Advanced Ferrocene Imine Palladacycle Catalysis for Scalable Aryl Nitrile Production
Introduction to Next-Generation Cyanation Technology
The landscape of organic intermediate synthesis is undergoing a significant transformation driven by the dual imperatives of safety and efficiency, a shift exemplified by the innovations detailed in patent CN101113138A. This pivotal intellectual property introduces a sophisticated synthesis method for aryl nitrile derivatives utilizing a ferrocene imine cyclopalladium complex as the primary catalyst. Unlike traditional methodologies that rely on hazardous cyanide sources, this approach leverages potassium ferrocyanide, a non-toxic and environmentally benign nitrile donor, thereby fundamentally altering the risk profile of large-scale nitrile production. The technology addresses critical pain points in the pharmaceutical and agrochemical sectors by offering a pathway that combines mild reaction conditions with exceptional reaction specificity and high yields. For R&D directors and process chemists, this represents a viable route to high-purity aryl nitrile derivatives that minimizes waste generation and simplifies downstream purification processes. The integration of a robust ferrocene-based palladacycle ensures stability and reactivity that surpasses conventional palladium sources, making it an attractive option for reliable aryl nitrile supplier networks seeking to modernize their manufacturing capabilities.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of substituted aromatic nitriles has been fraught with significant safety and operational challenges that hinder efficient cost reduction in pharmaceutical intermediates manufacturing. Traditional protocols frequently employ alkali metal cyanides such as KCN or NaCN, which are acutely toxic and pose severe environmental pollution risks requiring stringent containment and waste treatment measures. Alternative methods utilizing transition metal cyanides like CuCN or Zn(CN)2 generate substantial amounts of heavy metal waste, complicating disposal and increasing the overall ecological footprint of the process. Furthermore, existing palladium-catalyzed methods often necessitate the use of specialized and prohibitively expensive bisphosphine ligands to achieve acceptable conversion rates, driving up raw material costs. Additionally, standard catalysts like Pd(OAc)2 typically exhibit limited substrate scope, often failing to react efficiently with anything other than brominated aromatic compounds, thus restricting the versatility of the synthetic route. These cumulative factors create a bottleneck for supply chain heads who require consistent, scalable, and compliant production methods for complex organic intermediates.
The Novel Approach
The innovative methodology described in the patent circumvents these legacy issues by employing a ferrocenylimine cyclopalladated complex that operates effectively with a wide range of halogenated aromatic compounds. This novel approach utilizes potassium ferrocyanide as the cyanide source, which is not only non-toxic but also stable, eliminating the immediate hazards associated with free cyanide ions. The reaction system is designed to be highly versatile, accommodating various substituents on the aromatic ring, including electron-donating and electron-withdrawing groups, without compromising yield or purity. 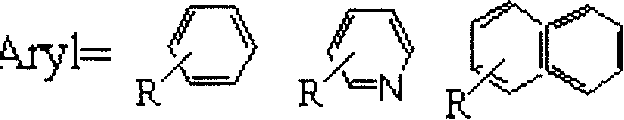 As illustrated in the substrate scope, the process tolerates groups such as methyl, methoxy, nitro, and acetyl, demonstrating remarkable chemoselectivity. The use of common organic solvents like N,N-dimethylformamide or N,N-dimethylacetamide further enhances the practicality of the method, allowing for seamless integration into existing reactor infrastructure. This broad applicability ensures that manufacturers can produce a diverse library of nitrile derivatives from a single, optimized platform, significantly reducing lead time for high-purity nitriles and streamlining the supply chain for downstream API synthesis.
As illustrated in the substrate scope, the process tolerates groups such as methyl, methoxy, nitro, and acetyl, demonstrating remarkable chemoselectivity. The use of common organic solvents like N,N-dimethylformamide or N,N-dimethylacetamide further enhances the practicality of the method, allowing for seamless integration into existing reactor infrastructure. This broad applicability ensures that manufacturers can produce a diverse library of nitrile derivatives from a single, optimized platform, significantly reducing lead time for high-purity nitriles and streamlining the supply chain for downstream API synthesis.
