Advanced Synthesis of 5-Fluoro-3-methylisobenzofuranone for Oncology Drug Manufacturing
Advanced Synthesis of 5-Fluoro-3-methylisobenzofuranone for Oncology Drug Manufacturing
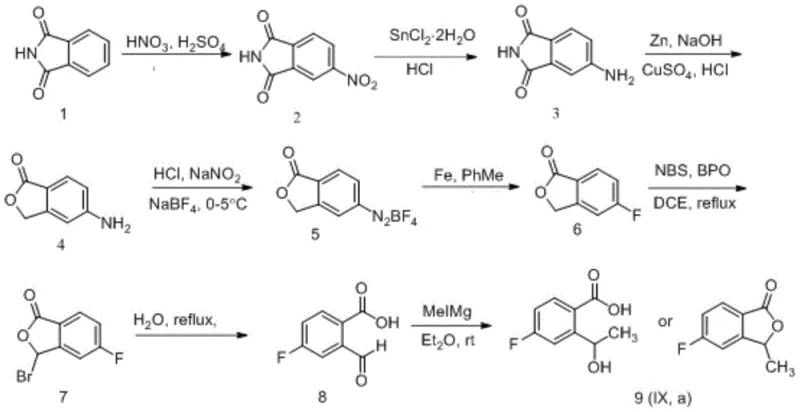
The pharmaceutical industry is constantly seeking robust and cost-effective pathways for the production of critical oncology intermediates, particularly for next-generation ALK inhibitors like Laratinib (PF-06463922). A significant technological breakthrough in this domain is detailed in patent CN109134410B, which discloses a novel eight-step synthesis method for 5-fluoro-3-methylisobenzofuran-1(3H)-one. This compound serves as a pivotal building block in the assembly of Laratinib, a drug designed to treat non-small cell lung cancer in patients who have developed resistance to first and second-generation ALK inhibitors. The disclosed methodology represents a strategic shift from previous complex routes, utilizing phthalimide as a foundational raw material to achieve the target molecule through a sequence involving nitration, reduction, cyclization, diazotization, and final lactonization. This approach not only streamlines the synthetic pathway but also addresses critical pain points regarding raw material availability and operational simplicity, making it a highly attractive candidate for commercial adoption by forward-thinking pharmaceutical manufacturers.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methods for synthesizing key intermediates for Laratinib, such as those described in patent WO 2013132376, have historically suffered from significant inefficiencies that hinder large-scale production. These conventional routes are often characterized by excessive step counts, which inherently accumulate yield losses and increase the burden on purification processes at each stage. Furthermore, the reliance on specialized or expensive starting materials in older methodologies drives up the cost of goods sold (COGS), creating a barrier to entry for generic manufacturers or companies aiming to optimize their supply chain economics. The complexity of post-treatment in these legacy processes often involves difficult separations or the use of hazardous reagents that require extensive safety protocols, thereby extending the overall production cycle time. For procurement managers and supply chain heads, these factors translate into higher volatility in supply continuity and reduced flexibility in responding to market demand fluctuations for the final API.
The Novel Approach
In stark contrast, the novel approach outlined in CN109134410B leverages phthalimide, a commodity chemical, to construct the core isobenzofuranone scaffold with remarkable efficiency. By initiating the synthesis with a straightforward nitration followed by a tandem reduction and cyclization sequence, the process rapidly establishes the necessary heterocyclic framework. The integration of a Balz-Schiemann type fluorination strategy allows for the precise introduction of the fluorine atom, a critical pharmacophore, using relatively inexpensive reagents like sodium fluoroborate and iron powder. This route effectively bypasses the need for exotic fluorinating agents or complex protecting group strategies often seen in alternative syntheses. The culmination of the synthesis involves a Grignard reaction that simultaneously installs the methyl group and facilitates ring closure, demonstrating a high degree of atom economy. This streamlined logic results in a total yield of over 6.1 percent across eight steps, which, while modest in isolation, is achieved through operations that are fundamentally simpler and more amenable to scale-up than previous iterations.
