Advanced Synthesis of Chiral (S)-3-Methylheptanoic Acid for Prostaglandin Manufacturing
Advanced Synthesis of Chiral (S)-3-Methylheptanoic Acid for Prostaglandin Manufacturing
The pharmaceutical industry continuously seeks efficient pathways for constructing chiral building blocks essential for complex drug synthesis. Patent CN101274891A introduces a groundbreaking methodology for the preparation of optically pure (S)-3-methylheptanoic acid, a critical intermediate in the total synthesis of cycloprostaglandin ISM10902 and Prostaglandin E1 analogues. This technology represents a significant paradigm shift by transforming waste products from steroidal sapogenin oxidative degradation into high-value chiral synthons. By leveraging the inherent chirality of (R)-γ-methyl-δ-valerolactone, a byproduct often discarded in steroid processing, this invention offers a sustainable and economically viable alternative to traditional asymmetric synthesis routes that rely on costly chiral auxiliaries or resolution techniques.
The strategic importance of (S)-3-methylheptanoic acid cannot be overstated in the context of modern medicinal chemistry, particularly for cardiovascular and inflammatory disease treatments. Conventional approaches, such as those developed by the Mukaiyama group in the late 1970s or the Badía group in 2006, often involve multi-step sequences utilizing chiral prosthetic groups like (S,S)-(+)-pseudoephedrine. While effective, these methods introduce significant operational complexity and cost burdens due to the stoichiometric requirement of chiral reagents and the necessity for their subsequent removal. In contrast, the methodology disclosed in CN101274891A streamlines the synthetic trajectory, utilizing a waste-derived starting material that already possesses the requisite stereochemistry, thereby eliminating the need for external chiral induction agents and reducing the overall environmental footprint of the manufacturing process.
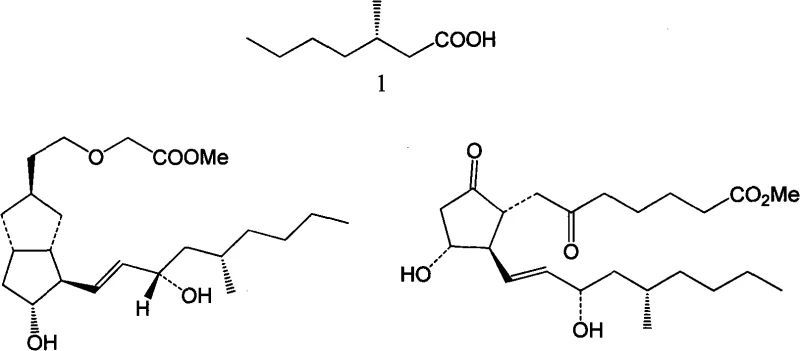
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3-substituted organic acids with high optical purity has been a formidable challenge for process chemists. Early methodologies, such as the Michael addition of Grignard reagents to unsaturated amides bearing chiral auxiliaries, require rigorous control of reaction conditions to maintain enantiomeric excess. These processes are inherently inefficient because the chiral auxiliary, which constitutes a significant portion of the molecular weight during the synthesis, must eventually be cleaved and ideally recovered, adding extra unit operations to the workflow. Furthermore, sourcing high-purity chiral starting materials like (S)-citronellol or (S)-ethyl 3-hydroxybutyrate can be subject to market volatility and supply chain disruptions, creating bottlenecks for large-scale production. The reliance on these specific natural products or specialized reagents limits the flexibility of the supply chain and often results in higher cost of goods sold (COGS) due to the premium pricing of enantiopure feedstocks.
The Novel Approach
The innovative route described in this patent circumvents these limitations by employing a clever strategy of "waste valorization." The process begins with (R)-γ-methyl-δ-valerolactone, a small fragment obtained from the degradation of steroidal sapogenins using hydrogen peroxide, a greener oxidant compared to traditional chromium-based reagents. This starting material is converted into a series of (S)-4-methyloctane derivatives through a sequence of highly selective carbon-carbon bond-forming reactions. The core of this novelty lies in the use of a diphenyl hydroxymethyl group as a temporary masking and chain-extending moiety, which is later removed via oxidative cleavage to reveal the target carboxylic acid. This approach not only simplifies the operational procedure by avoiding complex chiral chromatography or enzymatic resolutions but also aligns with green chemistry principles by upcycling industrial waste streams into valuable pharmaceutical intermediates.
