Advanced Synthesis of Orange Light-Emitting Platinum Complexes for WOLED Applications
The rapid evolution of the organic light-emitting diode (OLED) industry has placed immense pressure on material suppliers to deliver compounds that offer both superior photophysical performance and manufacturability. Patent CN108504351B introduces a groundbreaking orange light-emitting material, specifically a platinum alkynyl complex with the molecular formula C41H28N6Pt. This compound represents a significant advancement in the field of electroluminescent materials, addressing the critical need for stable, high-efficiency orange emitters required to construct high-quality White Organic Light-Emitting Devices (WOLEDs). The invention details a robust synthetic pathway that leverages the unique optical activity of Pt(II) ions and conjugated pi-systems to facilitate efficient electron transition and energy transfer. For R&D directors and procurement specialists alike, this technology offers a compelling value proposition by combining precise molecular architecture with a scalable production process.
In the context of global energy shortages, WOLEDs have emerged as a superior alternative to traditional lighting sources due to their flexibility, large-area potential, and lower manufacturing costs. However, the performance of these devices is heavily dependent on the quality of the emissive layers. The platinum complex described in this patent emits strong orange light with a maximum emission wavelength centered at 610nm, making it an ideal candidate for balancing the spectral output of white light panels. By integrating this material, manufacturers can achieve better color rendering and higher luminous efficiency, directly impacting the commercial viability of next-generation display and照明 technologies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of platinum-based phosphorescent emitters has been plagued by several significant challenges that hinder commercial adoption. Traditional routes often involve multi-step reactions requiring extreme temperatures, prolonged reaction times, and the use of hazardous solvents that complicate waste management and increase operational costs. Furthermore, conventional methods frequently suffer from poor regioselectivity, leading to complex mixtures of isomers that are difficult to separate. This lack of selectivity not only reduces the overall yield of the desired product but also introduces impurities that can act as quenching sites, severely degrading the luminescent efficiency of the final OLED device. Additionally, many existing protocols rely on expensive catalysts or precursors that are not readily available on a bulk scale, creating supply chain bottlenecks for large-scale manufacturing.
The Novel Approach
The methodology outlined in patent CN108504351B overcomes these historical barriers through a streamlined, three-step synthesis that prioritizes mild conditions and high selectivity. The process begins with the dimerization of 2-(2-pyridyl)benzimidazole under phase transfer catalysis conditions, utilizing potassium hydroxide and tetrabutylammonium bromide in dichloromethane at room temperature. This gentle approach minimizes side reactions and ensures the formation of the ligand precursor (1h) with high purity. Subsequent coordination with platinum and final alkynylation are conducted under controlled, moderate thermal conditions, avoiding the degradation issues common in high-temperature syntheses. This novel route not only simplifies the operational workflow but also significantly enhances the reproducibility of the material, a critical factor for industrial consistency.
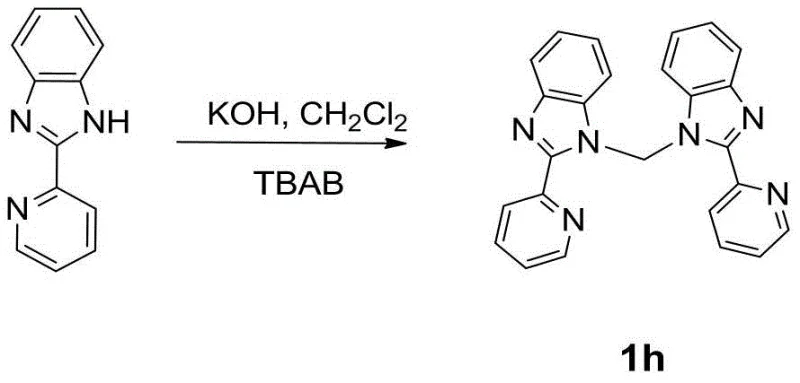
The first stage of this innovative process involves the efficient coupling of benzimidazole derivatives to form the bridging ligand structure. As illustrated in the reaction scheme, the use of dichloromethane as a solvent combined with phase transfer agents allows for effective mixing of the inorganic base and organic substrates. This results in a clean conversion to intermediate 1h, which serves as the foundational scaffold for the platinum complex. The simplicity of the workup procedure, involving solvent evaporation and column chromatography, demonstrates the practical feasibility of this method for scaling up production without requiring specialized high-pressure equipment.
