Advanced Catalytic Dehydration and Cyclization for Industrial Gamma-Decalactone Production
The global demand for high-quality flavor and fragrance ingredients continues to drive innovation in synthetic organic chemistry, particularly for lactones like gamma-decalactone which impart desirable peach and coconut notes. Patent CN111747910B introduces a significant technological advancement in this sector by detailing a robust preparation method utilizing 3-hydroxydecanoic acid as the primary precursor. This approach distinguishes itself through a sophisticated integration of acid-catalyzed dehydration and continuous product removal via a stripping agent, effectively addressing long-standing challenges in yield optimization and impurity control. By leveraging a bio-based starting material, the process not only aligns with the growing market preference for natural-identical ingredients but also streamlines the synthetic pathway to minimize environmental impact. For industry stakeholders, this represents a pivotal shift towards more sustainable and efficient manufacturing protocols that can be reliably scaled for commercial production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of gamma-decalactone has relied on several distinct pathways, each fraught with significant technical and economic drawbacks that hinder optimal production efficiency. Traditional methods often involve the co-heating of omega-decenoic acid with sulfuric acid, a process notorious for generating excessive byproducts due to double bond migration and non-selective lactonization, resulting in poor overall yields and difficult purification steps. Alternative routes utilizing high-valence metal acetates as oxidants introduce severe environmental liabilities, generating large volumes of wastewater contaminated with heavy metals that require costly treatment and disposal procedures. Furthermore, radical addition reactions between n-heptanol and acrylic acid, while capable of scale, frequently suffer from the accumulation of low and high boiling point impurities that detrimentally affect the sensory profile of the final fragrance. These legacy processes often struggle with selectivity issues, leading to complex mixtures that compromise the delicate aroma quality required by discerning flavor and fragrance applications.
The Novel Approach
In stark contrast to these conventional limitations, the methodology described in patent CN111747910B offers a streamlined and chemically elegant solution centered on the dehydration and cyclization of 3-hydroxydecanoic acid. This novel approach capitalizes on the specific reactivity of the hydroxy acid under acidic conditions to generate key unsaturated intermediates which subsequently cyclize into the target lactone. A critical innovation lies in the continuous introduction of a stripping agent, specifically water, which serves to continuously extract the formed gamma-decalactone from the reaction matrix. This dynamic removal prevents the product from undergoing reverse reactions or degradation within the harsh acidic environment, thereby driving the equilibrium forward and significantly enhancing conversion rates. By avoiding heavy metal catalysts and hazardous radical initiators, this process inherently reduces the generation of toxic waste streams, positioning it as a superior choice for modern green chemistry initiatives in the fine chemical industry.
Mechanistic Insights into Acid-Catalyzed Dehydration and Cyclization
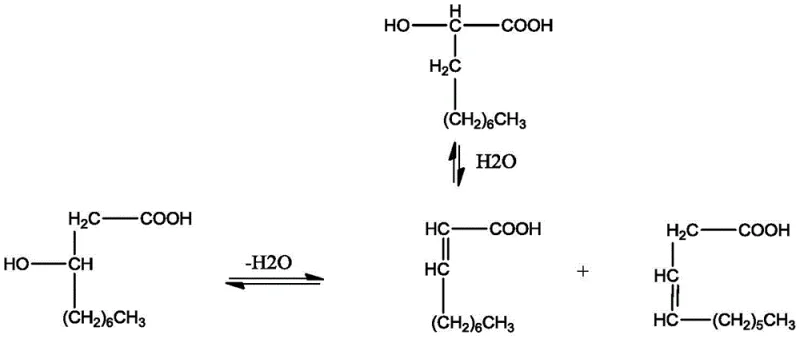
The core of this synthesis relies on a precise sequence of acid-catalyzed transformations where 3-hydroxydecanoic acid first undergoes dehydration to form a mixture of beta,gamma-unsaturated decenoic acid and alpha,beta-unsaturated decenoic acid. The beta,gamma-isomer is the direct precursor to the desired lactone, undergoing an intramolecular cyclization to close the five-membered ring structure characteristic of gamma-decalactone. However, the reaction landscape is complex, as the alpha,beta-unsaturated isomer does not directly cyclize to the target product and can instead revert to hydroxy acids via hydration, creating a dynamic equilibrium that must be carefully managed. The presence of strong acid catalysts like sulfuric or phosphoric acid facilitates these proton transfers and elimination steps, but also poses a risk of promoting polymerization of the highly reactive double bonds present in the unsaturated intermediates.
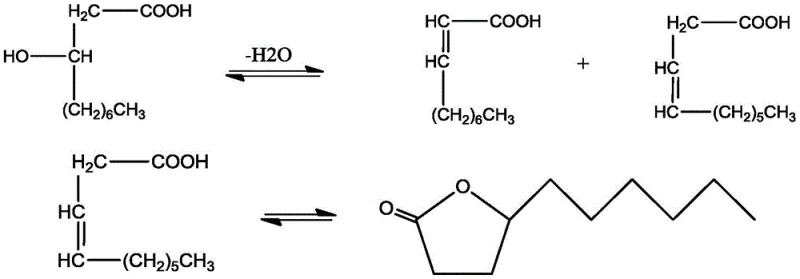
To mitigate these side reactions and maximize the yield of the desired beta,gamma-isomer, the process employs specific auxiliary agents such as o-nitrophenol or nitrobenzene which act as stabilizers against polymerization. The mechanistic pathway is further complicated by the reversible nature of the hydration-dehydration steps, where the alpha,beta-unsaturated acid can hydrate to form 2-hydroxydecanoic acid, which may then dehydrate back to the alpha,beta-isomer rather than progressing to the lactone. Understanding this intricate network of equilibria is crucial for optimizing reaction conditions, as the goal is to funnel the maximum amount of starting material through the beta,gamma-pathway while suppressing the formation of polymeric tars. The continuous stripping of the product effectively breaks these equilibria by removing the gamma-decalactone as soon as it forms, preventing it from participating in any reverse hydrolysis reactions that would lower the net yield.
