Scalable Synthesis of Trifluoromethyl-Substituted Bismaleimide for High-Performance Aerospace Composites
Scalable Synthesis of Trifluoromethyl-Substituted Bismaleimide for High-Performance Aerospace Composites
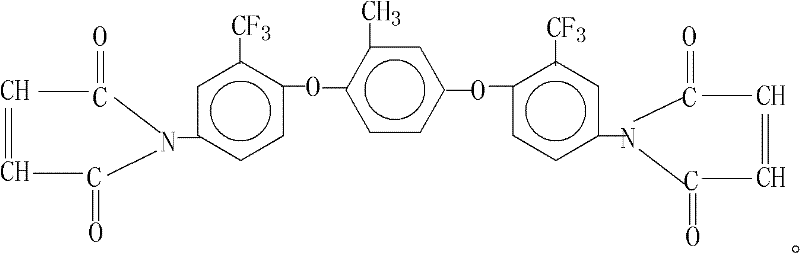
The development of advanced thermosetting resins capable of withstanding extreme thermal and mechanical stress is a cornerstone of modern aerospace engineering, and patent CN101941933B presents a breakthrough methodology for producing a highly specialized bismaleimide monomer known as 2-methyl-1,4-bis(2-trifluoromethyl-4-maleimidophenoxy)benzene. This specific chemical architecture integrates robust imide rings with electron-withdrawing trifluoromethyl groups, creating a molecular framework that offers exceptional thermal stability and reactivity for next-generation composite materials. The patent details a refined synthetic route that overcomes historical limitations in yield and purity, utilizing a unique catalytic system to facilitate efficient cyclodehydration under atmospheric conditions. By leveraging this technology, manufacturers can access a critical precursor for high-performance polyimides and epoxy-modified systems that are essential for aircraft structures, satellite components, and advanced electronic insulation. The following analysis dissects the technical merits and commercial viability of this process for global supply chain stakeholders.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis pathways for bismaleimide resins often rely on straightforward thermal dehydration of bismaleamic acids, a process that is frequently plagued by incomplete conversion and the formation of stubborn impurities that degrade the final material's performance. In many legacy protocols, the removal of water generated during the imidization step requires excessively high temperatures or prolonged reaction times, which can inadvertently trigger premature polymerization or degradation of the sensitive maleimide double bonds. Furthermore, conventional methods often struggle to achieve high purity without resorting to complex and costly recrystallization sequences or chromatographic purification, which are impractical for large-scale industrial production. The inability to effectively drive the equilibrium towards the cyclic imide structure often results in residual amic acid content, which acts as a defect site in the cured polymer network, compromising the thermal and mechanical integrity of the resulting composite materials used in critical aerospace applications.
The Novel Approach
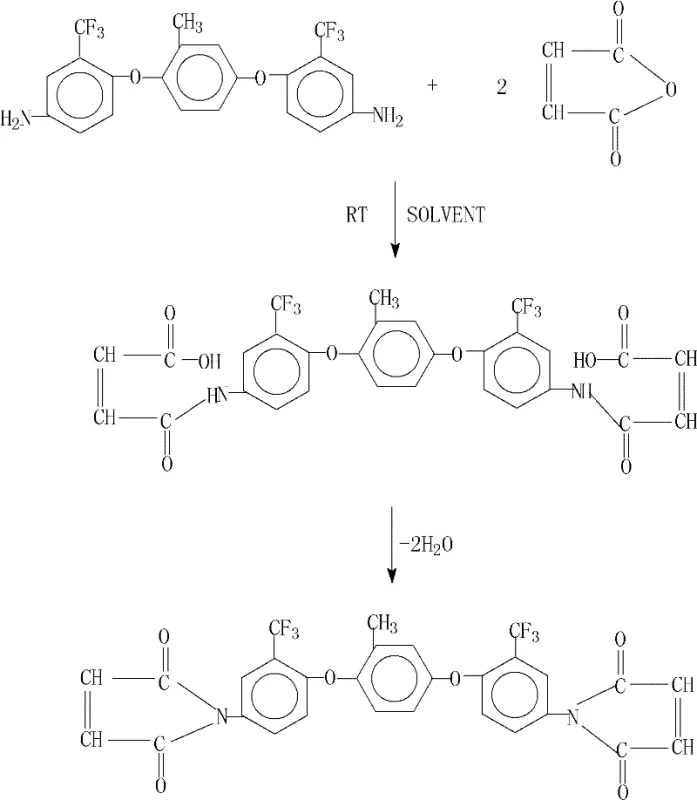
The methodology disclosed in patent CN101941933B introduces a sophisticated yet operationally simple strategy that utilizes a synergistic combination of anhydrous calcium chloride and N,N-dimethylaniline to catalyze the cyclization process with remarkable efficiency. By conducting the initial amidation at room temperature followed by a controlled azeotropic dehydration step, this novel approach ensures that the bismaleamic acid intermediate is formed cleanly before being rapidly converted to the target bismaleimide. The inclusion of solid anhydrous calcium chloride serves as a powerful dehydrating agent that actively sequesters water, shifting the reaction equilibrium decisively towards product formation without the need for harsh conditions. Comparative data within the patent highlights that this specific catalytic system can elevate yields from roughly two-thirds to nearly quantitative levels, demonstrating a profound improvement in process efficiency that translates directly to reduced waste and lower manufacturing costs for high-value specialty chemicals.