Mechanistic Insights into Ferrocene Imine Cyclopalladium Catalysis
The core of this technological breakthrough lies in the unique structure and reactivity of the ferrocene imine cyclopalladium complex, which serves as a highly efficient pre-catalyst for the cyanation reaction. 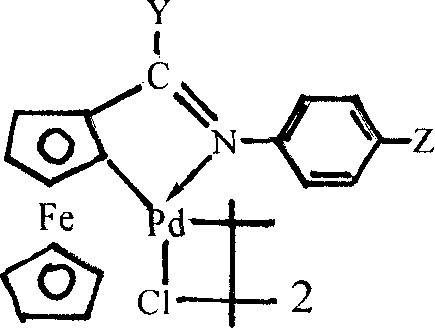 The catalyst features a robust ferrocene backbone coordinated with a palladium center through a cyclometalated imine ligand, creating a stable yet reactive species capable of facilitating the oxidative addition of aryl halides. This specific coordination environment stabilizes the palladium center against aggregation and deactivation, which are common failure modes in traditional palladium-catalyzed cross-coupling reactions. The mechanism likely proceeds through a standard catalytic cycle involving oxidative addition of the aryl halide to the Pd(II) center, followed by transmetallation with the ferrocyanide anion to transfer the cyano group. The final reductive elimination step releases the desired aryl nitrile product and regenerates the active palladium species. The presence of the ferrocene moiety may also impart electronic properties that lower the activation energy for the transmetallation step, which is often the rate-determining step in cyanation reactions using hexacyanoferrate sources.
The catalyst features a robust ferrocene backbone coordinated with a palladium center through a cyclometalated imine ligand, creating a stable yet reactive species capable of facilitating the oxidative addition of aryl halides. This specific coordination environment stabilizes the palladium center against aggregation and deactivation, which are common failure modes in traditional palladium-catalyzed cross-coupling reactions. The mechanism likely proceeds through a standard catalytic cycle involving oxidative addition of the aryl halide to the Pd(II) center, followed by transmetallation with the ferrocyanide anion to transfer the cyano group. The final reductive elimination step releases the desired aryl nitrile product and regenerates the active palladium species. The presence of the ferrocene moiety may also impart electronic properties that lower the activation energy for the transmetallation step, which is often the rate-determining step in cyanation reactions using hexacyanoferrate sources.
From an impurity control perspective, the high specificity of this catalyst system is paramount for producing high-purity aryl nitrile derivatives required in regulated industries. The strong chelation of the imine ligand prevents the formation of uncoordinated palladium black, which can catalyze unwanted side reactions such as homocoupling of the aryl halide or hydrodehalogenation. Furthermore, the mild reaction conditions, typically ranging from 100°C to 150°C, minimize thermal degradation of sensitive functional groups on the substrate, ensuring a cleaner crude reaction profile. This inherent selectivity reduces the burden on downstream purification units, such as chromatography or crystallization, leading to higher overall recovery rates and reduced solvent consumption. For quality assurance teams, this translates to a more predictable impurity profile and easier validation of the manufacturing process, aligning perfectly with the stringent purity specifications demanded by global regulatory bodies.
How to Synthesize Aryl Nitrile Derivatives Efficiently
The practical implementation of this synthesis route is straightforward and amenable to standard laboratory and pilot plant equipment, facilitating rapid technology transfer and commercial scale-up of complex organic intermediates. The process begins with the preparation of the specific ferrocene acetyl substituted phenylimine cyclopalladated derivative, which is synthesized by reacting palladium chloride and lithium chloride with the corresponding ferrocene imine ligand in methanol. Once the catalyst is prepared, the main reaction involves dissolving the catalyst, potassium ferrocyanide, and a base such as sodium carbonate in a polar aprotic solvent like N,N-dimethylacetamide. The halogenated aromatic substrate is then added under an inert nitrogen atmosphere, and the mixture is heated to the requisite temperature, typically around 120°C, for a duration of 1 to 20 hours depending on the reactivity of the specific substrate. Detailed standardized synthesis steps see the guide below.
- Prepare the ferrocene acetyl substituted phenylimine cyclopalladated derivative catalyst by reacting PdCl2 and LiCl with the ferrocene imine ligand in methanol.
- Dissolve the catalyst, potassium ferrocyanide, and a base such as sodium carbonate in an organic solvent like N,N-dimethylacetamide under nitrogen protection.
- Add the halogenated aromatic compound, heat the mixture to 100-150°C for 1-20 hours, then cool, wash with water, extract, and purify to obtain the aryl nitrile.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain strategists, the adoption of this ferrocene-catalyzed cyanation technology offers compelling economic and operational benefits that directly impact the bottom line. The shift away from toxic alkali cyanides to potassium ferrocyanide drastically simplifies EHS compliance and reduces the costs associated with hazardous waste disposal and worker safety monitoring. Moreover, the elimination of expensive bisphosphine ligands, which are often required in conventional palladium-catalyzed cyanations, results in significant raw material cost savings without sacrificing catalytic activity. The robustness of the catalyst allows for lower loading levels, typically between 0.1 to 5 mole percent, further optimizing the cost structure of the final product. These factors combined contribute to a more resilient supply chain capable of withstanding regulatory pressures and raw material price volatility.
