Scalable Synthesis of Aromia Bungii Aggregation Pheromone for Advanced Pest Control Solutions
The global agricultural sector faces increasing pressure from invasive species, particularly the pink-necked longhorn beetle, Aromia bungii, which devastates Prunus fruit trees. Effective management of this pest relies heavily on the availability of its male-produced aggregation pheromone, (E)-cis-6,7-epoxy-2-nonenal. Patent CN109678818B introduces a groundbreaking synthetic methodology that addresses the critical supply chain bottlenecks associated with this high-value agrochemical intermediate. By utilizing 1,4-butanediol as a foundational building block, this novel seven-step sequence achieves an overall yield of 6.5% while maintaining mild reaction conditions that are inherently safer and more cost-effective than previous iterations. For R&D directors and procurement specialists, this patent represents a pivotal shift towards sustainable and economically viable pheromone production, eliminating the reliance on scarce natural precursors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methodologies for synthesizing (E)-cis-6,7-epoxy-2-nonenal have been plagued by significant economic and operational hurdles that hinder widespread adoption in integrated pest management programs. Early approaches, such as those by Millar et al., relied on the direct epoxidation of violet leaf aldehyde, a raw material that is prohibitively expensive and difficult to source in bulk quantities, rendering the process unsuitable for industrial application. Subsequent improvements by Kenji Mori utilized cis-2-pentenol and Sharpless asymmetric epoxidation; however, these routes necessitated the use of harsh Grignard coupling reagents and complex olefin cross-metathesis steps. These traditional pathways not only incurred high operational costs due to expensive chiral catalysts and cryogenic conditions but also presented substantial safety risks and waste disposal challenges, creating a fragile supply chain for this critical pest control agent.
The Novel Approach
The innovative strategy outlined in CN109678818B fundamentally重构 s the synthetic landscape by employing 1,4-butanediol, a commodity chemical, as the starting material. This approach streamlines the carbon chain assembly through a logical sequence of protection, oxidation, and chain-extension reactions that avoid the pitfalls of earlier methods. The process initiates with the monoprotection of the diol followed by selective TEMPO-mediated oxidation, establishing the aldehyde functionality required for subsequent Wittig olefination. By replacing expensive chiral pool materials with a robust, linear synthesis, this method drastically simplifies the manufacturing workflow. The strategic use of mild oxidants and controlled Wittig conditions ensures high fidelity in stereochemistry without the need for complex purification protocols, thereby enhancing the overall process mass intensity and making it highly attractive for reliable agrochemical intermediate suppliers seeking to optimize their production portfolios.
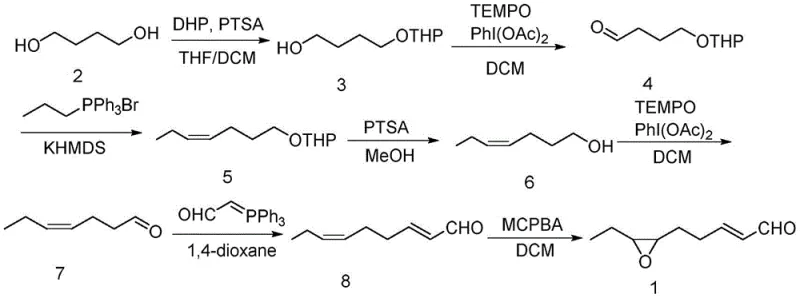
Mechanistic Insights into TEMPO-Mediated Oxidation and Stereoselective Wittig Olefination
The core chemical elegance of this synthesis lies in the repeated application of TEMPO (2,2,6,6-tetramethylpiperidin-1-yl)oxyl catalyzed oxidation coupled with stoichiometric PhI(OAc)2 as the terminal oxidant. This system operates via a catalytic cycle where the oxoammonium cation, generated in situ, selectively abstracts a hydride from the primary alcohol to form the corresponding aldehyde. This mechanism is particularly advantageous for R&D teams because it proceeds under neutral to slightly acidic conditions in dichloromethane, preventing the over-oxidation to carboxylic acids that often plagues chromium-based oxidations. Furthermore, the compatibility of TEMPO with the acid-labile tetrahydropyranyl (THP) protecting group allows for orthogonal reactivity, enabling the chemist to manipulate the carbon chain while preserving the protected alcohol moiety for later deprotection, thus ensuring a clean impurity profile throughout the multi-step sequence.