Mechanistic Insights into Pt(II) Coordination and Alkynylation
The core of this technology lies in the precise construction of the metal-ligand environment around the platinum center. The synthesis proceeds through a well-defined coordination mechanism where the nitrogen atoms of the benzimidazole and pyridine rings chelate the Pt(II) ion. This N^N bidentate coordination creates a rigid planar structure that restricts non-radiative decay pathways, thereby enhancing the phosphorescence quantum yield. The introduction of the alkynyl groups in the final step further extends the conjugation of the system. This extended pi-conjugation is crucial for tuning the HOMO-LUMO energy gap, allowing the material to emit in the desirable orange region of the spectrum. The high spin-orbit coupling inherent to the heavy platinum atom facilitates intersystem crossing, enabling the harvesting of both singlet and triplet excitons for theoretically 100% internal quantum efficiency.
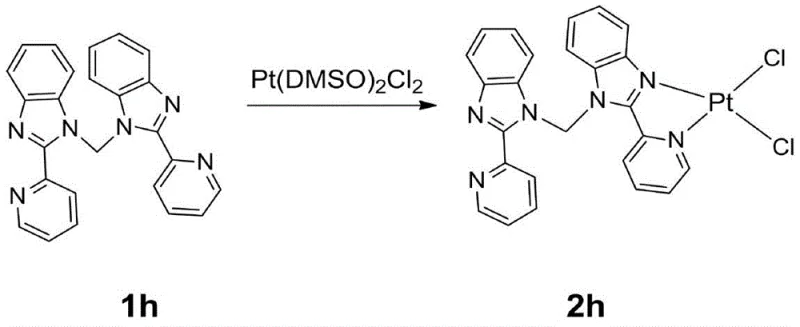
Following the ligand formation, the coordination step involves reacting the intermediate 1h with Pt(DMSO)2Cl2. This substitution reaction replaces the labile DMSO ligands with the rigid organic framework, locking the platinum into a specific geometry that is essential for its optical properties. The resulting intermediate 2h retains chloride ligands which are subsequently displaced in the final step. This modular approach allows for fine-tuning of the electronic properties by varying the alkyne component, although the patent specifically exemplifies the use of phenylacetylene to achieve the target orange emission profile. The mechanistic clarity of this pathway ensures that impurities are minimized, as each step produces distinct, separable intermediates.
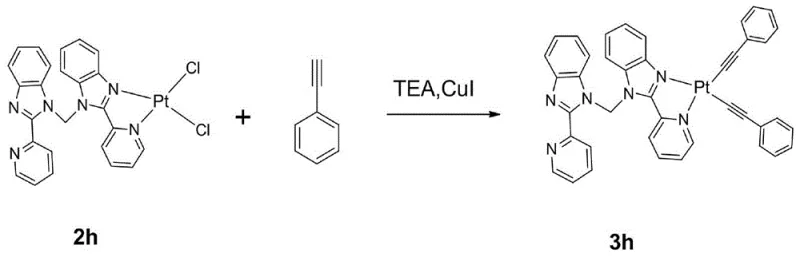
The final transformation utilizes a copper-catalyzed cross-coupling reaction, akin to a Sonogashira coupling, to install the phenylacetylene moieties. In this step, the chloride ligands on the platinum center are replaced by the alkynyl groups in the presence of triethylamine and cuprous iodide. This reaction is performed under reflux conditions in dichloromethane, ensuring complete conversion while maintaining the integrity of the sensitive platinum complex. The resulting product, 3h, possesses a definitive spatial structure confirmed by X-ray diffraction, exhibiting a triclinic crystal system. This structural precision is vital for ensuring consistent packing in the solid state, which influences the charge transport properties and overall device performance in OLED applications.
How to Synthesize N-[(2-pyridyl)benzimidazole]methylene-bi(2-pyridyl)benzimidazole Platinum (Diphenylacetylene) Complex Efficiently
To replicate the high-purity standards required for electronic grade materials, strict adherence to the patented protocol is essential. The synthesis is divided into three distinct stages, each optimized for yield and purity. The initial ligand synthesis requires careful control of the phase transfer conditions to ensure complete deprotonation and coupling. The subsequent metallation step demands stoichiometric precision to avoid the formation of mono-coordinated species. Finally, the alkynylation requires anhydrous conditions and precise temperature control to prevent homocoupling of the alkyne. Detailed standard operating procedures for each of these critical stages are provided below to guide process engineers in implementing this technology.
