Revolutionizing Tricyclic Derivative Production: A Safe, Scalable Route for High-Purity API Intermediates
Revolutionizing Tricyclic Derivative Production: A Safe, Scalable Route for High-Purity API Intermediates
The pharmaceutical industry constantly seeks robust manufacturing pathways for complex anticancer agents, particularly tricyclic derivatives known for their potent cytotoxicity against cancer cell lines and strong angiogenesis inhibition. Patent CN103772193A discloses a groundbreaking methodology for preparing these critical tricyclic derivatives and their key intermediates with exceptional yield and purity. Unlike traditional synthetic routes that rely on hazardous reagents and inefficient purification techniques, this invention introduces a chemically elegant solution that prioritizes industrial safety and economic feasibility. By replacing dangerous lithium borohydride with safer alternatives and substituting column chromatography with scalable recrystallization, this technology offers a reliable pharmaceutical intermediate supplier with a distinct competitive edge in the global market.
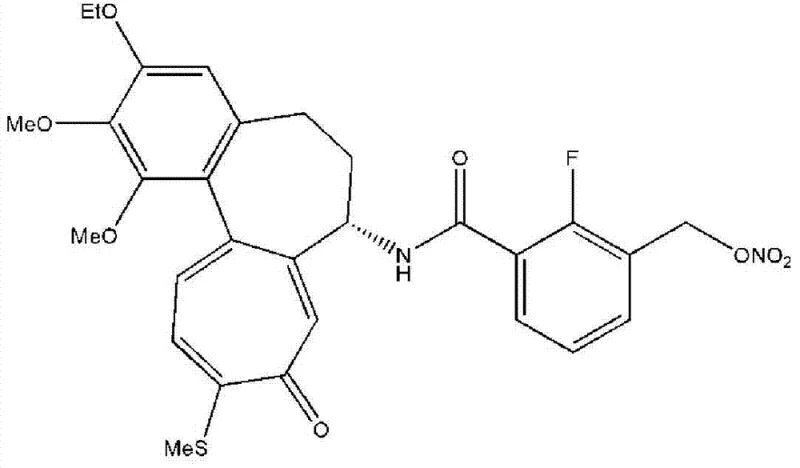
This technological advancement addresses the critical bottlenecks in the commercial scale-up of complex pharmaceutical intermediates. The patent details a multi-step synthesis starting from readily available 2-fluoroisophthalic acid, transforming it into high-value intermediates like 2-fluoro-3-(hydroxymethyl)benzoic acid. For R&D directors and procurement managers, understanding the nuances of this pathway is essential, as it directly impacts the cost of goods sold (COGS) and the reliability of the supply chain for downstream API manufacturing. The following analysis dissects the mechanistic innovations and commercial implications of this proprietary process.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of key intermediates for tricyclic derivatives has been plagued by significant safety hazards and low efficiency. As illustrated in the prior art reaction schemes, conventional methods often utilize lithium borohydride (LiBH4) as the primary reducing agent to convert ester groups into hydroxymethyl functionalities. While chemically effective on a small laboratory scale, LiBH4 poses severe risks in an industrial setting due to its violent reaction with water, which generates flammable hydrogen gas and creates a high probability of fire or explosion. Furthermore, traditional purification strategies heavily depend on silica gel column chromatography. Although effective for isolating milligrams of material for analysis, column chromatography is economically prohibitive for ton-scale production due to the excessive consumption of expensive silica and large volumes of organic solvents, leading to substantial waste disposal costs and environmental compliance issues.
