Advanced Alkylation Route for High-Purity PPARα Modulator Intermediates and Commercial Scale-Up
Advanced Alkylation Route for High-Purity PPARα Modulator Intermediates and Commercial Scale-Up
The pharmaceutical industry's relentless pursuit of effective treatments for metabolic syndrome, particularly Type 2 diabetes and associated cardiovascular risks, has placed significant emphasis on the development of potent PPARα modulators. Patent CN1809528A discloses a groundbreaking methodology for the preparation of (2S)-3-(4-{2-[amino]-2-oxoethoxy}phenyl)-2-ethoxypropionic acid derivatives, which serve as critical intermediates in this therapeutic class. This technical disclosure represents a paradigm shift from traditional peptide coupling strategies to a more efficient direct N-alkylation approach. For R&D directors and procurement specialists, understanding the nuances of this synthetic route is essential for securing a reliable pharmaceutical intermediate supplier capable of delivering high-purity materials. The patent details a robust process that not only enhances chemical efficiency but also addresses key supply chain vulnerabilities associated with complex amide bond formations. By leveraging specific reaction conditions involving polar aprotic solvents and inorganic bases, this technology enables the production of key drug substances with superior impurity profiles and stereochemical integrity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of complex amide-containing intermediates like those described in the background art relied heavily on activated coupling protocols. As illustrated in the prior art pathways, the conventional method typically involves reacting an S-enantiomer of a carboxylic acid derivative with an amine component in the presence of coupling agents such as carbodiimides (e.g., EDC or DCC). 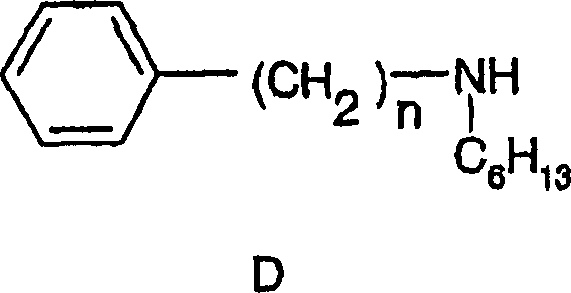 This traditional approach, while chemically valid, introduces significant logistical and economic burdens for large-scale manufacturing. The use of stoichiometric coupling reagents generates substantial amounts of urea byproducts, which are notoriously difficult to remove completely from the final API intermediate, often requiring extensive chromatographic purification. Furthermore, the cost of activating agents and additives like HOBt or DMAP can drastically inflate the bill of materials. From a safety and environmental perspective, the accumulation of these byproducts complicates waste stream management and increases the E-factor of the process. For procurement managers, reliance on such chemistry means exposure to volatile pricing of specialized reagents and potential supply bottlenecks for high-purity coupling agents.
This traditional approach, while chemically valid, introduces significant logistical and economic burdens for large-scale manufacturing. The use of stoichiometric coupling reagents generates substantial amounts of urea byproducts, which are notoriously difficult to remove completely from the final API intermediate, often requiring extensive chromatographic purification. Furthermore, the cost of activating agents and additives like HOBt or DMAP can drastically inflate the bill of materials. From a safety and environmental perspective, the accumulation of these byproducts complicates waste stream management and increases the E-factor of the process. For procurement managers, reliance on such chemistry means exposure to volatile pricing of specialized reagents and potential supply bottlenecks for high-purity coupling agents.
The Novel Approach
In stark contrast, the invention disclosed in CN1809528A introduces a streamlined alkylation strategy that bypasses the need for activation entirely. The core innovation lies in the direct reaction of a secondary amide precursor (Formula II) with an alkylating agent (Formula III, where X is a leaving group) under basic conditions. 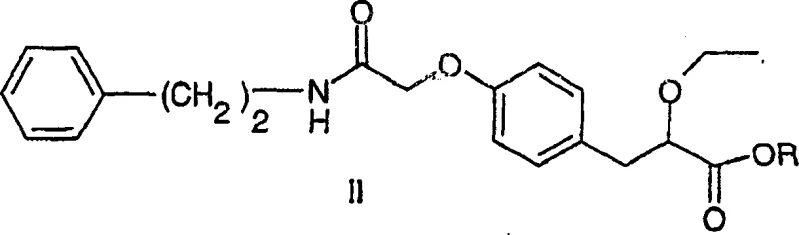 This method utilizes simple inorganic bases such as potassium hydroxide or sodium hydride in inert solvents like dimethylsulfoxide (DMSO) or N-methylpyrrolidone (NMP). By shifting the mechanism from nucleophilic acyl substitution to nucleophilic substitution at an alkyl carbon, the process eliminates the formation of difficult-to-remove urea wastes. The reaction operates effectively across a broad temperature range of -25°C to 150°C, offering flexibility for process optimization. This transition represents a significant cost reduction in pharmaceutical intermediate manufacturing, as alkyl halides are generally far cheaper and more readily available than peptide coupling reagents. Additionally, the simplified workup procedure, often involving straightforward extraction and crystallization, enhances the overall throughput and reduces the operational complexity for contract development and manufacturing organizations (CDMOs).
