Advanced Manufacturing of 2-Substituted Pyrimidine Carboxylic Acids for Global Supply Chains
The chemical landscape for heterocyclic intermediates is constantly evolving, driven by the need for more efficient and scalable manufacturing routes. Patent CN101171237B introduces a transformative methodology for the preparation of optionally 2-substituted 1,6-dihydro-6-oxo-4-pyrimidinecarboxylic acids, specifically compounds of formula 1. This innovation addresses long-standing challenges in the synthesis of these critical scaffolds, which serve as pivotal building blocks for a wide array of bioactive molecules, including herbicides and potential pharmaceutical agents. By shifting away from traditional, uncontrolled condensation reactions, this patented process leverages precise pH modulation to achieve superior reaction kinetics and product purity. For global supply chain leaders, understanding this technology is essential for securing a reliable agrochemical intermediate supplier capable of meeting stringent quality demands while optimizing production costs.
Historically, the synthesis of 1,6-dihydro-6-oxo-4-pyrimidinecarboxylic acids has been plagued by inefficiencies inherent in early methodologies. Seminal work by G.D. Daves, Jr. et al. described a process where diethyl oxalacetate and formamidine were mixed simultaneously in aqueous sodium hydroxide without specific pH control. This粗放 approach typically resulted in modest yields of approximately 63%, with significant material loss attributed to uncontrolled side reactions and hydrolysis. Other reports utilizing similar oxalic acid diester condensations have disclosed even lower efficiencies, creating a bottleneck for commercial scale-up. The lack of selectivity in these conventional methods often necessitates extensive purification steps, driving up both the cost of goods sold and the environmental footprint of the manufacturing process. Consequently, there has been a persistent industry demand for new methods that can provide higher yields alongside low cost, high efficiency, and reliability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
The primary deficiency of legacy synthesis routes lies in their inability to discriminate between competing reaction pathways during the critical cyclization phase. When reagents are combined without regard for the protonation state of the intermediates, the reactive diester species can undergo non-selective hydrolysis or polymerization before the desired ring closure occurs. This results in a complex crude reaction mixture containing numerous impurities that are structurally similar to the target molecule, making downstream purification arduous and yield-destructive. Furthermore, the exothermic nature of uncontrolled base-catalyzed condensations can lead to thermal runaways, posing safety risks in large-scale reactors. These factors collectively undermine the economic viability of producing high-purity pyrimidine carboxylic acids, forcing procurement teams to accept higher prices or compromise on specification limits.
The Novel Approach
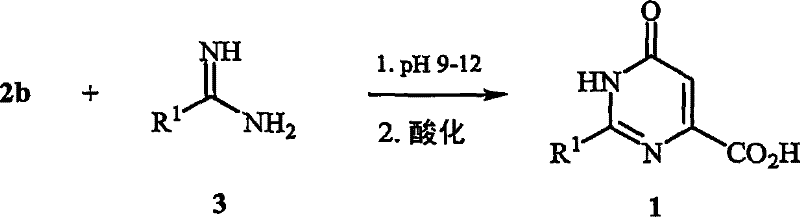
In stark contrast, the process disclosed in CN101171237B decouples the hydrolysis and cyclization events through a sophisticated, multi-step protocol. The innovation begins with the selective saponification of a diester salt (formula 2a) under strictly controlled alkaline conditions, specifically maintaining a pH range of about 10 to 14. This ensures the quantitative formation of the monoester salt (formula 2b) without degrading the sensitive oxalacetate backbone. Subsequently, this activated intermediate is reacted with a formamidine derivative (formula 3) in a second distinct phase where the pH is carefully adjusted to a range of about 9 to 12. This precise orchestration of reaction conditions maximizes the nucleophilic attack required for ring closure while suppressing degradation pathways, thereby delivering substantially improved yields and a cleaner impurity profile compared to the prior art.
Mechanistic Insights into pH-Controlled Cyclization
The core mechanistic advantage of this invention resides in the stabilization of the enolate intermediate during the initial hydrolysis step. By maintaining the reaction medium at a pH of 10 to 14, typically using alkali metal hydroxides like sodium hydroxide, the process ensures that the ester group at the OM twin position on formula 2a is selectively saponified to form the corresponding CO2M group of formula 2b. This mono-anionic species is crucial because it retains the electrophilicity necessary for the subsequent condensation while being stable enough to prevent premature decomposition. The use of aqueous alkanol solvents, such as ethanol-water mixtures, further facilitates this transformation by solubilizing both the organic esters and the inorganic bases, creating a homogeneous reaction environment that promotes consistent kinetics across large batch sizes.
Following the generation of the monoester, the cyclization mechanism proceeds through a concerted condensation with the formamidine reagent. As illustrated in the reaction scheme below, the interaction between the activated methylene group of the oxalacetate derivative and the amidine functionality is highly dependent on the protonation state of the nitrogen atoms. Maintaining the pH between 9 and 12 ensures that the formamidine is sufficiently nucleophilic to attack the carbonyl center, yet not so basic as to cause rapid hydrolysis of the newly formed pyrimidine ring. This delicate balance allows for the efficient construction of the heterocyclic core, ultimately yielding the target 1,6-dihydro-6-oxo-4-pyrimidinecarboxylic acid (formula 1) after a final acidification step.