Mechanistic Insights into the Synthetic Sequence and Chiral Control
The chemical elegance of this synthesis lies in its strategic manipulation of functional groups to drive reactivity towards the desired lactone structure. The initial nitration of phthalimide under mixed acid conditions is carefully controlled at temperatures between 10-30°C to ensure regioselectivity, favoring the 4-nitro isomer which is essential for the subsequent ortho-cyclization. The reduction of the nitro group using stannous chloride followed by zinc-mediated ring closure is a classic yet powerful transformation that converts the planar phthalimide into the three-dimensional isobenzofuranone core. Crucially, the diazotization step is performed at low temperatures (below 0°C) to stabilize the diazonium salt before its conversion to the fluoro-borate complex. The thermal decomposition of this complex in the presence of iron powder is a heterogeneous reaction that requires efficient heat transfer management, typically achieved in solvents like toluene at reflux, to ensure complete conversion to the fluoro-intermediate without generating excessive tar or polymeric byproducts.
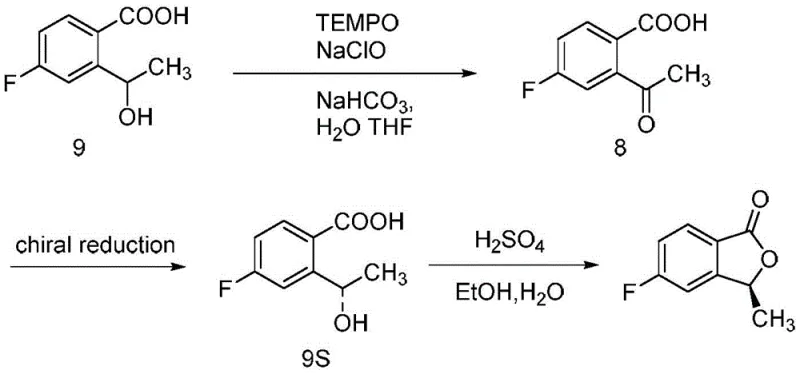
Impurity control is paramount in the later stages, particularly during the radical bromination and subsequent hydrolysis. The use of N-bromosuccinimide (NBS) with benzoyl peroxide (BPO) initiation allows for selective bromination at the benzylic position, but care must be taken to prevent over-bromination or ring degradation. The subsequent hydrolysis of the bromo-lactone to the formyl-benzoic acid is a delicate equilibrium that is driven to completion by refluxing in water, a green solvent choice that simplifies waste management. Finally, the addition of methyl magnesium iodide to the aldehyde-acid precursor is a highly exothermic Grignard reaction that must be managed under inert atmosphere to prevent quenching by moisture. The resulting intermediate spontaneously or upon acid workup undergoes lactonization to form the final product. For the chiral variant, the process employs a TEMPO-mediated oxidation followed by a chiral reduction or resolution step, ensuring that the stereocenter at the 3-position is established with high fidelity, which is critical for the biological activity of the final Laratinib drug substance.
How to Synthesize 5-Fluoro-3-methylisobenzofuranone Efficiently
Executing this synthesis requires strict adherence to the specified reaction parameters to maximize yield and purity. The process begins with the preparation of 4-nitrophthalimide, followed by reduction to the amine and immediate cyclization to the lactam. Operators must maintain precise temperature control during the diazotization phase to prevent the premature decomposition of the diazonium species. The fluorination step involves handling potentially toxic gases like HF and BF3, necessitating appropriate scrubbing systems. Following the formation of the fluoro-lactone, the radical bromination should be monitored via TLC to prevent over-reaction. The final Grignard addition is the most sensitive step, requiring anhydrous conditions and slow addition rates to manage the exotherm. For a detailed, step-by-step breakdown of the standardized operating procedures, including specific molar ratios and workup protocols, please refer to the technical guide below.
- Nitration of phthalimide using mixed acid (H2SO4/HNO3) to form 4-nitrophthalimide.
- Reduction of the nitro group using SnCl2/HCl followed by Zn/NaOH mediated cyclization to form 5-aminoisobenzofuran-1-one.
- Diazotization with NaNO2/HCl and NaBF4, followed by thermal decomposition with Fe powder to introduce the fluorine atom.
