Scaling High-Efficiency ACDB Photosensitizer Production For Next-Generation Photodynamic Therapy
Scaling High-Efficiency ACDB Photosensitizer Production For Next-Generation Photodynamic Therapy
The rapidly evolving landscape of oncological therapeutics demands precision tools that can effectively target malignant cells while minimizing systemic toxicity. In this context, Patent CN111592560A introduces a groundbreaking advancement in the field of biomedical imaging and treatment through the development of the ACDB photosensitizer probe. This specific molecular architecture represents a significant leap forward in Boron-Dipyrromethene (BODIPY) chemistry, addressing the longstanding challenges of low singlet oxygen quantum yields and poor photostability that have plagued earlier generations of photosensitizing agents. By strategically incorporating heavy atoms into the BODIPY core, this innovation unlocks superior reactive oxygen species (ROS) generation capabilities, making it an ideal candidate for clinical photodynamic therapy (PDT) and real-time fluorescence monitoring. For global pharmaceutical developers seeking a reliable photosensitizer supplier, understanding the technical nuances of this patented route is critical for securing a competitive edge in the burgeoning market of image-guided cancer therapies.
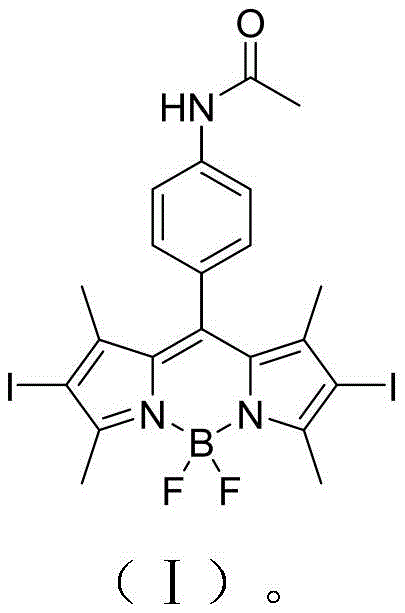
The commercial viability of this technology hinges not just on its biological efficacy but on the robustness of its manufacturing process. The patent delineates a concise, four-step synthetic pathway that transforms readily available commodity chemicals into a high-value specialty intermediate. This approach mitigates many of the supply chain risks associated with complex heterocyclic synthesis, offering a streamlined solution for cost reduction in pharmaceutical intermediate manufacturing. As we delve deeper into the technical specifics, it becomes evident that this methodology offers a balanced proposition of high purity, operational safety, and scalability, positioning ACDB as a cornerstone molecule for next-generation theranostic applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the development of effective photosensitizers for photodynamic therapy has been hindered by intrinsic limitations in molecular design and synthetic accessibility. Traditional BODIPY dyes, while possessing excellent fluorescence quantum yields, often suffer from inefficient intersystem crossing (ISC), which is the critical physical process required to generate the triplet state necessary for singlet oxygen production. Without efficient ISC, the absorbed light energy is merely re-emitted as fluorescence or heat, rendering the molecule ineffective for killing tumor cells. Furthermore, conventional synthesis routes for functionalized BODIPY derivatives frequently involve harsh reaction conditions, expensive transition metal catalysts that are difficult to remove, and multi-step protection-deprotection sequences that drastically lower overall yields. These factors contribute to high production costs and inconsistent batch-to-batch quality, creating significant bottlenecks for procurement managers aiming to secure a stable supply of high-purity OLED material or biomedical intermediates.
The Novel Approach
The methodology outlined in Patent CN111592560A circumvents these historical obstacles through a clever integration of the heavy atom effect directly into the synthetic workflow. By introducing iodine atoms at the 2 and 6 positions of the pyrrole rings early in the synthesis, the novel approach fundamentally alters the electronic properties of the BODIPY core, promoting rapid spin-orbit coupling and enhancing triplet state formation. This strategic modification ensures that the resulting ACDB probe exhibits a singlet oxygen quantum yield of approximately 0.56, a substantial improvement over non-iodinated analogues. Moreover, the synthetic route employs mild conditions, utilizing common reagents like N-iodosuccinimide for halogenation and hydrazine hydrate for reduction, thereby eliminating the need for exotic catalysts. This simplification not only accelerates the reaction kinetics but also streamlines the downstream purification processes, ensuring that the final product meets the stringent purity specifications required for in vivo applications without exorbitant processing costs.