How to Synthesize Gamma-Decalactone Efficiently
Executing this synthesis requires careful control of reaction parameters to balance the rate of dehydration with the efficiency of product removal. The process begins by charging a reactor with 3-hydroxydecanoic acid, a controlled amount of water, and a mineral acid catalyst, along with a small quantity of a polymerization inhibitor to ensure reaction stability. As the mixture is heated to the optimal temperature range of 150-200°C, the dehydration reaction initiates, and the continuous addition of water as a stripping agent is commenced to maintain a constant liquid level while carrying the volatile lactone out of the reactor. Detailed standardized synthetic steps for implementing this continuous stripping protocol are provided in the guide below to ensure reproducibility and safety during scale-up operations.
- Charge the reactor with 3-hydroxydecanoic acid, water, an acid catalyst like sulfuric acid, and a polymerization inhibitor.
- Heat the mixture to 150-200°C and initiate the continuous addition of water as a stripping agent to maintain liquid level.
- Collect the condensate containing gamma-decalactone, separate the oil phase, and purify to obtain the final high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented technology offers tangible benefits that extend beyond mere chemical yield, impacting the overall cost structure and reliability of the supply base. The shift towards a bio-based starting material like 3-hydroxydecanoic acid not only enhances the marketability of the final fragrance ingredient as natural-identical but also diversifies the raw material sourcing away from purely petrochemical feedstocks which are subject to volatile pricing fluctuations. By eliminating the need for expensive and environmentally burdensome heavy metal oxidants or specialized radical initiators, the process significantly simplifies the waste treatment infrastructure required at the manufacturing site. This reduction in hazardous waste generation translates directly into lower operational expenditures related to environmental compliance and disposal, making the overall cost of goods sold more competitive in the global marketplace.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts and the avoidance of complex multi-step sequences found in Reformatsky or radical addition routes drastically simplify the production workflow. This simplification reduces the consumption of solvents and reagents, leading to substantial cost savings in raw material procurement and utility usage. Furthermore, the continuous nature of the product removal minimizes the residence time of materials in the reactor, allowing for higher throughput and better utilization of existing capital equipment without the need for extensive retrofitting.
- Enhanced Supply Chain Reliability: Utilizing 3-hydroxydecanoic acid, which can be sourced from established biological fermentation processes, provides a stable and renewable supply chain foundation that is less susceptible to the geopolitical and logistical disruptions often associated with petroleum derivatives. The robustness of the acid-catalyzed chemistry ensures consistent batch-to-batch quality, reducing the risk of production delays caused by off-spec material that requires reprocessing. This reliability is critical for maintaining uninterrupted supply to downstream customers in the food and beverage industry who operate on tight just-in-time inventory schedules.
- Scalability and Environmental Compliance: The process design inherently supports easy scale-up from pilot to commercial production due to its reliance on standard unit operations like distillation and stirred-tank reaction. The significant reduction in three wastes (wastewater, waste gas, and solid waste) aligns perfectly with increasingly stringent global environmental regulations, future-proofing the manufacturing asset against tighter emission standards. This environmental stewardship not only mitigates regulatory risk but also enhances the brand reputation of the supplier as a responsible partner in sustainable chemistry.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this gamma-decalactone synthesis method. These insights are derived directly from the experimental data and process descriptions within the patent documentation to provide clarity on operational feasibility. Understanding these nuances is essential for technical teams evaluating the integration of this technology into their existing manufacturing portfolios.
Q: What are the advantages of using 3-hydroxydecanoic acid over traditional petrochemical routes?
A: Using 3-hydroxydecanoic acid, often derived from biological fermentation, allows for the production of bio-based gamma-decalactone which possesses a superior, more natural fragrance profile compared to petrochemical equivalents. Additionally, this route avoids the use of heavy metal oxidants or hazardous radical initiators found in older methods.
Q: How does the continuous water stripping method improve yield?
A: The continuous addition of water as a stripping agent facilitates the removal of gamma-decalactone from the reaction zone as it forms. This shifts the chemical equilibrium towards product formation according to Le Chatelier's principle and prevents the degradation or ring-opening of the lactone that can occur if it remains in the acidic reaction medium for too long.
Q: What role do auxiliary agents play in this synthesis?
A: Auxiliary agents such as o-nitrophenol or nitrobenzene are critical for suppressing side reactions. The intermediate unsaturated acids are highly reactive and prone to polymerization, which leads to viscous byproducts and lower yields. These additives stabilize the intermediates, ensuring a cleaner reaction profile and easier downstream purification.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Gamma-Decalactone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of delivering high-purity flavor ingredients that meet the exacting standards of the global food and fragrance industries. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory processes like the one described in CN111747910B can be successfully translated into robust industrial operations. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of gamma-decalactone delivers the consistent peach and coconut aroma profile that your formulations demand. Our commitment to quality assurance ensures that the transition from petrochemical to bio-based precursors is seamless and reliable for your supply chain.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can be tailored to your specific volume requirements and cost targets. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits of switching to this greener manufacturing method. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive value and sustainability in your product lines.
