Advanced Synthesis of High-Nitrogen Phosphazene-DOPO Compounds for Next-Generation Flame Retardants
Advanced Synthesis of High-Nitrogen Phosphazene-DOPO Compounds for Next-Generation Flame Retardants
The landscape of polymer safety is undergoing a critical transformation, driven by stringent environmental regulations and the demand for higher performance materials. A pivotal advancement in this sector is detailed in patent CN103435652A, which outlines a sophisticated preparation method for novel compounds containing both phosphaphenanthrene and phosphazene dual-effect functional groups. This technology represents a significant leap forward in the design of halogen-free flame retardants, addressing the longstanding challenge of balancing fire safety with environmental compliance. By utilizing a branched structure with cyclotriphosphazene as the core and 9,10-dihydro-9-oxa-10-phosphaphenanthrene-10-oxide (DOPO) as side groups, the invention achieves a nitrogen content of up to 6.1%, vastly superior to previous iterations. For R&D directors and procurement specialists in the polymer industry, this patent offers a roadmap to producing high-purity additives that leverage phosphorus-nitrogen synergy for maximum efficiency.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the development of effective flame retardants has been plagued by a trade-off between performance and toxicity. Traditional halogenated flame retardants, while effective, release corrosive and toxic gases upon combustion, leading to their phase-out in many jurisdictions. Earlier generations of phosphorus-based retardants often suffered from low thermal stability or insufficient nitrogen content, limiting their char-forming capabilities. Specifically, prior art methods, such as those cited in the background of the patent, yielded compounds with nitrogen content as low as 1.9%. This low nitrogen level restricts the synergistic potential with phosphorus, resulting in the need for higher loading rates in the final polymer matrix, which can adversely affect the mechanical properties of the plastic. Furthermore, conventional synthesis routes often involved harsh conditions or complex multi-step purifications that hindered commercial scalability and increased the overall cost of goods sold.
The Novel Approach
The methodology presented in CN103435652A fundamentally restructures the molecular architecture to overcome these deficits. By employing hexa(4-amino-phenoxy)-cyclotriphosphazene as a reactive hub, the process allows for the grafting of multiple DOPO units through robust P-O-C linkages. This approach not only drastically increases the nitrogen density within the molecule but also incorporates bulky phenyl groups that enhance thermal stability and compatibility with polymer matrices. The synthesis is streamlined into two primary pathways: a direct grafting via the Atherton-Todd reaction and a condensation via the Kabachnik-Fields reaction. This flexibility allows manufacturers to tailor the final additive properties by selecting different aldehydes in the second pathway. The result is a versatile family of compounds that offer superior flame retardancy at lower loading levels, preserving the physical integrity of the host polymer while ensuring compliance with global safety standards.
Mechanistic Insights into Atherton-Todd and Kabachnik-Fields Functionalization
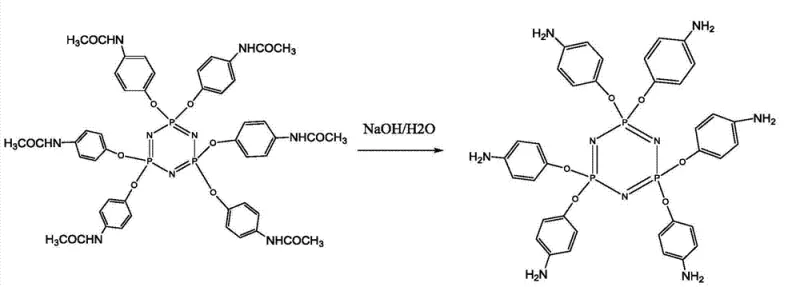
The core chemical innovation lies in the efficient functionalization of the phosphazene ring. The process begins with the preparation of the key intermediate, hexa-(4-aminophenoxy)cyclotriphosphazene (Compound 1). As illustrated in the reaction scheme, this is achieved through the hydrolysis of a functionalized amide precursor using concentrated sodium hydroxide in a polar solvent like acetone. The reaction proceeds at a moderate temperature of 70°C over 10 hours, yielding the highly reactive amine species necessary for subsequent grafting. This step is critical because the six amino groups provide the nucleophilic sites required to attach the bulky DOPO moieties. The high yield and purity of this intermediate are essential for ensuring that the final flame retardant has a consistent molecular weight distribution, which is a key quality metric for R&D teams evaluating batch-to-batch reproducibility.