Stereochemical control is rigorously maintained during the chain extension phases through the careful selection of bases for the Wittig reactions. The formation of the Z-alkene geometry in the intermediate (Z)-4-heptenal is achieved by generating the ylide using potassium hexamethyldisilazide (KHMDS) at low temperatures, typically around -78°C. This kinetic control favors the formation of the Z-isomer over the thermodynamically more stable E-isomer, which is crucial for the biological activity of the final pheromone. The subsequent epoxidation with meta-chloroperoxybenzoic acid (MCPBA) exploits the electronic difference between the conjugated E-alkene and the isolated Z-alkene; the electron-rich isolated double bond reacts preferentially to form the cis-epoxide ring. This mechanistic understanding allows for precise tuning of reaction parameters to minimize geometric isomers, ensuring the production of high-purity insect pheromone that meets stringent bioassay requirements.
How to Synthesize (E)-cis-6,7-epoxy-2-nonenal Efficiently
Executing this synthesis requires strict adherence to the optimized conditions detailed in the patent to maximize yield and purity. The process begins with the protection of 1,4-butanediol using 3,4-dihydro-2H-pyran (DHP) in a THF/DCM solvent system catalyzed by p-toluenesulfonic acid. Following isolation, the mono-protected alcohol undergoes oxidation to the aldehyde, which is immediately subjected to Wittig olefination with n-propyltriphenylphosphonium bromide. The detailed standardized synthesis steps, including specific molar ratios, temperature controls, and workup procedures for each of the seven transformations, are provided in the technical guide below to ensure reproducibility at scale.
- Protect 1,4-butanediol with DHP and oxidize to aldehyde using TEMPO/PhI(OAc)2.
- Perform Wittig reaction with propyltriphenylphosphonium salt to extend the carbon chain with Z-selectivity.
- Deprotect, oxidize to aldehyde, perform second Wittig reaction, and finalize with MCPBA epoxidation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this 1,4-butanediol-based route offers transformative benefits that directly impact the bottom line and operational resilience. The substitution of exotic starting materials with a bulk commodity chemical eliminates the volatility associated with niche raw material markets, ensuring a stable and continuous supply of feedstock. Moreover, the elimination of cryogenic Grignard reactions and expensive chiral catalysts significantly reduces the complexity of the manufacturing infrastructure, allowing for production in standard multipurpose reactors without the need for specialized low-temperature equipment. This simplification translates directly into lower capital expenditure and reduced operational overhead, positioning this method as a superior choice for cost reduction in pheromone manufacturing.
- Cost Reduction in Manufacturing: The utilization of 1,4-butanediol as the primary feedstock represents a substantial decrease in raw material costs compared to violet aldehyde or cis-2-pentenol. Additionally, the avoidance of precious metal catalysts and the use of recyclable solvents like dichloromethane and THF further drive down the cost of goods sold. The streamlined seven-step sequence minimizes unit operations, reducing labor costs and energy consumption associated with heating and cooling cycles, thereby delivering significant economic advantages for large-volume production runs.
- Enhanced Supply Chain Reliability: By relying on globally available commodity chemicals, the risk of supply disruption is markedly reduced. The robust nature of the TEMPO oxidation and Wittig steps ensures consistent batch-to-batch quality, minimizing the need for reprocessing or scrapping off-spec material. This reliability is critical for maintaining the continuity of pest control programs, particularly during peak seasons when demand for pheromone traps surges, ensuring that downstream formulators receive their orders on time without delay.
- Scalability and Environmental Compliance: The mild reaction conditions and the absence of heavy metal waste streams simplify the environmental compliance burden. The process generates less hazardous waste compared to chromium or manganese-based oxidations, facilitating easier disposal and treatment. Furthermore, the high atom economy of the Wittig reactions and the efficient protection-deprotection strategy make this route highly scalable from kilogram to multi-ton production, supporting the commercial scale-up of complex agrochemical intermediates without compromising safety or environmental standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of this aggregation pheromone. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on the feasibility and advantages of this specific manufacturing route for potential partners and technical stakeholders.
Q: What is the primary advantage of using 1,4-butanediol as a starting material?
A: 1,4-butanediol is significantly cheaper and more readily available than previous starting materials like violet aldehyde or cis-2-pentenol, drastically reducing raw material costs for large-scale manufacturing.
Q: How does this method improve stereocontrol compared to prior art?
A: The use of KHMDS in the Wittig reaction ensures high Z-selectivity for the internal double bond, while the final epoxidation with MCPBA selectively targets the electron-rich alkene to form the cis-epoxide.
Q: Is this synthesis route suitable for industrial scale-up?
A: Yes, the patent explicitly states the conditions are mild and operations are simple, avoiding harsh reagents like Grignard reagents or expensive chiral catalysts, making it ideal for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (E)-cis-6,7-epoxy-2-nonenal Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality pheromones play in modern sustainable agriculture. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that utilize advanced analytical techniques to verify stereochemical integrity and impurity profiles, guaranteeing that every batch meets the highest standards for field efficacy.
We invite you to collaborate with us to leverage this innovative synthesis technology for your pest control formulations. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can enhance your supply chain efficiency and reduce your overall procurement costs.
