Advanced Synthesis of 4,4,4-Trifluoro-2-Butenoate for Commercial Pharmaceutical Applications
The pharmaceutical and agrochemical industries are constantly seeking robust methods to incorporate fluorine atoms into organic scaffolds, as the trifluoromethyl group significantly enhances metabolic stability and lipophilicity. Patent CN102911054A discloses a highly efficient preparation method for 4,4,4-trifluoro-2-butenoate, a critical building block for advanced functional materials and bioactive compounds. This technology addresses the longstanding challenges associated with introducing trifluoromethyl groups under mild conditions, utilizing a novel combination of Togni reagents and zinc iodide catalysis. The process described in the patent not only achieves high product purity but also ensures that the reaction solvents are easily recoverable and recyclable, aligning with modern green chemistry principles. For R&D directors and procurement specialists, this represents a significant opportunity to optimize supply chains for fluorine-containing intermediates while maintaining stringent quality standards. The strategic implementation of this synthesis route can lead to substantial improvements in process reliability and cost-efficiency for downstream applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional methods for synthesizing beta-trifluoromethyl-alpha,beta-unsaturated esters often rely on harsh reaction conditions that can compromise the integrity of sensitive functional groups. Many conventional routes require the use of gaseous trifluoromethylating agents which pose significant safety hazards and require specialized equipment for handling and containment. Furthermore, older methodologies frequently suffer from poor regioselectivity and low yields, necessitating complex purification steps that increase both production time and waste generation. The use of strong bases or high temperatures in these traditional processes can lead to unwanted side reactions, resulting in impurity profiles that are difficult to manage during scale-up. These factors collectively contribute to higher operational costs and longer lead times, creating bottlenecks for manufacturers aiming to bring fluorine-containing products to market efficiently. Consequently, there is a critical need for alternative synthetic strategies that offer better control over reaction parameters and improved safety profiles.
The Novel Approach
The methodology outlined in the patent introduces a transformative two-step sequence that overcomes the drawbacks of legacy technologies by employing stable solid reagents and mild catalytic conditions. By utilizing Togni reagents as the trifluoromethyl source, the process eliminates the need for hazardous gases and allows for precise stoichiometric control during the addition phase. The subsequent elimination step uses DBU at room temperature, which preserves the stereochemistry of the double bond and prevents thermal degradation of the product. This approach simplifies the workflow significantly, as the reaction can be conducted under normal atmospheric pressure without the need for cryogenic cooling or high-pressure vessels. The ability to recycle solvents such as chloroform and dichloromethane further enhances the economic viability of this route, making it an attractive option for cost-sensitive manufacturing environments. Overall, this novel approach provides a streamlined pathway to high-value fluorochemical intermediates with superior consistency and reliability.
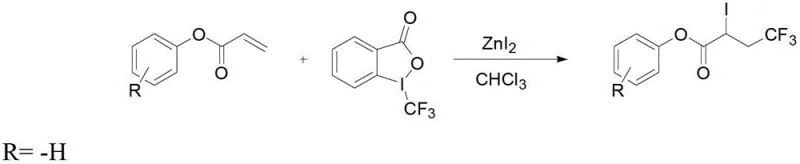
Mechanistic Insights into ZnI2-Catalyzed Trifluoromethylation and Elimination
The core of this synthesis lies in the zinc iodide-catalyzed radical or ionic trifluoromethylation of the acrylate double bond, which proceeds with high regioselectivity to form the 4,4,4-trifluoro-2-iodobutyrate intermediate. The zinc iodide acts as a Lewis acid to activate the Togni reagent, facilitating the transfer of the trifluoromethyl group to the beta-position of the acrylate substrate. This activation mechanism ensures that the reaction proceeds smoothly under reflux conditions, typically completing within 1 to 3 hours depending on the specific substituents on the aromatic ring. The presence of the iodine atom at the alpha-position is crucial for the subsequent elimination step, serving as an excellent leaving group that enables the formation of the double bond under mild basic conditions. Understanding this mechanistic pathway is essential for R&D teams aiming to adapt this chemistry for diverse substrates, as electronic effects from substituents like methoxy or nitro groups can influence reaction rates and yields. The robustness of this catalytic system allows for a broad scope of application, making it a versatile tool in the synthetic chemist's arsenal.
Following the initial trifluoromethylation, the elimination of hydrogen iodide is mediated by 1,8-diazabicyclo[5.4.0]undec-7-ene (DBU), a non-nucleophilic base that promotes E2 elimination without attacking the ester functionality. This step is remarkably fast, often reaching completion within 10 to 30 minutes at room temperature, which minimizes energy consumption and exposure time. The mildness of this condition is particularly beneficial for preserving sensitive functional groups that might otherwise decompose under stronger basic or thermal stress. The resulting 4,4,4-trifluoro-2-butenoate features a conjugated system where the strong electron-withdrawing trifluoromethyl group enhances the reactivity of the double bond for further downstream transformations. This increased reactivity is advantageous for subsequent conjugate addition reactions or polymerization processes, expanding the utility of the final product in material science and drug discovery. The combination of these two distinct chemical transformations creates a powerful synergy that drives the overall efficiency of the synthesis.