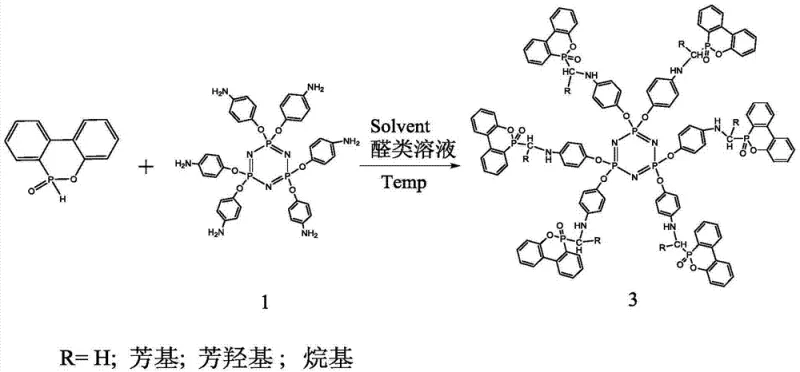
Following the synthesis of Compound 1, the patent describes two distinct mechanisms for introducing the phosphaphenanthrene groups. In the first pathway, Compound 1 reacts with DOPO in the presence of carbon tetrachloride (CCl4) via an Atherton-Todd reaction. This coupling occurs at temperatures around 90°C in ethanol, forming stable P-O-C bonds directly between the amine-derived oxygen and the phosphorus of the DOPO group. Alternatively, the Kabachnik-Fields reaction allows for the introduction of a methylene bridge between the amine and the DOPO unit by reacting with an aldehyde. This pathway, conducted at 70-80°C, offers greater structural diversity, as the 'R' group on the methylene bridge can be varied (hydrogen, aryl, alkyl) to tune solubility and compatibility. Both mechanisms are characterized by high atom economy and generate minimal hazardous byproducts, aligning with green chemistry principles that are increasingly important for supply chain sustainability.
How to Synthesize High-Nitrogen Phosphazene Derivatives Efficiently
Implementing this synthesis route requires precise control over reaction parameters to maximize the nitrogen content and ensure complete conversion of the reactive amines. The process is designed to be operationally simple, avoiding the need for exotic catalysts or extreme pressures. The initial hydrolysis step to generate the amine intermediate must be carefully monitored to prevent degradation of the phosphazene ring, although the patent indicates high stability under the specified alkaline conditions. Subsequent coupling reactions benefit from the high nucleophilicity of the amino groups, driving the reaction to completion with yields exceeding 90% in optimized examples. For technical teams looking to replicate or scale this process, the focus should be on maintaining strict stoichiometric ratios, particularly the excess of DOPO relative to the phosphazene core, to ensure full substitution of the six active sites. Detailed standardized synthesis steps for this high-performance intermediate are provided below.
- Synthesize the key intermediate, hexa-(4-aminophenoxy)cyclotriphosphazene (Compound 1), by hydrolyzing the functionalized amide precursor with concentrated NaOH in acetone at 70°C for 10 hours.
- For Compound 2, react Compound 1 with DOPO and carbon tetrachloride in a polar solvent like ethanol at 90°C for 10 hours via the Atherton-Todd reaction.
- For Compound 3, react Compound 1 with DOPO and an aldehyde solution in a polar solvent at 70-80°C for 10-15 hours using the Kabachnik-Fields addition reaction.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this new class of flame retardants offers tangible strategic benefits beyond mere technical performance. The synthesis route relies on commodity chemicals such as DOPO, hexachlorocyclotriphosphazene, and common aldehydes, all of which are produced at a massive global scale. This reliance on established feedstocks mitigates the risk of raw material shortages that often plague niche specialty chemical markets. Furthermore, the reaction conditions are relatively mild, operating at temperatures below 100°C and using standard polar solvents like ethanol and acetone. This reduces the energy intensity of the manufacturing process compared to high-temperature melt polycondensations, directly contributing to cost reduction in flame retardant manufacturing without compromising on output quality.