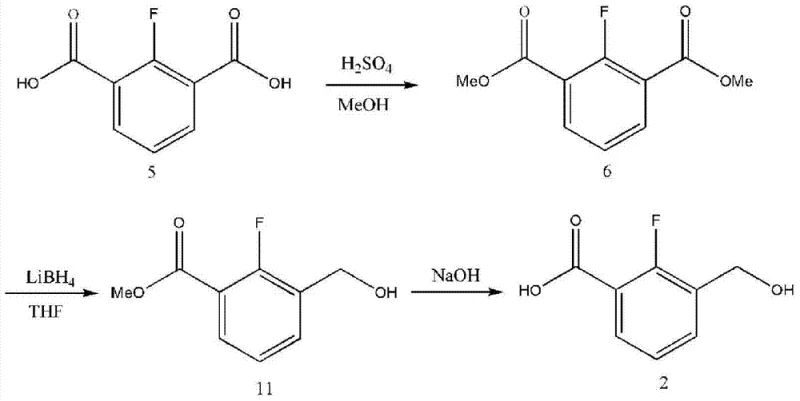
The Novel Approach
The methodology described in patent CN103772193A fundamentally re-engineers the synthetic route to eliminate these industrial barriers. The novel approach replaces the hazardous lithium borohydride with sodium borohydride (NaBH4) or lithium aluminum hydride (LiAlH4), reagents that offer a much lower fire risk and are manageable in large-scale reactors. Crucially, the invention introduces a strategic protection-deprotection sequence using piperidine to form an amide bond. This structural modification prevents the non-selective reduction of both ester groups, a common side reaction when using standard hydride reducers on diesters. Additionally, the process abandons column chromatography entirely in favor of recrystallization techniques using solvent systems like acetonitrile and ethyl acetate. This shift not only enhances the purity of the final high-purity pharmaceutical intermediate to levels exceeding 99% but also drastically simplifies the workflow, making it ideally suited for continuous manufacturing and large-batch processing.
Mechanistic Insights into Selective Ester Reduction via Amide Protection
The core chemical innovation lies in the precise control of chemoselectivity during the reduction phase. In the synthesis of 2-fluoro-3-(hydroxymethyl)benzoic acid, the starting material, 2-fluoroisophthalic acid, contains two carboxylic acid groups. Direct reduction would typically lead to a diol or a mixture of products. The patented process first converts the diacid into a mono-ester (Compound 10) and then transforms the remaining free acid into a piperidine amide (Compound 3). This amide functionality is robust against mild hydride reduction conditions. When sodium borohydride is introduced, it selectively reduces the methyl ester moiety to the primary alcohol (hydroxymethyl group) while leaving the amide bond intact. This selectivity is the key to achieving high yields, reported at approximately 94.4% in the reduction step, compared to the significantly lower yields of unprotected routes.
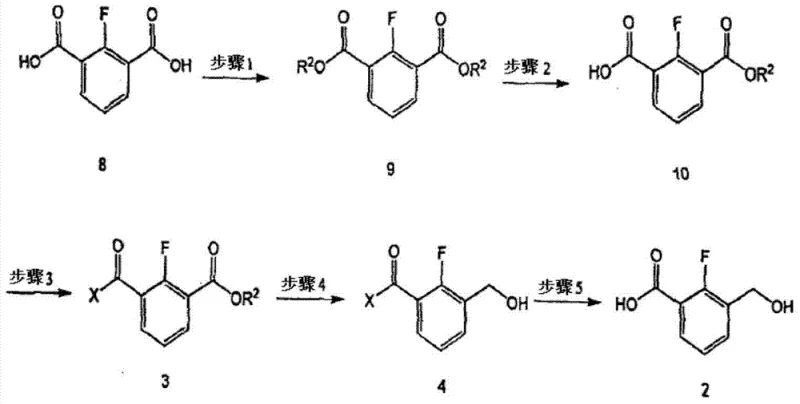
Following the selective reduction, the final step involves the hydrolysis of the piperidine amide under basic conditions (using NaOH or KOH) to regenerate the carboxylic acid, yielding the target intermediate (Compound 2). This sequence ensures that the sensitive hydroxymethyl group is not compromised during the final deprotection. The mechanism demonstrates a sophisticated understanding of functional group tolerance, allowing for the use of cheaper, safer reagents without sacrificing the structural integrity of the molecule. For process chemists, this route represents a paradigm shift from relying on exotic, dangerous reagents to optimizing standard unit operations through clever molecular design.
How to Synthesize 2-Fluoro-3-(hydroxymethyl)benzoic Acid Efficiently
The synthesis of this critical intermediate is achieved through a streamlined five-step sequence that maximizes atom economy and operational safety. The process begins with the Fischer esterification of 2-fluoroisophthalic acid, followed by a controlled partial hydrolysis to generate the mono-ester. Subsequent amidation with piperidine sets the stage for the pivotal reduction step. Detailed standardized operating procedures, including specific solvent ratios, temperature controls, and workup protocols for each transformation, are essential for replicating the high purity and yield reported in the patent data.