This method utilizes simple inorganic bases such as potassium hydroxide or sodium hydride in inert solvents like dimethylsulfoxide (DMSO) or N-methylpyrrolidone (NMP). By shifting the mechanism from nucleophilic acyl substitution to nucleophilic substitution at an alkyl carbon, the process eliminates the formation of difficult-to-remove urea wastes. The reaction operates effectively across a broad temperature range of -25°C to 150°C, offering flexibility for process optimization. This transition represents a significant cost reduction in pharmaceutical intermediate manufacturing, as alkyl halides are generally far cheaper and more readily available than peptide coupling reagents. Additionally, the simplified workup procedure, often involving straightforward extraction and crystallization, enhances the overall throughput and reduces the operational complexity for contract development and manufacturing organizations (CDMOs).
Mechanistic Insights into Base-Mediated N-Alkylation
The mechanistic elegance of this process centers on the generation of a highly nucleophilic amide anion. In the presence of a strong base like potassium hydroxide in a polar aprotic solvent such as DMSO, the acidic proton on the nitrogen atom of the acetamide moiety is abstracted. This deprotonation creates a resonance-stabilized anion that retains sufficient nucleophilicity to attack the electrophilic carbon of the alkyl halide (e.g., 1-bromohexane). The choice of solvent is critical; DMSO not only solubilizes the inorganic base and the organic substrate but also enhances the reactivity of the anion by minimizing solvation shells, thereby accelerating the SN2 reaction kinetics. Crucially, the reaction conditions are mild enough to preserve the stereochemical integrity of the adjacent chiral center at the 2-position of the propionic acid chain. The patent data confirms that the (2S)-enantiomer is maintained throughout the transformation, yielding products with high enantiomeric excess (e.e.), which is a non-negotiable requirement for biological efficacy in PPAR modulation.
Impurity control is another vital aspect of this mechanistic pathway. Unlike coupling reactions that can lead to racemization via oxazolone formation, this alkylation route minimizes such risks. The primary side reactions to monitor include O-alkylation versus N-alkylation selectivity and potential elimination of the alkyl halide. However, the specific electronic environment of the amide nitrogen, coupled with the optimized base strength and temperature control described in the examples, favors N-alkylation overwhelmingly. The patent examples demonstrate that by carefully controlling the addition rate of the alkylating agent and maintaining appropriate stoichiometry, the formation of bis-alkylated byproducts or hydrolysis of the ester protecting group (if present) can be effectively suppressed. This results in a crude product profile that is amenable to simple crystallization, ensuring that the final commercial scale-up of complex pharmaceutical intermediates meets stringent purity specifications without the need for preparative HPLC.
How to Synthesize (2S)-2-Ethoxy-3-(4-{2-[amino]-2-oxoethoxy}phenyl)propanoic Acid Derivatives Efficiently
The practical execution of this synthesis involves a sequential two-step protocol that maximizes yield and purity. Initially, if the starting material is protected as an ester, a hydrolysis step is performed using lithium hydroxide in a THF/water mixture to reveal the free carboxylic acid, which facilitates subsequent solubility and reactivity adjustments. Following isolation, the key alkylation step is conducted by suspending the acid in anhydrous DMSO and treating it with powdered KOH to generate the reactive species. This methodology is designed for reproducibility and safety, avoiding the exothermic hazards often associated with carbodiimide activations. The detailed standardized synthetic steps provided in the guide below ensure that technical teams can replicate the high yields reported in the patent literature.
- Prepare the free acid precursor by hydrolyzing the ethyl ester using lithium hydroxide in a THF/water mixture, followed by acidification and extraction.
