Advanced Synthesis of N-Substituted 1,2-Diphenylethylamine Compounds for Scalable Pharmaceutical Manufacturing
Introduction to Advanced Diphenylethylamine Synthesis
The pharmaceutical and fine chemical industries continuously seek robust synthetic pathways for complex amine intermediates, particularly N-substituted 1,2-diphenylethylamine compounds which serve as critical building blocks for a wide array of bioactive molecules. Patent CN101475491B introduces a groundbreaking methodology that addresses longstanding challenges in synthesizing these structurally diverse amines. This technology leverages a unique three-step sequence involving nitrone intermediates, offering a distinct advantage over conventional routes by utilizing mild reaction conditions and readily available starting materials. The structural versatility of this approach allows for extensive substitution patterns on the phenyl rings, making it highly applicable for the development of new drug candidates including beta-blockers and antitumor agents. By shifting the synthetic paradigm from unstable imines to stable nitrones, this process significantly enhances the reliability of production for high-value pharmaceutical intermediates.
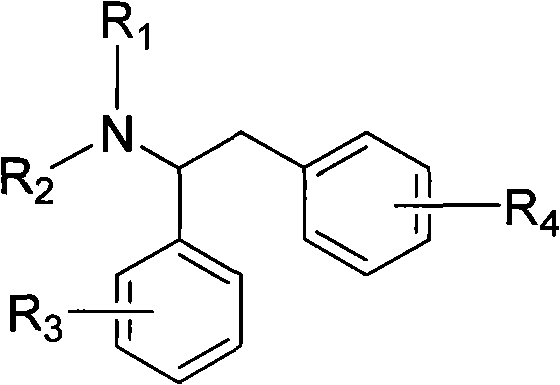
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for 1,2-diphenylethylamine derivatives often rely on the direct addition of Grignard reagents to imines or the reduction of nitrostyrenes, both of which present significant operational hurdles in a commercial setting. The direct addition to imines frequently suffers from poor regioselectivity and the inherent instability of the imine bond, which can lead to hydrolysis or polymerization under the rigorous conditions required for Grignard reactions. Furthermore, the nitrostyrene reduction pathway often necessitates the use of hazardous reagents and high-pressure hydrogenation equipment, introducing safety risks and complicating waste management protocols. These conventional methods often result in lower yields due to competitive side reactions and require extensive purification steps to remove heavy metal residues or polymeric byproducts, thereby inflating the cost of goods and extending production lead times for critical API intermediates.
The Novel Approach
In stark contrast, the novel methodology disclosed in the patent utilizes a nitrone intermediate strategy that fundamentally alters the reaction landscape for improved efficiency and safety. By first condensing an aromatic aldehyde with a hydroxylamine derivative, the process generates a stable nitrone species that acts as a superior electrophile for subsequent nucleophilic attack. This intermediate is robust enough to withstand isolation and purification, ensuring high purity before the critical carbon-carbon bond-forming step. The subsequent addition of organometallic reagents proceeds under much milder thermal conditions compared to traditional imine chemistry, drastically reducing energy consumption and equipment stress. This streamlined approach not only simplifies the workflow but also opens up a broader scope of compatible functional groups, enabling the synthesis of complex derivatives that were previously difficult to access via standard protocols.
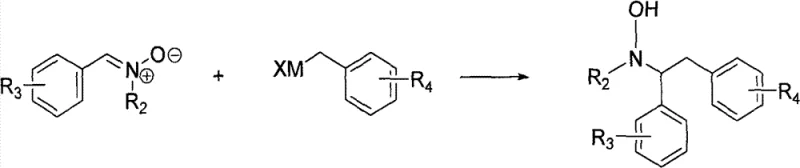
Mechanistic Insights into Nitrone-Based Coupling and Reduction
The core mechanistic advantage of this synthesis lies in the electronic properties of the nitrone functional group, which facilitates a highly controlled nucleophilic addition. In the second step of the process, the organometallic reagent, typically a Grignard or organolithium species, attacks the electrophilic carbon of the C=N bond in the nitrone. Unlike imines, nitrones possess an adjacent oxygen atom that stabilizes the transition state through resonance, lowering the activation energy required for the addition. This results in a hydroxylamine adduct with excellent stereochemical integrity and minimal formation of homocoupling byproducts. The reaction can be finely tuned by adjusting the temperature between -80°C and 100°C, allowing chemists to optimize kinetics for specific substrate combinations without compromising the structural integrity of sensitive functional groups on the aromatic rings.
Following the coupling step, the conversion of the hydroxylamine intermediate to the final amine product is achieved through a versatile reduction protocol that supports both chemical and catalytic methods. The patent details the use of metal reducing agents such as zinc powder or stannous chloride in the presence of acid, which effectively cleaves the N-O bond to release the free amine. Alternatively, transition metal catalysts like palladium on carbon or Raney nickel can be employed under a hydrogen atmosphere, offering a greener alternative with potentially easier workup procedures. This dual-pathway flexibility is crucial for impurity control, as it allows manufacturers to select the reduction method that best minimizes specific byproduct profiles for their target molecule, ensuring the final API intermediate meets stringent regulatory purity specifications.