Mechanistic Insights into Iodinated BODIPY Functionalization
The chemical elegance of the ACDB synthesis lies in its sequential logic, which builds complexity while maintaining control over regioselectivity and functional group tolerance. The process initiates with the acid-catalyzed condensation of p-nitrobenzaldehyde and 2,4-dimethylpyrrole, forming the dipyrromethene precursor which is subsequently complexed with boron trifluoride to establish the rigid BODIPY scaffold. This core structure serves as the foundation for all subsequent modifications. The critical mechanistic step occurs during the iodination phase, where N-iodosuccinimide (NIS) acts as a mild electrophilic iodinating agent. Due to the electron-rich nature of the BODIPY pyrrole rings, the iodination proceeds selectively at the alpha-positions (2,6-positions), avoiding unwanted substitution on the phenyl ring or the methyl groups. This regioselectivity is paramount, as placing the heavy atoms at these specific locations maximizes the perturbation of the excited state dynamics, directly correlating to the enhanced therapeutic efficacy observed in biological assays.
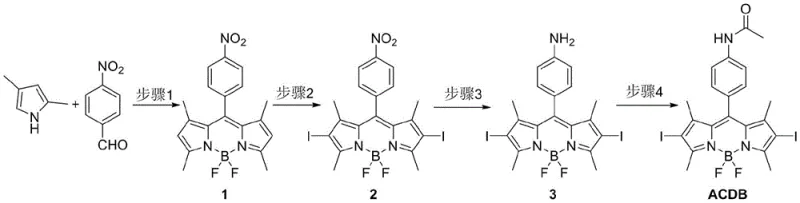
Following the installation of the heavy atoms, the synthesis addresses biocompatibility through a reduction-acetylation sequence. The nitro group on the phenyl ring, initially present from the starting aldehyde, serves as a masked handle for further functionalization. The patent describes a catalytic hydrogenation alternative using hydrazine hydrate and palladium on carbon, a method chosen for its operational simplicity and safety profile compared to high-pressure hydrogen gas. This reduction converts the electron-withdrawing nitro group into an electron-donating amino group, which can influence the intramolecular charge transfer (ICT) properties of the dye. Finally, the acetylation of this amine with acetyl chloride stabilizes the molecule against metabolic oxidation and fine-tunes its lipophilicity. This final structural adjustment is crucial for ensuring that the probe can effectively penetrate cell membranes and accumulate within tumor tissues, thereby fulfilling its dual role as both a therapeutic agent and a diagnostic marker in complex biological environments.
How to Synthesize ACDB Photosensitizer Probe Efficiently
Executing the synthesis of ACDB requires precise control over reaction parameters to maximize yield and minimize impurity profiles, particularly given the sensitivity of the BODIPY core to acidic or oxidative degradation. The patented procedure offers a standardized protocol that balances reaction time, temperature, and stoichiometry to achieve optimal results. The initial condensation step is typically conducted under an inert nitrogen atmosphere to prevent oxidative polymerization of the pyrrole species, followed by careful pH control during the workup to isolate the intermediate boron complex. Subsequent steps, including the iodination and reduction, rely on monitoring reaction progress via thin-layer chromatography or HPLC to prevent over-reaction or side-product formation. For technical teams looking to implement this process, adhering to the specified solvent systems—such as ultra-dry dichloromethane for the halogenation and ethanol for the reduction—is essential for maintaining reproducibility. Detailed standardized synthesis steps are provided in the guide below to assist R&D departments in replicating this high-performance pathway.
- Condense p-nitrobenzaldehyde with 2,4-dimethylpyrrole using TFA and DDQ, followed by BF3 complexation to form the BODIPY core.
- Perform regioselective iodination at the 2,6-positions using N-iodosuccinimide (NIS) to enhance intersystem crossing.
- Reduce the nitro group to an amine using hydrazine hydrate and Pd/C catalyst under reflux conditions.