- Suspend the resulting acid in anhydrous DMSO and treat with powdered potassium hydroxide to generate the nucleophilic amide anion.
- Add 1-bromohexane slowly to the reaction mixture at controlled temperatures, stir to completion, and isolate the product via ether extraction and acidification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this alkylation technology translates into tangible strategic advantages beyond mere chemical curiosity. The shift away from proprietary or expensive coupling reagents towards commodity chemicals like alkyl bromides and potassium hydroxide fundamentally alters the cost structure of the intermediate. This structural change in the bill of materials insulates the supply chain from the volatility often seen in the specialty reagent market. Furthermore, the robustness of the reaction conditions allows for processing in standard stainless steel reactors without the need for specialized glass-lined equipment often required for highly corrosive coupling byproducts, thereby increasing asset utilization rates.
- Cost Reduction in Manufacturing: The elimination of stoichiometric coupling agents such as EDC or HATU removes a major cost driver from the production budget. These reagents are not only expensive per kilogram but also require equimolar amounts, doubling the mass intensity of the input materials. By replacing them with inexpensive inorganic bases and alkyl halides, the process achieves substantial cost savings. Additionally, the simplified purification workflow reduces solvent consumption and energy usage associated with extensive chromatography or multiple recrystallizations, further driving down the cost of goods sold (COGS) for the final active pharmaceutical ingredient.
- Enhanced Supply Chain Reliability: The starting materials for this route, including phenethylamine derivatives and simple alkyl halides, are produced on a massive global scale for various industrial applications. This abundance ensures a stable and continuous supply, reducing the risk of production stoppages due to raw material shortages. Unlike specialized chiral coupling catalysts that may have limited suppliers, the reagents here are commoditized. This diversity of supply sources empowers procurement teams to negotiate better terms and secure long-term contracts, ensuring reducing lead time for high-purity pharmaceutical intermediates and maintaining consistent production schedules for downstream API synthesis.
- Scalability and Environmental Compliance: The process demonstrates excellent scalability, as evidenced by the patent examples which operate effectively on hundred-gram scales with potential for tonnage production. The use of DMSO, while requiring careful recovery, is a well-understood solvent in the industry with established recycling protocols. Moreover, the absence of heavy metal catalysts or persistent organic pollutants typical of older coupling methods simplifies waste treatment. The aqueous workup and extraction procedures generate waste streams that are easier to treat and dispose of in compliance with increasingly stringent environmental regulations, making this a sustainable choice for modern green chemistry initiatives in fine chemical manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the experimental data and claims within CN1809528A, providing clarity for technical teams evaluating this technology for their pipeline. Understanding these specifics is crucial for risk assessment and process validation during the technology transfer phase.
Q: How does the novel alkylation method improve upon traditional amide coupling for this intermediate?
A: The novel method replaces expensive carbodiimide coupling agents with cost-effective alkyl halides and inorganic bases, significantly reducing raw material costs and simplifying purification by avoiding urea byproducts.
Q: What represents the critical quality attribute for the stereocenter in this synthesis?
A: Maintaining the (2S)-configuration is paramount for biological activity as a PPARα modulator; the patented process ensures high enantiomeric excess (e.e. >97%) through mild basic conditions that prevent racemization.
Q: Is this process suitable for multi-ton commercial production?
A: Yes, the use of robust solvents like DMSO and common inorganic bases like KOH, combined with high corrected yields (over 90% in examples), indicates excellent scalability for industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (2S)-3-(4-{2-[amino]-2-oxoethoxy}phenyl)-2-ethoxypropionic acid derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development of metabolic disease therapeutics. Our technical team has thoroughly analyzed the alkylation methodology described in CN1809528A and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this chemistry to life. We are equipped with rigorous QC labs and state-of-the-art analytical instrumentation to ensure that every batch meets stringent purity specifications, including precise control over enantiomeric excess and residual solvent levels. Our commitment to quality assurance means that we can deliver the (2S)-enantiomer with the consistency demanded by global regulatory bodies.
We invite pharmaceutical partners to collaborate with us to leverage this cost-effective and scalable synthetic route. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to obtain specific COA data and route feasibility assessments that demonstrate how our manufacturing capabilities can optimize your supply chain for PPARα modulator programs. Let us be your partner in transforming innovative patent chemistry into commercial reality.