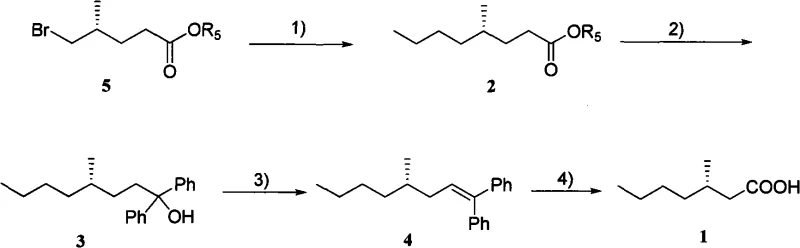
Mechanistic Insights into Grignard Addition and Oxidative Cleavage
The chemical elegance of this synthesis is rooted in the precise control of organometallic reactivity. The first critical transformation involves the reaction of the bromo-ester or lactone precursor (Compound 5) with n-propylmagnesium bromide in the presence of copper salts such as Li2CuCl4. This copper-catalyzed conjugate addition or nucleophilic acyl substitution is pivotal for establishing the carbon skeleton while preserving the stereocenter at the 4-position. The presence of additives like N-methylpyrrolidone (NMP) enhances the solubility and reactivity of the organocuprate species, ensuring high conversion rates even at moderate temperatures ranging from -78°C to reflux. This step effectively extends the carbon chain by three carbons, setting the stage for the subsequent introduction of the cleavable handle.
Following the initial chain extension, the intermediate ester or ketone (Compound 2) undergoes a second Grignard reaction with phenylmagnesium bromide. This step generates a tertiary alcohol (Compound 3) bearing two phenyl rings, which serves a dual purpose: it protects the carbonyl functionality and provides the necessary unsaturation for the final cleavage. The subsequent dehydration of this tertiary alcohol, catalyzed by reagents like trimethylchlorosilane or p-toluenesulfonic acid, yields a stilbene derivative (Compound 4). The final step employs powerful oxidative cleavage reagents, such as ozone or a potassium permanganate/sodium periodate system, to sever the double bond. This oxidative scission removes the diphenyl fragment as benzophenone or benzoic acid byproducts, leaving behind the desired (S)-3-methylheptanoic acid with the chiral methyl group intact and unaltered throughout the harsh oxidative conditions.
How to Synthesize (S)-3-Methylheptanoic Acid Efficiently
The practical implementation of this synthesis requires careful attention to moisture exclusion and temperature control, typical of organometallic chemistry. The process is divided into four distinct stages: the initial copper-mediated alkylation, the phenyl Grignard addition, the acid-catalyzed dehydration, and the final oxidative workup. Each step has been optimized in the patent examples to demonstrate robustness across different solvent systems, including THF, dichloromethane, and tert-butanol. For process engineers looking to adopt this technology, the detailed standardized synthesis steps provided below outline the specific molar ratios, reaction times, and quenching procedures necessary to achieve high yields and optical purity consistent with the patent data.
- Perform a copper-catalyzed Grignard addition of n-propylmagnesium bromide to the chiral lactone precursor (Compound 5) to form the ketone/ester intermediate (Compound 2).
- React the intermediate with phenylmagnesium bromide to introduce the diphenyl hydroxymethyl group, yielding the tertiary alcohol (Compound 3).
