Advanced Manufacturing of Idelalisib: A Green Four-Step Synthetic Route for Global Supply Chains
Advanced Manufacturing of Idelalisib: A Green Four-Step Synthetic Route for Global Supply Chains
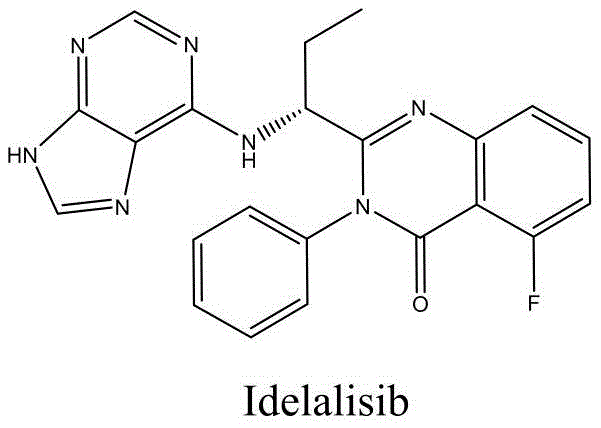
The pharmaceutical landscape for oncology treatments continues to evolve with the demand for highly selective kinase inhibitors, exemplified by Idelalisib, a potent PI3K-delta inhibitor approved for treating relapsed chronic lymphocytic leukemia and follicular lymphoma. As global demand for this critical active pharmaceutical ingredient (API) surges, the industry faces significant pressure to optimize manufacturing processes that are not only cost-effective but also environmentally sustainable and capable of delivering ultra-high purity. Patent CN108409740B introduces a groundbreaking preparation method that fundamentally restructures the synthetic pathway, moving away from traditional, hazardous multi-step sequences toward a streamlined, four-step protocol. This innovation addresses long-standing bottlenecks in the supply chain, offering a robust alternative for reliable pharmaceutical intermediate suppliers aiming to secure long-term contracts with major generic and branded drug manufacturers. By leveraging a unique Hexamethyldisilazane (HMDS) and Lewis acid catalytic system, this technology promises to redefine the economic and technical feasibility of producing complex heterocyclic drugs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
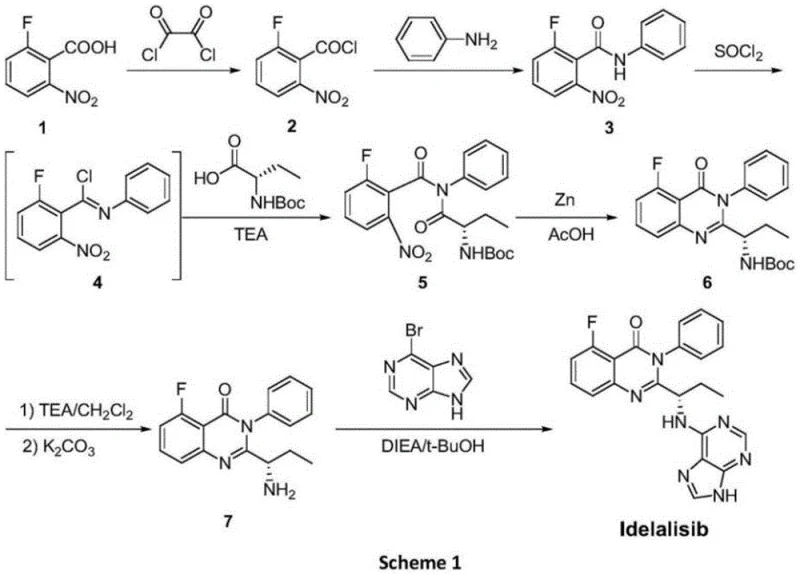
Historically, the industrial synthesis of Idelalisib has been plagued by severe technical inefficiencies and safety hazards inherent in legacy pathways, such as those described in WO2005113554. A primary bottleneck in these conventional routes is the reliance on unstable acyl chloride intermediates, which necessitate rigorous anhydrous and anaerobic conditions to prevent hydrolysis, thereby complicating reactor operations and increasing energy consumption. Furthermore, the traditional cyclization steps frequently employ Zinc powder in acetic acid, a reagent combination that generates substantial quantities of zinc acetate waste and poses a critical risk of heavy metal residue in the final drug substance. These metal contaminants are notoriously difficult to remove to ppm levels required by regulatory bodies, often necessitating expensive additional purification stages that erode profit margins. Additionally, the use of expensive halogenated starting materials like 6-bromopurine not only drives up raw material costs but also results in the generation of bromine-containing wastewater, creating significant environmental compliance burdens for manufacturing facilities.