Mechanistic Insights into Catalytic Azeotropic Dehydration
The core of this synthetic innovation lies in the precise management of the dehydration equilibrium during the conversion of the bismaleamic acid intermediate into the cyclic imide structure. The reaction initiates with the nucleophilic attack of the amine groups on the maleic anhydride carbonyls at room temperature, forming the soluble bismaleamic acid species in a polar aprotic solvent medium such as N,N-dimethylacetamide or N-methyl-2-pyrrolidone. Upon the addition of the dehydrating system, the reaction mixture is heated to reflux, where the azeotropic agent, typically toluene or xylene, forms a low-boiling mixture with the water produced by the cyclization. The presence of N,N-dimethylaniline likely acts as a basic catalyst that facilitates the proton transfer steps required for ring closure, while the anhydrous calcium chloride physically binds the liberated water molecules, preventing the reverse hydrolysis reaction. This dual-action mechanism ensures that the cyclization proceeds rapidly and completely, preserving the integrity of the polymerizable double bonds which are essential for the subsequent curing of the resin.
Impurity control in this process is inherently managed by the mildness of the initial amidation step and the selectivity of the dehydration catalyst. By maintaining the first step at room temperature, the formation of side products associated with thermal stress is effectively eliminated, ensuring a clean starting profile for the cyclization phase. The use of ethanol for precipitation in the final workup further enhances purity, as the target bismaleimide product exhibits low solubility in ethanol while many potential organic impurities and residual solvents remain in the mother liquor. This crystallization-induced purification allows for the isolation of the product as a high-purity powder with a sharp melting point, as evidenced by differential scanning calorimetry data showing a distinct endotherm at approximately 213°C. Such high purity is critical for R&D directors who require consistent rheological and curing behaviors in their formulation development for advanced composite matrices.
How to Synthesize 2-methyl-1,4-bis(2-trifluoromethyl-4-maleimidophenoxy)benzene Efficiently
The synthesis protocol outlined in the patent provides a robust framework for laboratory and pilot-scale production, emphasizing the importance of stoichiometric precision and temperature control to maximize yield. The process begins by dissolving the trifluoromethyl-substituted diamine in a suitable polar solvent, followed by the controlled addition of maleic anhydride to manage the exotherm and ensure complete conversion to the amic acid.
- React 2-methyl-1,4-bis(2-trifluoromethyl-4-aminophenoxy)benzene with maleic anhydride in a polar aprotic solvent at room temperature to form the amic acid intermediate.
- Add anhydrous calcium chloride, an azeotropic dehydrating agent, and N,N-dimethylaniline, then heat to reflux to effect cyclodehydration and water removal.