Furthermore, the process accounts for the tautomeric equilibrium inherent to the pyrimidine system. The compounds of formula 1 exist in equilibrium with their respective tautomeric counterparts, formula 1a, as shown in the patent exhibits. The robust nature of the synthetic method ensures that the thermodynamic product is favored, regardless of the specific tautomeric form isolated. This mechanistic understanding is vital for R&D directors focusing on impurity control, as it confirms that the process is designed to drive the reaction to completion, minimizing the presence of open-chain precursors or hydrolysis byproducts that could complicate regulatory filings for downstream active ingredients.
How to Synthesize 2-Substituted 1,6-Dihydro-6-Oxo-4-Pyrimidinecarboxylic Acids Efficiently
Implementing this novel synthetic route requires careful attention to the sequential addition of reagents and real-time monitoring of reaction parameters. The process begins by preparing a mixture of the diester salt, a lower alkanol, and water, followed by the controlled addition of a base to achieve the initial hydrolysis. Once the monoester intermediate is generated, the formamidine is introduced, and the pH is fine-tuned to facilitate cyclization. The detailed standardized synthesis steps see the guide below.
- Perform selective saponification of formula 2a diester in aqueous alkanol at pH 10-14 to generate formula 2b monoester salt.
- React the formula 2b solution with formula 3 formamidine at pH 9-12 and elevated temperature to form the pyrimidine ring.
- Acidify the reaction mixture to pH below 3 to precipitate the final formula 1 carboxylic acid product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology offers tangible strategic benefits beyond mere chemical elegance. The shift towards a pH-controlled, stepwise process eliminates the reliance on expensive transition metal catalysts or exotic reagents that often characterize modern cross-coupling approaches. Instead, it utilizes commodity chemicals like diethyl oxalacetate salts and simple formamidines, which are readily available from multiple global sources. This raw material accessibility drastically reduces supply chain risk, ensuring that production schedules are not disrupted by the scarcity of specialized inputs. Moreover, the simplified workup procedure, which often involves direct crystallization upon acidification, minimizes the need for resource-intensive chromatography or multiple extraction cycles.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the substantial improvement in yield and the reduction of waste generation. By avoiding the low-efficiency pitfalls of conventional simultaneous mixing methods, manufacturers can extract significantly more product from the same amount of starting material, directly lowering the unit cost of production. Additionally, the elimination of complex purification steps reduces solvent consumption and energy usage associated with distillation and drying. The use of aqueous ethanol systems further lowers costs compared to anhydrous organic solvents, providing a clear pathway for cost reduction in agrochemical intermediate manufacturing without compromising on quality standards.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route translates directly into improved delivery performance for customers. Because the reaction conditions are mild and tolerant of minor variations in reagent quality, the process is less prone to batch failures or off-spec results that can delay shipments. The ability to operate effectively in water-containing solvent systems also simplifies logistics, as strict moisture control is not required for the bulk solvents. This operational resilience ensures a consistent flow of high-purity pyrimidine carboxylic acids, allowing downstream formulators to maintain their own production schedules with confidence and reducing lead time for high-purity intermediates.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this process aligns perfectly with modern green chemistry principles. The avoidance of heavy metal catalysts means there is no need for costly and technically challenging metal scavenging steps to meet residual metal specifications. The aqueous nature of the reaction media facilitates easier wastewater treatment and reduces the volume of hazardous organic waste requiring incineration. These factors make the commercial scale-up of complex pyrimidine derivatives significantly easier to permit and operate within strict regulatory frameworks, ensuring long-term sustainability for the manufacturing site.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and embodiments provided in the patent documentation, offering clarity on process capabilities and limitations. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: How does the new pH-controlled method improve yield compared to conventional processes?
A: Conventional methods often mix components simultaneously without pH control, resulting in yields around 63%. The novel process utilizes a stepwise approach with precise pH regulation (10-14 for hydrolysis, 9-12 for cyclization), significantly minimizing side reactions and improving overall isolation yields.
Q: What are the key advantages for large-scale manufacturing of these intermediates?
A: The process utilizes inexpensive, readily available starting materials like diethyl oxalacetate salts and avoids complex transition metal catalysts. The use of aqueous ethanol systems simplifies solvent recovery and waste treatment, enhancing scalability and environmental compliance.
Q: Can this synthetic route be adapted for different R1 substituents?
A: Yes, the methodology is highly versatile regarding the R1 group. The patent demonstrates successful preparation with various carbon moieties including cyclopropyl and substituted phenyl groups, making it suitable for generating diverse libraries of agrochemical and pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,6-Dihydro-6-Oxo-4-Pyrimidinecarboxylic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of next-generation agrochemicals and pharmaceuticals. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that utilize advanced analytical techniques to verify every batch against the highest industry standards. Our facility is equipped to handle the specific pH-controlled requirements of this patented process, guaranteeing consistency and reliability for your supply chain.
We invite you to collaborate with us to leverage this advanced manufacturing technology for your specific project needs. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this efficient route can optimize your overall budget. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a competitive advantage in the global market.