- Esterify 2-fluoroisophthalic acid with methanol using sulfuric acid catalyst to form dimethyl 2-fluoroisophthalate.
- Perform selective hydrolysis using potassium hydroxide to obtain mono-ester, followed by amidation with piperidine to protect the carboxylic acid.
- Reduce the remaining ester group using sodium borohydride (NaBH4) to introduce the hydroxymethyl group, then hydrolyze the amide to release the final acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition from the conventional LiBH4 route to this novel NaBH4/recrystallization protocol offers profound economic and logistical benefits. The elimination of pyrophoric reagents reduces the need for specialized containment infrastructure and lowers insurance premiums associated with hazardous chemical storage. Furthermore, the switch from column chromatography to recrystallization significantly reduces the consumption of silica gel and organic solvents, leading to substantial cost savings in raw materials and waste treatment. This process optimization translates directly into a more competitive pricing structure for the final API intermediate, enhancing the margin potential for downstream drug manufacturers.
- Cost Reduction in Manufacturing: The replacement of lithium borohydride with sodium borohydride eliminates the need for expensive, specialty reducing agents that require strict anhydrous conditions. Additionally, the removal of silica gel column chromatography removes a major cost center; silica is expensive, and its disposal adds to environmental fees. By utilizing simple recrystallization, the process minimizes solvent usage and labor hours, resulting in a drastically simplified production cost profile without compromising quality.
- Enhanced Supply Chain Reliability: Safety is a critical component of supply continuity. Processes involving high-fire-risk reagents like LiBH4 are prone to regulatory shutdowns or accidents that can halt production for months. By adopting a safer chemistry with NaBH4, the manufacturing facility ensures consistent uptime and reduces the risk of force majeure events. The use of commodity chemicals (methanol, piperidine, NaBH4) also ensures that raw material sourcing remains stable and unaffected by the supply volatility often seen with niche specialty reagents.
- Scalability and Environmental Compliance: The ability to purify intermediates via recrystallization rather than chromatography is the defining factor for scalability. Chromatography is notoriously difficult to scale beyond pilot plant sizes, whereas crystallization is a standard unit operation in multi-ton reactors. This scalability ensures that the commercial scale-up of complex pharmaceutical intermediates can proceed smoothly from kilogram to metric ton quantities. Moreover, the reduced solvent waste aligns with modern green chemistry principles, facilitating easier compliance with increasingly stringent environmental regulations regarding VOC emissions and hazardous waste disposal.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the experimental data and beneficial effects described in patent CN103772193A, providing clarity on how this technology outperforms legacy methods in terms of safety, yield, and purity.
Q: Why is the new reduction method safer than conventional lithium borohydride processes?
A: Conventional methods use lithium borohydride (LiBH4), which reacts violently with water producing flammable gases. The patented method utilizes sodium borohydride (NaBH4) or lithium aluminum hydride (LiAlH4), which significantly lowers fire risks and is suitable for industrial mass production.
Q: How does this process improve purity compared to column chromatography?
A: Instead of using expensive and waste-generating silica gel column chromatography, this method employs recrystallization (e.g., using acetonitrile/ethyl acetate mixtures). This achieves high purity (up to 99.8% in final steps) while drastically reducing solvent waste and processing time.
Q: What is the key chemical strategy to prevent over-reduction of ester groups?
A: The process introduces a piperidinyl amide group prior to reduction. This amide bond protects one carboxyl function, allowing the reducing agent to selectively target only the remaining ester group to form the hydroxymethyl moiety without reducing the entire molecule.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tricyclic Derivatives Supplier
The synthesis of tricyclic derivatives represents a high-barrier entry point in oncology drug development, requiring partners who possess both deep chemical expertise and robust manufacturing capabilities. NINGBO INNO PHARMCHEM stands at the forefront of this sector, leveraging advanced process technologies like the one described in CN103772193A to deliver superior results. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions seamlessly from R&D to market. We maintain stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of high-purity pharmaceutical intermediate meets the exacting standards required for clinical and commercial API synthesis.
We invite you to collaborate with us to optimize your supply chain and reduce your manufacturing costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our optimized synthesis of tricyclic derivatives can accelerate your drug development timeline while maximizing economic efficiency.