- Cost Reduction in Manufacturing: The economic viability of this process is underpinned by its high reaction yields and simplified purification workflow. In the provided examples, the final products are isolated simply by filtration and washing with solvents like acetone or methanol, followed by vacuum drying. This eliminates the need for expensive and time-consuming column chromatography or distillation steps often required for high-purity fine chemicals. By removing these complex downstream processing stages, manufacturers can significantly lower operational expenditures and reduce solvent consumption. Additionally, the high nitrogen content means that less additive is required to achieve the same UL-94 V-0 rating in the final polymer, effectively reducing the cost-in-use for the end customer.
- Enhanced Supply Chain Reliability: The robustness of the chemical synthesis ensures a stable supply of critical polymer additives. Because the reaction tolerances are wide and the reagents are non-sensitive to moisture compared to organometallic catalysts, the risk of batch failure due to minor environmental fluctuations is minimized. This reliability is crucial for maintaining continuous production lines in the plastics and electronics industries, where downtime is prohibitively expensive. The ability to produce these compounds in high purity (reported up to 99.2% in examples) without complex refining steps means that lead times can be shortened, allowing suppliers to respond more agilely to market demand spikes.
- Scalability and Environmental Compliance: From a regulatory and scaling perspective, this technology is exceptionally well-positioned. The process is halogen-free, avoiding the generation of dioxins or furans during both synthesis and the eventual end-of-life incineration of the plastic product. This aligns perfectly with global directives like RoHS and REACH, future-proofing the supply chain against tightening environmental legislation. The scalability is further enhanced by the use of solution-phase reactions which are easily transferred from laboratory glassware to industrial stainless steel reactors. The waste stream primarily consists of recoverable solvents and benign salts, simplifying wastewater treatment and reducing the environmental footprint of the manufacturing facility.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this phosphazene-DOPO technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing a factual basis for decision-making. Understanding these nuances is essential for stakeholders evaluating the feasibility of integrating these novel additives into their existing product portfolios.
Q: What is the primary advantage of the phosphazene-DOPO compounds described in CN103435652A?
A: The primary advantage is the significantly higher nitrogen content, reaching up to 6.1%, compared to previous generations which typically contained around 1.9%. This high nitrogen content, combined with the phosphorus from DOPO and the phosphazene core, creates a powerful synergistic effect that enhances char formation and flame retardant efficiency while remaining halogen-free.
Q: How does the new synthesis method improve supply chain reliability for polymer additives?
A: The method utilizes readily available starting materials like DOPO and hexachlorocyclotriphosphazene derivatives. The reaction conditions are mild (70-90°C) and the purification process involves simple filtration and washing with common solvents like acetone and methanol, eliminating the need for complex chromatographic separations and ensuring consistent, scalable production.
Q: Are these compounds suitable for halogen-free applications?
A: Yes, absolutely. The design specifically targets halogen-free flame retardancy. By relying on the P-N synergy between the phosphaphenanthrene groups and the phosphazene ring, these compounds achieve high fire resistance without releasing toxic halogenated gases, making them ideal for eco-friendly electronic materials and construction polymers.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polymer Additive Supplier
The synthesis of high-nitrogen phosphazene-DOPO derivatives represents a sophisticated intersection of organic synthesis and materials science, requiring a partner with deep technical expertise. NINGBO INNO PHARMCHEM stands ready to support your transition to these advanced flame retardants with our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facilities are equipped with rigorous QC labs capable of verifying the stringent purity specifications required for high-performance polymer applications, ensuring that every batch meets the exacting standards of the global electronics and automotive sectors. We understand that consistency is key, and our process engineering teams are dedicated to optimizing these specific Atherton-Todd and Kabachnik-Fields reactions for maximum efficiency and yield.
We invite you to collaborate with us to unlock the full potential of this patented technology for your specific application needs. Whether you require custom modifications to the 'R' group for enhanced compatibility or need a Customized Cost-Saving Analysis for switching from traditional halogenated systems, our technical team is prepared to assist. Please contact our technical procurement team today to request specific COA data and route feasibility assessments tailored to your project requirements. Let us help you build a safer, more sustainable, and cost-effective supply chain for your next generation of flame-retardant materials.