- Cost Reduction in Manufacturing: The economic advantages of this process are multifaceted, stemming primarily from the substitution of high-cost reagents with affordable, commodity-grade chemicals. By utilizing potassium ferrocyanide instead of specialized cyanating agents, manufacturers avoid the premium pricing and supply constraints often associated with toxic cyanide salts. Additionally, the catalyst system does not require ancillary ligands that are costly and sensitive to air and moisture, thereby reducing inventory holding costs and waste. The high yields reported in the patent examples, often exceeding 90%, mean that less raw material is wasted, and the throughput per batch is maximized. This efficiency translates directly into a lower cost of goods sold (COGS), providing a competitive edge in the market for fine chemical intermediates.
- Enhanced Supply Chain Reliability: The reliance on widely available and stable raw materials enhances the reliability of the supply chain, mitigating the risk of production stoppages due to reagent shortages. Potassium ferrocyanide and common aryl halides are produced on a massive industrial scale, ensuring a steady flow of inputs even during market fluctuations. The mild reaction conditions also reduce the stress on reactor vessels and auxiliary equipment, extending asset life and minimizing unplanned maintenance downtime. For supply chain heads, this predictability is crucial for meeting delivery commitments to downstream pharmaceutical clients who operate on tight Just-In-Time schedules. The ability to source materials locally and process them safely adds a layer of security to the global supply network.
- Scalability and Environmental Compliance: Scaling this process from gram to ton scale is facilitated by the use of standard solvents and simple workup procedures involving water washing and extraction. The absence of highly toxic reagents simplifies the permitting process for new manufacturing lines and reduces the regulatory burden on existing facilities. Environmental compliance is significantly improved as the process generates less hazardous waste, aligning with green chemistry principles and corporate sustainability goals. The ease of purification, often achievable through simple crystallization or column chromatography as demonstrated in the examples, ensures that the final product meets rigorous quality standards with minimal environmental impact. This scalability ensures that the technology can grow with demand, supporting the commercial expansion of key therapeutic areas.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this ferrocene-catalyzed cyanation technology, based on the detailed experimental data provided in the patent literature. These insights are intended to clarify the operational parameters and benefits for potential partners evaluating this synthetic route for their own portfolios. Understanding the nuances of catalyst loading, substrate scope, and safety profiles is essential for making informed decisions about process adoption. We encourage technical teams to review these answers as a starting point for deeper discussions regarding specific project requirements.
Q: Why is potassium ferrocyanide preferred over traditional cyanide sources?
A: Potassium ferrocyanide is significantly less toxic than KCN or NaCN, reducing environmental hazards and safety risks associated with handling highly toxic cyanide salts in large-scale manufacturing.
Q: What are the advantages of the ferrocene imine cyclopalladium complex catalyst?
A: This catalyst eliminates the need for expensive bisphosphine ligands, offers broader substrate scope including electron-deficient aromatics, and operates under milder conditions compared to traditional Pd(OAc)2 systems.
Q: Can this method tolerate sensitive functional groups like nitro or acetyl?
A: Yes, the patent demonstrates high compatibility with various substituents including nitro, acetyl, and methoxy groups, achieving high yields without significant side reactions or deprotection issues.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aryl Nitrile Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the ferrocene imine cyclopalladium complex in driving innovation within the fine chemical sector. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab bench to full-scale manufacturing is seamless and efficient. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that validate every batch against the highest industry standards. We understand that the synthesis of aryl nitrile derivatives is often a critical step in the production of life-saving medications and crop protection agents, and we treat every project with the urgency and precision it deserves. Our team of expert chemists is ready to adapt this patented methodology to your specific molecular targets, optimizing conditions to maximize yield and minimize impurities.
We invite you to collaborate with us to unlock the full commercial potential of your aryl nitrile projects through our tailored manufacturing solutions. Please contact our technical procurement team to request a Customized Cost-Saving Analysis that details how implementing this greener, more efficient route can improve your margin structure. We are prepared to provide specific COA data from our pilot runs and comprehensive route feasibility assessments to support your regulatory filings. By partnering with NINGBO INNO PHARMCHEM, you gain access to a reliable aryl nitrile supplier dedicated to excellence, sustainability, and long-term strategic growth in the global pharmaceutical marketplace.
