensive transition metal catalysts that introduce difficult-to-remove impurities, complicating the purification of high-purity electronic chemicals. The sensitivity of boron complexes to moisture and hydrolysis in open systems further exacerbates these challenges, requiring stringent anhydrous conditions that drive up capital expenditure for specialized reactor equipment.
The Novel Approach
In stark contrast, the methodology disclosed in CN108530474B employs a streamlined one-pot strategy that dramatically enhances process efficiency. By conducting the acid-catalyzed condensation of pyrrole aldehydes and 2-hydrazinobenzothiazoles followed directly by base treatment and boron trifluoride coordination in the same vessel, the process eliminates the need for intermediate isolation. This telescoped approach minimizes material handling, reduces solvent waste, and significantly shortens the overall production cycle time. The use of commercially available starting materials, such as substituted pyrrole aldehydes and benzothiazole derivatives, ensures that the supply chain remains robust and cost-effective. Moreover, the reaction tolerates a wide range of functional groups, allowing for the synthesis of diverse derivatives without the need for protecting group strategies, which further simplifies the synthetic route and reduces raw material costs.
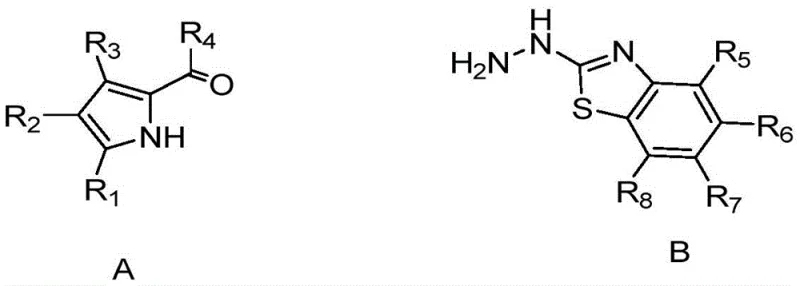
Mechanistic Insights into One-Pot Condensation and Coordination
The chemical mechanism underpinning this synthesis involves a carefully orchestrated sequence of acid-base catalysis and Lewis acid coordination. Initially, under acidic conditions provided by catalysts like p-toluenesulfonic acid, the nucleophilic nitrogen of the hydrazine moiety attacks the electrophilic carbonyl carbon of the pyrrole aldehyde. This condensation step proceeds through a tetrahedral intermediate to form a hydrazone linkage, releasing water as the byproduct. The acidity of the medium is critical here, as it activates the carbonyl group while preventing premature decomposition of the sensitive hydrazine reactant. Once the hydrazone formation is complete, the addition of a base such as N,N-diisopropylethylamine serves a dual purpose: it neutralizes the acidic environment and deprotonates the hydrazone nitrogen, generating a nucleophilic species ready for metal coordination.
Subsequent addition of boron trifluoride diethyl etherate triggers the final cyclization and complexation step. The boron atom, acting as a strong Lewis acid, coordinates with the nitrogen atoms of the hydrazone and the adjacent heterocyclic nitrogen, forming a rigid five-membered chelate ring. This coordination locks the molecule into a planar conformation, which is essential for maximizing pi-conjugation across the entire molecular framework. The resulting planarity restricts intramolecular rotation, thereby minimizing non-radiative decay pathways and leading to the observed high fluorescence quantum yields. The presence of two boron atoms in the final structure, as seen in some derivatives, further enhances the rigidity and electronic delocalization, contributing to the dye's exceptional photophysical properties and stability against photobleaching.
How to Synthesize Pyrrole Hydrazone Difluoroboron Dye Efficiently
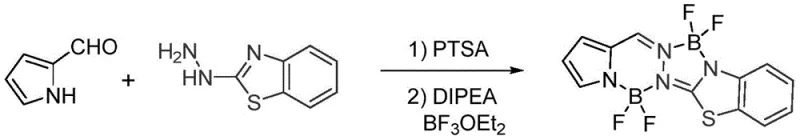
Implementing this synthesis on a pilot or commercial scale requires precise control over reaction parameters to maximize yield and purity. The process begins with the dissolution of the pyrrole aldehyde derivative and the 2-hydrazinobenzothiazole derivative in a suitable solvent such as 1,2-dichloroethane or toluene. An acid catalyst is introduced to initiate the condensation, and the mixture is heated to reflux temperatures between 70°C and 120°C for several hours until the starting aldehyde is consumed. Following this, the reaction mixture is treated with a base to adjust the pH to a slightly alkaline range, preparing the system for the critical boron coordination step. The detailed standardized synthesis steps, including specific molar ratios and workup procedures, are outlined below to ensure reproducibility and quality control.