- Finalize the probe by acetylating the amine group with acetyl chloride in the presence of a base.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, the ACDB synthesis route presents compelling advantages that align with the goals of cost efficiency and supply chain resilience. The reliance on bulk commodity chemicals such as p-nitrobenzaldehyde, 2,4-dimethylpyrrole, and acetyl chloride means that raw material procurement is not subject to the volatility often seen with specialized fine chemicals. This abundance of starting materials ensures a consistent supply flow, reducing the risk of production stoppages due to vendor shortages. Furthermore, the elimination of precious metal catalysts in favor of more accessible reagents like palladium on carbon (which can be recovered and recycled) contributes to substantial cost savings in pharmaceutical intermediate manufacturing. The simplified purification protocols, primarily involving standard column chromatography and solvent extraction, reduce the consumption of expensive stationary phases and decrease the overall cycle time per batch, allowing for higher throughput in existing manufacturing facilities.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by the high atom economy of the condensation reaction and the use of inexpensive halogenating agents. By avoiding complex protecting group strategies and utilizing a linear synthetic sequence, the total number of unit operations is minimized. This reduction in processing steps directly translates to lower labor costs, reduced energy consumption for heating and cooling, and decreased solvent waste disposal fees. Additionally, the high selectivity of the iodination step minimizes the formation of difficult-to-separate isomers, which significantly lowers the cost associated with final purification and quality control testing.
- Enhanced Supply Chain Reliability: The robustness of the synthetic route enhances supply chain security by reducing dependency on single-source suppliers for exotic reagents. The solvents employed, including tetrahydrofuran, dichloromethane, and ethanol, are standard industrial grades available from multiple global vendors, mitigating geopolitical or logistical risks. Moreover, the reaction conditions are relatively mild, with most steps proceeding at room temperature or moderate reflux, which lowers the safety risks associated with high-pressure or cryogenic operations. This operational safety profile facilitates easier regulatory approval for manufacturing sites and ensures continuous production capability even under stringent environmental health and safety regulations.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is facilitated by the homogeneous nature of the reactions and the absence of gaseous reagents that require specialized handling equipment. The use of hydrazine hydrate for reduction, while requiring careful handling, avoids the need for high-pressure hydrogenation reactors, simplifying the engineering requirements for scale-up. From an environmental standpoint, the process generates manageable waste streams that can be treated using standard effluent protocols. The high yield of the final acetylation step ensures that raw material utilization is maximized, aligning with green chemistry principles and supporting corporate sustainability goals by minimizing the carbon footprint per kilogram of produced active ingredient.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of the ACDB photosensitizer probe. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a clear understanding of the technology's capabilities and limitations. Understanding these details is vital for stakeholders evaluating the feasibility of integrating this intermediate into their broader drug development pipelines.
Q: What is the primary advantage of the ACDB probe structure?
A: The ACDB structure incorporates iodine atoms at the 2,6-positions of the BODIPY core, which induces a heavy atom effect. This significantly enhances the intersystem crossing rate, leading to a high singlet oxygen quantum yield essential for effective photodynamic therapy.
Q: Is the synthesis of ACDB scalable for industrial production?
A: Yes, the patented process utilizes standard organic solvents like dichloromethane, ethanol, and THF, and avoids extreme cryogenic conditions. The use of commodity starting materials such as p-nitrobenzaldehyde ensures that the route is highly amenable to commercial scale-up.
Q: How does the acetylation step impact the probe's performance?
A: The final acetylation step converts the reactive amine into an acetamide group. This modification improves the metabolic stability and lipophilicity of the probe, facilitating better cellular uptake and retention during biological imaging and therapeutic applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable ACDB Photosensitizer Probe Supplier
As the demand for advanced theranostic agents continues to surge, partnering with an experienced chemical manufacturer is essential for translating laboratory innovations into commercial realities. NINGBO INNO PHARMCHEM stands at the forefront of this industry, leveraging extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the specific requirements of BODIPY dye synthesis, ensuring that every batch of ACDB meets stringent purity specifications through our rigorous QC labs. We understand that the consistency of your raw materials directly impacts the efficacy of your final therapeutic products, which is why we prioritize process optimization and quality assurance at every stage of the manufacturing lifecycle.
We invite global partners to collaborate with us to unlock the full potential of this patented technology. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical needs. By engaging with our technical procurement team, you can access specific COA data and route feasibility assessments that will empower you to make informed decisions for your supply chain. Let us support your journey in developing next-generation photodynamic therapies with a reliable partner committed to excellence, innovation, and long-term supply stability.