The Novel Approach
In stark contrast to these cumbersome legacy methods, the novel synthesis route disclosed in CN108409740B offers a paradigm shift by utilizing stable, commercially available starting materials and eliminating hazardous reagents entirely. This innovative pathway condenses the synthesis into just four distinct chemical transformations, drastically reducing the cumulative yield loss typically associated with longer linear sequences. The core of this improvement lies in the strategic selection of intermediates that avoid the formation of moisture-sensitive acyl chlorides, instead favoring robust ester and amide linkages that tolerate standard industrial handling conditions. By replacing the toxic Zinc/acetic acid reduction system with a mild HMDS/Lewis acid catalytic cycle, the process inherently prevents metal contamination, ensuring a cleaner crude product profile that simplifies downstream purification. This approach not only enhances the overall atom economy but also aligns perfectly with modern green chemistry principles, making it an ideal solution for cost reduction in pharmaceutical intermediates manufacturing while simultaneously mitigating environmental risks.
Mechanistic Insights into HMDS/Lewis Acid Catalyzed Cyclization
The cornerstone of this technological advancement is the sophisticated ring-closure mechanism facilitated by the Hexamethyldisilazane (HMDS) and Lewis acid catalytic system, which replaces the harsh reductive conditions of the past. In this critical step, the intermediate F undergoes a cyclization reaction where HMDS acts as a silylating agent, activating the amide nitrogen for nucleophilic attack on the adjacent electrophilic center. The presence of a weak Lewis acid, such as iodine or zinc chloride, further polarizes the reacting bonds, lowering the activation energy required for the formation of the quinazolinone core. This mechanistic pathway is exceptionally mild, typically proceeding at temperatures between 10°C and 80°C, which minimizes thermal degradation of the sensitive purine moiety and prevents the formation of polymeric by-products. The result is a highly selective transformation that preserves the stereochemical integrity of the chiral center at the propyl chain, a factor crucial for maintaining the biological potency of the final API.
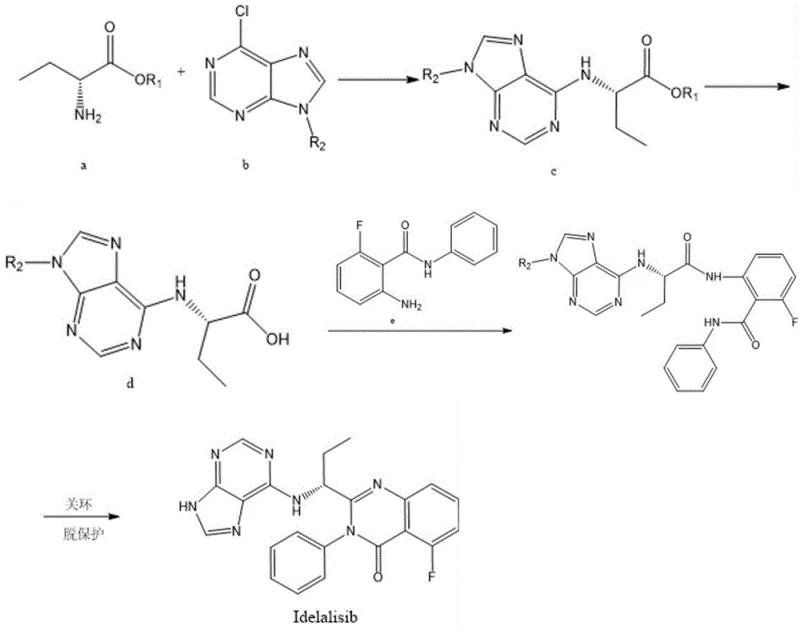
Beyond the primary cyclization event, this catalytic system plays a pivotal role in impurity control, a key concern for high-purity pharmaceutical intermediates. Traditional acid-catalyzed cyclizations often lead to racemization or over-reaction, generating structurally related impurities that are difficult to separate chromatographically. However, the specific coordination chemistry of the HMDS/Lewis acid complex ensures a controlled reaction environment that favors the desired intramolecular condensation over competing intermolecular side reactions. Following the cyclization, the subsequent deprotection step is seamlessly integrated, allowing for the removal of protecting groups under mild acidic conditions without compromising the newly formed heterocyclic ring. This dual functionality of the catalytic system—promoting ring closure while facilitating clean deprotection—significantly reduces the complexity of the work-up procedure, leading to a final product with purity levels exceeding 98% as confirmed by HPLC analysis in the patent examples.
How to Synthesize Idelalisib Efficiently
The implementation of this four-step synthesis requires precise control over reaction parameters to maximize yield and minimize impurity formation, particularly during the nucleophilic substitution and condensation phases. The process begins with the coupling of an amino acid ester with a protected purine derivative, followed by hydrolysis to the free acid, which then serves as the substrate for amide bond formation with a fluorinated aniline derivative. The final and most critical stage involves the HMDS-mediated cyclization, which must be carefully monitored to ensure complete conversion before proceeding to deprotection. For process chemists and plant managers looking to adopt this technology, understanding the specific solvent systems and stoichiometric ratios detailed in the patent is essential for successful technology transfer. The detailed standardized synthesis steps for implementing this route are outlined below.
- Perform nucleophilic substitution between Compound A and Compound B in the presence of an acid-binding agent to obtain Intermediate C.