- Conduct the first contact reaction between pyrrole aldehyde derivatives and 2-hydrazinobenzothiazole derivatives under acidic conditions (pH 5.1-6.9) at 70-120°C.
- Perform alkali treatment on the reaction system to adjust pH to 7.1-10 using organic or inorganic bases.
- Add boron trifluoride diethyl etherate and heat to 80-120°C for the second contact reaction to form the final difluoroboron complex.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this technology translates into tangible operational benefits that extend beyond simple yield metrics. The reliance on commodity chemicals as starting materials mitigates the risk of supply chain disruptions often associated with exotic or custom-synthesized reagents. Since the raw materials are widely produced for other pharmaceutical and agrochemical applications, sourcing is straightforward, and pricing remains stable even during periods of market volatility. The simplification of the synthetic route also implies a reduction in the number of unit operations required, which directly correlates to lower labor costs and reduced energy consumption per kilogram of product. This efficiency gain is particularly significant when scaling from laboratory grams to multi-tonne commercial batches.
- Cost Reduction in Manufacturing: The elimination of intermediate isolation steps drastically reduces the consumption of solvents and stationary phases used in purification. By avoiding multiple chromatographic separations, the process lowers the cost of goods sold (COGS) and minimizes the generation of hazardous chemical waste. The one-pot nature of the reaction means that reactor occupancy time is reduced, allowing for higher throughput in existing manufacturing facilities without the need for significant capital investment in new equipment. Furthermore, the high atom economy of the condensation-coordination sequence ensures that a greater proportion of raw material mass is incorporated into the final product, enhancing overall process sustainability.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions, which tolerate a range of temperatures and pH levels, makes the process less susceptible to minor fluctuations in operating parameters. This tolerance reduces the likelihood of batch failures due to operator error or equipment variance, ensuring a consistent supply of high-quality material. The use of stable intermediates and the avoidance of highly reactive or unstable species further enhance safety profiles, facilitating easier transport and storage of materials within the supply chain. Consequently, lead times for high-purity electronic chemicals can be significantly shortened, enabling faster response to customer demand.
- Scalability and Environmental Compliance: The process is inherently scalable, as demonstrated by the successful synthesis of various derivatives with different steric and electronic properties. The ability to tune the reaction by simply swapping substituents allows for the rapid development of a portfolio of related dyes without re-engineering the entire process. From an environmental perspective, the reduction in solvent usage and waste generation aligns with increasingly stringent global regulations on chemical manufacturing. The potential for solvent recovery and recycling in a closed-loop system further diminishes the environmental footprint, making this technology attractive for companies committed to green chemistry principles.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these advanced fluorescent dyes. These answers are derived directly from the technical specifications and experimental data provided in the patent documentation, ensuring accuracy and relevance for industry professionals. Understanding these nuances is essential for integrating this material into your existing product development pipelines.
Q: What are the key advantages of the one-pot synthesis method described in CN108530474B?
A: The one-pot method significantly simplifies the process by combining condensation and coordination steps, reducing solvent consumption and operational time compared to multi-step traditional syntheses.
Q: What are the optical properties of these pyrrole hydrazone difluoroboron dyes?
A: These dyes exhibit high molar absorption coefficients, high fluorescence quantum yields (ranging from 0.53 to 0.87 in various solvents), and excellent photostability, making them ideal for OLED applications.
Q: Are the raw materials for this synthesis commercially available?
A: Yes, the synthesis utilizes commercially available pyrrole aldehyde derivatives and 2-hydrazinobenzothiazole derivatives, ensuring a stable and scalable supply chain for industrial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyrrole Hydrazone Difluoroboron Dye Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-performance fluorescent dyes play in the advancement of OLED technology and electronic displays. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from R&D to mass manufacturing is seamless. We adhere to stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch meets the exacting standards required for electronic grade materials. Our commitment to quality assurance means that you receive products with consistent optical properties and minimal impurity profiles, essential for maintaining the performance of your final devices.
We invite you to collaborate with us to leverage this innovative synthesis technology for your specific application needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements and purity targets. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise in fine chemical intermediates can drive value and efficiency in your supply chain.