- Distill off the dehydrating agent, cool the mixture, and precipitate the final bismaleimide product using ethanol followed by filtration and drying.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this patented synthesis route offers substantial strategic benefits centered around cost efficiency, operational safety, and environmental compliance. The ability to conduct the reaction under atmospheric pressure eliminates the need for expensive high-pressure reactors and the associated safety infrastructure, thereby reducing capital expenditure and maintenance overheads for manufacturing facilities. Additionally, the process design incorporates solvent recovery loops where the azeotropic dehydrating agents and mother liquors can be distilled and reused, significantly lowering the consumption of raw materials and minimizing the volume of hazardous waste requiring disposal. This closed-loop approach not only aligns with increasingly stringent environmental regulations but also insulates the supply chain from volatility in solvent pricing, ensuring more predictable production costs over the long term.
- Cost Reduction in Manufacturing: The dramatic improvement in reaction yield driven by the calcium chloride catalytic system means that less raw material is wasted per unit of finished product, directly lowering the cost of goods sold. By avoiding the need for complex purification steps or multiple recrystallizations, the process reduces labor hours and energy consumption associated with downstream processing. The elimination of corrosive reagents further extends the lifespan of reactor vessels and piping, reducing replacement costs and downtime for maintenance. These cumulative efficiencies create a leaner manufacturing profile that allows for competitive pricing in the high-performance polymer market without sacrificing margin.
- Enhanced Supply Chain Reliability: The starting materials, including the specific trifluoromethyl diamine and maleic anhydride, are commercially available from established chemical suppliers, ensuring a stable and secure feedstock supply. The simplicity of the one-pot reaction sequence reduces the risk of batch failures due to operational complexity, leading to more consistent output and reliable delivery schedules for customers. Furthermore, the robustness of the process allows for flexibility in scaling production volumes to meet fluctuating demand in the aerospace and electronics sectors without requiring significant process re-engineering. This reliability is crucial for maintaining continuity in the supply of critical materials for defense and commercial aviation programs.
- Scalability and Environmental Compliance: The use of standard organic solvents and atmospheric conditions makes this process highly scalable from kilogram to multi-ton production scales using conventional chemical processing equipment. The recovery and recycling of solvents significantly reduce the environmental footprint of the manufacturing process, aiding compliance with green chemistry initiatives and reducing waste disposal fees. The absence of heavy metal catalysts or toxic reagents simplifies the handling of effluent streams and reduces the regulatory burden associated with hazardous material management. This environmentally friendly profile enhances the brand value of the final product and facilitates easier approval for use in sensitive applications where material toxicity is a concern.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this specialized bismaleimide resin, based on the detailed disclosures within the patent literature.
Q: What is the key advantage of using calcium chloride in this bismaleimide synthesis?
A: According to patent CN101941933B, the addition of solid anhydrous calcium chloride acts as a crucial dehydrating aid that significantly drives the equilibrium towards the imide formation, resulting in drastically higher yields compared to traditional thermal dehydration methods.
Q: Can the solvents used in this process be recycled?
A: Yes, the process is designed for industrial sustainability; the azeotropic dehydrating agent (such as toluene or xylene) is distilled off and can be recovered for reuse, while the mother liquor from the ethanol precipitation can also be fractionated and purified for recycling.
Q: What thermal properties does this specific bismaleimide resin exhibit?
A: The synthesized resin demonstrates a sharp melting point around 213°C and excellent thermal stability, with exothermic curing behavior initiating near 324°C, making it highly suitable for high-temperature structural adhesives and composite matrices.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-methyl-1,4-bis(2-trifluoromethyl-4-maleimidophenoxy)benzene Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-performance intermediates play in the development of next-generation aerospace and electronic materials, and we are uniquely positioned to support your supply needs with expertise and precision. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements whether you are in the R&D phase or full-scale manufacturing. We operate stringent purity specifications and maintain rigorous QC labs to guarantee that every batch of 2-methyl-1,4-bis(2-trifluoromethyl-4-maleimidophenoxy)benzene meets the exacting standards required for advanced composite applications. Our commitment to quality assurance ensures that the thermal and mechanical properties of your final cured resins remain consistent and reliable.
We invite you to contact our technical procurement team to discuss how we can tailor our production capabilities to your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our optimized synthesis routes can reduce your overall material costs. We encourage you to reach out today to obtain specific COA data and route feasibility assessments that will demonstrate our capability to be your trusted partner in the global supply of high-performance polyimide precursors.