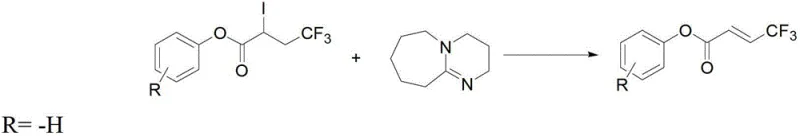
How to Synthesize 4,4,4-Trifluoro-2-Butenoate Efficiently
Implementing this synthesis route requires careful attention to the stoichiometry of reagents and the purity of solvents to ensure optimal yields and reproducibility. The patent specifies a molar ratio of 1:2:2 for acrylate, trifluoromethylation reagent, and iodide, which is critical for driving the first step to completion while minimizing excess reagent waste. Operators must maintain an inert nitrogen atmosphere throughout the reaction to prevent oxidation of sensitive intermediates and ensure the stability of the catalytic species. Detailed standardized synthesis steps see the guide below for precise operational parameters regarding temperature control and workup procedures. Adhering to these guidelines will help manufacturing teams achieve consistent batch quality and maximize the throughput of their production lines. Proper training on handling Togni reagents and zinc salts is also recommended to maintain safety standards in the laboratory and plant environments.
- Dissolve acrylate and Togni reagent in organic solvent under nitrogen, add zinc iodide, and reflux for 1-3 hours to form the iodo-intermediate.
- Quench the reaction with water, extract with dichloromethane, dry, and purify via column chromatography to isolate 4,4,4-trifluoro-2-iodobutyrate.
- React the iodo-intermediate with DBU in dichloromethane at room temperature for 10-30 minutes, then purify to obtain the final 4,4,4-trifluoro-2-butenoate.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthesis route offers compelling benefits that directly address the pain points of procurement managers and supply chain leaders in the fine chemical sector. The elimination of hazardous gaseous reagents reduces the regulatory burden and insurance costs associated with storing and handling dangerous materials, thereby lowering the total cost of ownership for the manufacturing process. Furthermore, the ability to recover and recycle solvents like chloroform and dichloromethane significantly reduces raw material consumption and waste disposal fees, contributing to a more sustainable and cost-effective operation. The simplicity of the reaction conditions, which operate at atmospheric pressure and moderate temperatures, allows for the use of standard glass-lined or stainless-steel reactors without the need for specialized high-pressure equipment. These factors combine to create a manufacturing process that is not only economically advantageous but also resilient to supply chain disruptions related to specialized reagents or equipment.
- Cost Reduction in Manufacturing: The use of stable solid reagents instead of gases eliminates the need for expensive containment systems and reduces safety compliance costs significantly. Additionally, the high selectivity of the reaction minimizes the formation of by-products, which reduces the load on purification units and lowers solvent usage during chromatography. The recyclability of the reaction medium further drives down variable costs, allowing for better margin protection in competitive markets. By streamlining the synthesis into two straightforward steps, labor hours and utility consumption are also optimized, leading to a leaner production model.
- Enhanced Supply Chain Reliability: Sourcing stable solid reagents like Togni reagents and zinc iodide is generally more reliable than securing compressed gases, which can be subject to transportation restrictions and supply volatility. The robustness of the reaction conditions means that production is less susceptible to minor fluctuations in environmental parameters, ensuring consistent output quality. This stability allows for more accurate forecasting and inventory planning, reducing the risk of stockouts for critical downstream customers. Moreover, the scalability of the process ensures that supply can be ramped up quickly to meet surges in demand without compromising on product specifications.
- Scalability and Environmental Compliance: The process operates under normal pressure and uses common organic solvents that are well-understood in terms of environmental impact and waste treatment. The absence of heavy metal catalysts or toxic gases simplifies the effluent treatment process, making it easier to meet strict environmental regulations in various jurisdictions. The short reaction times and mild conditions also reduce the energy footprint of the manufacturing process, aligning with corporate sustainability goals. This environmental compatibility facilitates smoother regulatory approvals and enhances the brand reputation of the manufacturer as a responsible supplier.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route for 4,4,4-trifluoro-2-butenoate. These answers are derived directly from the technical specifications and beneficial effects described in the patent documentation to ensure accuracy and relevance. Understanding these details is crucial for stakeholders evaluating the feasibility of integrating this chemistry into their existing production portfolios. The information provided here aims to clarify the operational advantages and technical nuances that define the value proposition of this method.
Q: What are the key advantages of using Togni reagent in this synthesis?
A: Togni reagent provides a stable and efficient source of trifluoromethyl groups, enabling mild reaction conditions and high product purity compared to traditional gaseous reagents.
Q: How does the DBU elimination step impact overall yield?
A: The DBU-mediated elimination occurs rapidly at room temperature, minimizing side reactions and thermal degradation, which preserves the integrity of the sensitive double bond.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the use of recyclable solvents and standard atmospheric pressure conditions makes this method highly adaptable for commercial scale-up with reduced environmental impact.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4,4,4-Trifluoro-2-Butenoate Supplier
NINGBO INNO PHARMCHEM stands ready to leverage this advanced synthesis technology to deliver high-quality fluorochemical intermediates to the global market. As a seasoned CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our facilities are equipped with stringent purity specifications and rigorous QC labs to guarantee that every batch meets the highest industry standards for pharmaceutical and agrochemical applications. We understand the critical nature of supply chain continuity and are committed to providing a stable source of this valuable intermediate for your long-term projects.
We invite you to engage with our technical procurement team to discuss how this synthesis route can be optimized for your specific requirements. Request a Customized Cost-Saving Analysis to understand the potential economic benefits of switching to this method for your production needs. Our team is prepared to provide specific COA data and route feasibility assessments to support your decision-making process. By partnering with us, you gain access to a wealth of technical expertise and manufacturing capacity dedicated to advancing your chemical supply chain.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