How to Synthesize N-Substituted 1,2-Diphenylethylamine Efficiently
Implementing this synthesis route requires careful attention to solvent selection and stoichiometric ratios to maximize yield and minimize waste. The process begins with the condensation of aromatic aldehydes and hydroxylamine derivatives in solvents such as tetrahydrofuran or dichloromethane, typically requiring a molar ratio of 1:1 to 1:5 to drive the equilibrium towards nitrone formation. Once the nitrone is secured, the addition of the organometallic reagent must be performed under inert atmosphere to prevent quenching by moisture, followed by a controlled reduction step that can be adapted based on available plant infrastructure. The detailed standardized synthesis steps below outline the precise operational parameters derived from the patent examples to ensure reproducible high-quality output.
- Condense aromatic aldehydes with hydroxylamine derivatives in organic solvents at 0-150°C to form stable nitrone intermediates.
- React the isolated nitrone with organometallic reagents (Grignard or Organolithium) in aprotic solvents at -80 to 100°C to generate hydroxylamine adducts.
- Reduce the hydroxylamine intermediate using metal reducing agents like zinc or transition metal catalysts under hydrogen to yield the final amine.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, this synthetic route offers substantial benefits by decoupling production from volatile supply chains associated with exotic catalysts or hazardous precursors. The reliance on commodity chemicals such as benzaldehydes and simple hydroxylamines ensures a stable and cost-effective supply of raw materials, mitigating the risk of production stoppages due to原料 shortages. Furthermore, the operational simplicity of the process reduces the dependency on specialized high-pressure reactors or cryogenic equipment, allowing for manufacturing in standard multipurpose facilities which lowers capital expenditure and overhead costs significantly. This accessibility translates directly into more competitive pricing structures for downstream pharmaceutical clients seeking reliable long-term partners for intermediate supply.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts in the coupling step and the ability to use common metal powders for reduction drastically lowers the direct material costs per kilogram of product. Additionally, the mild reaction conditions reduce energy consumption for heating and cooling, while the high selectivity of the nitrone route minimizes the loss of valuable starting materials to side products. This efficiency gain means less solvent is required for purification, further driving down waste disposal costs and improving the overall economic viability of large-scale production runs.
- Enhanced Supply Chain Reliability: By utilizing a three-step sequence with isolable intermediates, the process builds in quality checkpoints that prevent the propagation of impurities to the final stage, ensuring consistent batch-to-batch quality. The robustness of the nitrone intermediate allows for potential stockpiling, providing a buffer against supply chain disruptions and enabling just-in-time delivery models for urgent client demands. This flexibility is critical for maintaining continuity in the production of life-saving medications where intermediate availability is often the bottleneck.
- Scalability and Environmental Compliance: The process avoids the generation of nitro-containing waste streams associated with nitrostyrene reductions, simplifying environmental compliance and wastewater treatment requirements. The use of standard organic solvents that are easily recoverable through distillation supports green chemistry initiatives and reduces the environmental footprint of the manufacturing site. This alignment with sustainability goals not only meets regulatory standards but also enhances the brand value of the supply chain for eco-conscious pharmaceutical partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this nitrone-based synthesis technology. These insights are derived directly from the experimental data and claims within the patent documentation to provide clarity on feasibility and performance. Understanding these details helps stakeholders make informed decisions about integrating this chemistry into their existing manufacturing portfolios.
Q: What are the primary advantages of the nitrone route over traditional imine reduction?
A: The nitrone route offers superior stability compared to imines, allowing for milder reaction conditions and reduced side reactions such as polymerization, leading to higher overall purity and yield.
Q: Is this synthesis method scalable for industrial production?
A: Yes, the process utilizes readily available raw materials and standard solvents like THF and dichloromethane, with reaction temperatures ranging from ambient to moderate heating, facilitating safe commercial scale-up.
Q: What reducing agents are compatible with this protocol?
A: The patent specifies flexibility in reduction methods, supporting both chemical reduction using zinc or tin powders and catalytic hydrogenation using palladium or nickel catalysts, allowing optimization based on facility capabilities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Substituted 1,2-Diphenylethylamine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development of new therapeutic agents. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of N-substituted 1,2-diphenylethylamine meets the exacting standards required by global regulatory bodies. Our commitment to technical excellence ensures that your supply chain remains robust, reliable, and capable of supporting your most ambitious drug development timelines.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this nitrone-based methodology for your specific target molecules. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, allowing us to demonstrate concretely how our manufacturing capabilities can drive value and efficiency for your organization.