- Synthesize the bis-benzimidazole ligand (1h) by reacting 2-(2-pyridyl)benzimidazole with KOH and TBAB in dichloromethane at room temperature.
- Coordinate the ligand with Pt(DMSO)2Cl2 in dichloromethane to form the chloro-bridged platinum intermediate (2h).
- Perform a copper-catalyzed alkynylation using phenylacetylene and triethylamine to replace chloride ligands, yielding the final orange-emitting complex (3h).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis route offers tangible strategic benefits beyond mere technical performance. The reliance on commercially available starting materials, such as 2-(2-pyridyl)benzimidazole and phenylacetylene, mitigates the risk of raw material shortages. Furthermore, the use of dichloromethane, a ubiquitous industrial solvent, simplifies solvent recovery and recycling processes, aligning with modern sustainability goals. The mild reaction conditions reduce energy consumption significantly compared to high-temperature alternatives, contributing to a lower carbon footprint for the manufacturing process. These factors collectively enhance the resilience of the supply chain against market volatility.
- Cost Reduction in Manufacturing: The streamlined nature of this synthesis directly translates to reduced operational expenditures. By eliminating the need for exotic catalysts or extreme reaction conditions, the process lowers both capital investment and utility costs. The high selectivity of the reactions minimizes the formation of by-products, which reduces the burden on purification systems and decreases solvent usage during chromatography. Additionally, the ability to perform key steps at room temperature removes the energy costs associated with heating and cooling large reactors, resulting in substantial cost savings in electronic chemical manufacturing.
- Enhanced Supply Chain Reliability: The robustness of this chemical pathway ensures consistent production output, which is critical for maintaining just-in-time delivery schedules for display manufacturers. The intermediates generated in this process are stable and can be stockpiled if necessary, providing a buffer against demand fluctuations. Moreover, the simplicity of the workup procedures—primarily involving filtration and evaporation—allows for faster batch turnover times. This efficiency enables suppliers to respond more agilely to customer orders, reducing lead time for high-purity OLED materials and strengthening partnerships with downstream device fabricators.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is facilitated by the absence of hazardous reagents or dangerous exothermic events. The use of standard organic solvents allows for the utilization of existing infrastructure in fine chemical plants without major retrofitting. From an environmental perspective, the high atom economy of the coupling reactions and the potential for solvent recycling support compliance with increasingly stringent environmental regulations. This scalability ensures that the material can be produced in metric ton quantities to meet the growing global demand for energy-efficient lighting and display solutions.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this material is essential for stakeholders evaluating its integration into their product lines. The following questions address common inquiries regarding the material's performance, stability, and production feasibility. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a reliable basis for decision-making.
Q: Why is this specific orange emitter critical for White OLED (WOLED) development?
A: White OLEDs typically require a combination of blue, green, and red/orange emitters to achieve high-quality white light. This platinum complex provides a stable, high-efficiency orange component with a maximum emission wavelength around 610nm, which is essential for balancing the color spectrum and improving the Color Rendering Index (CRI) in display and lighting applications.
Q: What are the thermal stability characteristics of this platinum alkynyl material?
A: The material exhibits exceptional thermal stability with a melting point exceeding 300°C. This high thermal decomposition threshold ensures that the material can withstand the rigorous processing conditions often encountered during vacuum deposition or solution processing in device fabrication without degrading.
Q: How does the patented synthesis method improve upon traditional platinum complex preparation?
A: Unlike traditional methods that may require harsh temperatures or toxic solvents, this patented route utilizes mild conditions (room temperature to 70°C) and common solvents like dichloromethane. The use of phase transfer catalysis in the first step significantly enhances reaction efficiency and selectivity, leading to higher purity intermediates and a simplified downstream purification process.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Platinum Alkynyl Complex Supplier
As the demand for high-performance optoelectronic materials continues to surge, partnering with an experienced CDMO is crucial for success. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and reliability. Our facility is equipped with rigorous QC labs capable of verifying stringent purity specifications, including trace metal analysis and thermal stability testing, which are paramount for OLED applications. We understand the critical nature of electronic chemicals and are committed to delivering materials that consistently meet the highest industry standards.
We invite you to collaborate with us to leverage this advanced technology for your next-generation display projects. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and process constraints. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you optimize your supply chain and accelerate your time-to-market with our premium orange light-emitting materials.