- Hydrolyze Compound C under acidic or basic conditions to generate the carboxylic acid Intermediate D.
- Condense Intermediate D with Compound E using a coupling agent to form the amide linkage in Intermediate F.
- Execute the final ring-closure reaction on Intermediate F using a Hexamethyldisilazane (HMDS) and Lewis acid catalytic system, followed by deprotection.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route translates directly into tangible operational efficiencies and risk mitigation strategies that strengthen the overall supply network. By eliminating the need for expensive and hazardous reagents like 6-bromopurine and Zinc powder, manufacturers can significantly reduce their raw material expenditure and waste disposal costs, creating a more resilient cost structure. The reduction in synthesis steps from seven to four inherently shortens the production cycle time, allowing for faster turnaround on orders and improved responsiveness to market fluctuations in demand for oncology drugs. Furthermore, the avoidance of heavy metal catalysts removes a major regulatory hurdle, reducing the likelihood of batch failures due to out-of-specification metal residues, which is a common cause of supply disruption in the pharmaceutical industry.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the drastic simplification of the synthetic sequence and the elimination of costly purification steps associated with metal removal. By avoiding the use of Zinc powder and acetic acid, the process removes the need for extensive washing and chelation treatments required to meet strict residual solvent and heavy metal guidelines, thereby lowering utility and consumable costs. Additionally, the higher overall yield achieved through fewer reaction steps means that less starting material is required to produce the same amount of final API, directly improving the cost of goods sold (COGS). The use of common, non-proprietary solvents like tert-butanol and ethyl acetate further ensures that supply chain volatility for specialized reagents does not impact production budgets.
- Enhanced Supply Chain Reliability: From a logistics perspective, the stability of the intermediates generated in this route offers a significant advantage over traditional methods that rely on moisture-sensitive acyl chlorides. Because the intermediates do not require strict anhydrous storage or transport conditions, the risk of material degradation during warehousing and shipping is minimized, ensuring consistent quality upon arrival at the manufacturing site. The robustness of the reaction conditions also allows for greater flexibility in scheduling, as the process is less susceptible to minor variations in temperature or humidity that might halt a more sensitive legacy process. This reliability is crucial for maintaining continuous production schedules and meeting the just-in-time delivery requirements of major pharmaceutical clients.
- Scalability and Environmental Compliance: The environmental profile of this synthesis is markedly superior, characterized by the absence of bromine-containing wastewater and heavy metal sludge, which simplifies effluent treatment and reduces the facility's environmental footprint. The mild reaction conditions and the use of recyclable solvents make the process highly amenable to scale-up from pilot plant to commercial tonnage without the need for exotic equipment or specialized containment systems. This scalability ensures that suppliers can rapidly ramp up production capacity to meet surging global demand without facing the engineering bottlenecks often associated with hazardous chemical processes. Consequently, this route supports sustainable manufacturing goals while ensuring long-term regulatory compliance in increasingly strict environmental jurisdictions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced Idelalisib synthesis route, providing clarity for stakeholders evaluating its potential for integration into their supply chains. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, highlighting the practical benefits of the HMDS catalytic system. Understanding these nuances is vital for making informed decisions about process adoption and vendor qualification.
Q: How does this new synthesis route improve upon previous methods regarding metal contamination?
A: Unlike prior art methods that utilize Zinc powder and acetic acid for reduction and cyclization, which often leave difficult-to-remove metal residues, this novel process employs an HMDS/Lewis acid catalytic system. This completely eliminates the risk of heavy metal contamination, ensuring the final API meets stringent pharmacopeial standards without complex purification steps.
Q: What are the stability advantages of the intermediates in this process?
A: Conventional routes involve the formation of highly unstable acyl chloride intermediates that require strict anhydrous and anaerobic conditions, leading to low yields and operational complexity. The disclosed method utilizes stable ester and amide intermediates that can be handled under standard industrial conditions, significantly simplifying the workflow and improving overall throughput.
Q: Is this synthetic route suitable for large-scale commercial production?
A: Yes, the process is specifically designed for scalability. By reducing the total step count from seven to four and utilizing mild reaction conditions with easily recoverable solvents, the method offers substantial improvements in cost-efficiency and environmental compliance, making it highly viable for multi-kilogram to ton-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Idelalisib Supplier
As the global pharmaceutical industry increasingly prioritizes green chemistry and supply chain resilience, NINGBO INNO PHARMCHEM stands at the forefront of adopting these cutting-edge synthetic methodologies to deliver superior value to our partners. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We are committed to maintaining stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of Idelalisib intermediate meets the highest international standards for safety and efficacy. By integrating this novel four-step route into our manufacturing portfolio, we are able to offer a product that is not only cost-competitive but also environmentally responsible and free from the risks associated with traditional heavy metal catalysis.
We invite forward-thinking procurement leaders and R&D directors to engage with us to explore how this optimized synthesis can enhance your drug development pipeline and reduce overall manufacturing costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our advanced capabilities can support your long-term strategic goals in the oncology therapeutic sector.