- Execute an acid-catalyzed dehydration of the tertiary alcohol to generate the stilbene derivative (Compound 4), followed by oxidative cleavage to release the final carboxylic acid.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, this technology offers compelling advantages by decoupling production from the volatile market of specialized chiral reagents. By utilizing a waste stream from the steroid industry as the primary feedstock, manufacturers can secure a low-cost, abundant source of chirality that is not subject to the same supply constraints as natural terpenes or synthetic chiral auxiliaries. This shift fundamentally alters the cost structure of the intermediate, potentially lowering the barrier to entry for generic prostaglandin manufacturers and enabling more competitive pricing for downstream API production. The elimination of stoichiometric chiral auxiliaries also reduces the material intensity of the process, leading to significant waste reduction and lower disposal costs associated with spent reagents.
- Cost Reduction in Manufacturing: The most significant economic driver of this process is the substitution of expensive chiral starting materials with value-added waste products. Traditional routes often incur high costs due to the purchase and recovery of chiral auxiliaries; this method bypasses that expense entirely. Furthermore, the reagents used—n-propyl bromide, bromobenzene, and magnesium—are commodity chemicals available in bulk quantities at stable prices. The streamlined workflow reduces the number of isolation and purification steps, which directly translates to lower labor costs, reduced solvent consumption, and decreased energy usage per kilogram of product, resulting in substantial overall cost savings for the manufacturing facility.
- Enhanced Supply Chain Reliability: Relying on waste derivatives from the massive steroidal sapogenin industry ensures a consistent and scalable supply of the chiral backbone. Unlike niche chiral pool materials that may suffer from harvest variability or geopolitical supply issues, the feedstock for this process is a byproduct of established large-scale operations. Additionally, the synthetic steps utilize robust, well-understood chemical transformations (Grignard reactions and oxidations) that can be performed in standard glass-lined or stainless steel reactors without the need for specialized exotic catalysts. This operational simplicity minimizes the risk of production delays caused by equipment incompatibility or the unavailability of specialized catalytic systems.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, utilizing solvents and reagents that are manageable in large-volume batch processes. The avoidance of heavy metal catalysts (other than trace copper which can be scavenged) and chromium-based oxidants aligns with increasingly stringent environmental regulations regarding heavy metal residues in pharmaceutical intermediates. The use of hydrogen peroxide in the upstream generation of the starting lactone and the potential for recycling solvent streams in the Grignard steps further enhance the environmental profile. This compliance reduces the regulatory burden on the supply chain head, facilitating smoother audits and faster approval times for new drug master files (DMFs).
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These insights are derived directly from the experimental data and claims within patent CN101274891A, providing a clear understanding of the process capabilities and limitations for potential licensees or manufacturing partners. Understanding these nuances is critical for R&D teams evaluating the feasibility of integrating this technology into their existing production pipelines.
Q: What is the primary advantage of this synthesis method over traditional chiral auxiliary routes?
A: Unlike traditional methods requiring expensive chiral auxiliaries like pseudoephedrine or complex resolution steps, this patent utilizes waste lactones from steroidal sapogenin degradation, significantly reducing raw material costs and simplifying the purification process.
Q: How is optical purity maintained during the Grignard reactions?
A: The optical purity is inherent to the starting material, (R)-γ-methyl-δ-valerolactone, which is derived from natural steroidal sapogenins. The subsequent Grignard additions and dehydration steps proceed without racemization at the chiral center, preserving the stereochemical integrity required for prostaglandin synthesis.
Q: Can this process be scaled for industrial production of prostaglandin intermediates?
A: Yes, the process relies on standard unit operations such as Grignard formation, extraction, and column chromatography or crystallization. The use of common reagents like n-propyl bromide and bromobenzene ensures that supply chains are robust and scalable for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (S)-3-Methylheptanoic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of efficient chiral synthesis in the development of next-generation therapeutics. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from laboratory bench to industrial plant is seamless and compliant. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including chiral HPLC analysis to guarantee the optical integrity of intermediates like (S)-3-methylheptanoic acid. We are committed to delivering high-quality pharmaceutical intermediates that meet the exacting standards of global regulatory bodies.
We invite you to collaborate with our technical team to explore how this waste-valorization technology can optimize your supply chain and reduce manufacturing costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to support your project timelines and help you secure a reliable supply of this critical prostaglandin precursor.