- Radical bromination at the 3-position using NBS/BPO, followed by hydrolysis to the aldehyde-acid and final Grignard methylation/lactonization.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthetic route offers compelling advantages that directly address the priorities of procurement managers and supply chain directors. The primary driver for cost reduction is the substitution of complex, custom-synthesized starting materials with phthalimide, a bulk chemical available from multiple global suppliers at competitive prices. This shift significantly de-risks the supply chain by reducing dependency on single-source vendors for exotic precursors. Furthermore, the operational simplicity of the route, characterized by standard unit operations like filtration, reflux, and extraction, minimizes the requirement for specialized equipment or highly skilled labor, thereby lowering the overall manufacturing overhead. The use of common solvents such as water, toluene, and ether further contributes to cost efficiency and simplifies solvent recovery and recycling programs, aligning with modern sustainability goals.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the use of stoichiometric reagents like iron and zinc powders drastically reduces the raw material cost profile. Additionally, the simplified post-treatment procedures, which often involve simple precipitation or crystallization rather than complex chromatography, lead to substantial savings in processing time and consumable costs. This lean manufacturing approach ensures that the cost of goods remains low even when scaling to multi-ton quantities, providing a significant margin advantage for the final API producer.
- Enhanced Supply Chain Reliability: By anchoring the synthesis on phthalimide, manufacturers can leverage a robust and mature supply network for the starting material, ensuring consistent availability and mitigating the risk of shortages. The short reaction times and straightforward workups associated with this method allow for faster batch turnover, enabling facilities to respond more agilely to fluctuating demand signals from downstream API customers. This reliability is crucial for maintaining uninterrupted production schedules for life-saving oncology medications.
- Scalability and Environmental Compliance: The process is inherently scalable, utilizing reaction conditions that are easily translated from laboratory glassware to industrial stainless steel reactors. The avoidance of cryogenic conditions (except for the brief diazotization step) and the use of water as a solvent in the hydrolysis step reduce the energy footprint of the process. Moreover, the generation of inorganic salts as byproducts, which are easier to treat than complex organic waste streams, facilitates compliance with increasingly stringent environmental regulations regarding effluent discharge.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering a clear view of the process capabilities and limitations. Understanding these details is essential for R&D teams evaluating the feasibility of technology transfer and for procurement teams assessing the long-term viability of the supply source.
Q: What are the key advantages of this synthesis route for Laratinib intermediates?
A: This route utilizes phthalimide, a readily available and inexpensive starting material, avoiding the long and costly sequences described in prior art like WO 2013132376. It features simple post-treatment operations such as filtration and crystallization, significantly reducing processing time and solvent consumption.
Q: How is chirality controlled in the production of (S)-5-fluoro-3-methylisobenzofuran-1(3H)-one?
A: Chirality is introduced via a resolution strategy. The racemic alcohol intermediate is oxidized to a ketone, which then undergoes chiral reduction or resolution to obtain the (S)-enantiomer. This is followed by acid-catalyzed lactonization to secure the stereochemistry in the final lactone ring.
Q: Is this process scalable for industrial manufacturing?
A: Yes, the patent explicitly states the method is beneficial for realizing industrialization. The reactions utilize standard reagents like iron powder, zinc, and common solvents (toluene, ether, DCE), and the conditions (mostly ambient to reflux temperatures) are compatible with standard stainless steel reactor setups.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Fluoro-3-methylisobenzofuranone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development and commercialization of advanced oncology therapies. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial plant is seamless and efficient. We are committed to delivering 5-fluoro-3-methylisobenzofuranone with stringent purity specifications, supported by our rigorous QC labs that employ state-of-the-art analytical techniques to verify identity and assay. Our capability to handle complex heterocyclic chemistry, including sensitive Grignard reactions and chiral resolutions, positions us as a strategic partner for pharmaceutical companies seeking to secure their supply chains for Laratinib and related analogs.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic potential of switching to this phthalimide-based pathway. We encourage potential partners to contact us for specific COA data and route feasibility assessments, allowing us to demonstrate our commitment to quality, transparency, and collaborative innovation in the fine chemical sector.
