Scaling High-Performance Fluorinated Bismaleimide Resins for Aerospace and Electronic Applications
Introduction to Next-Generation Bismaleimide Technology
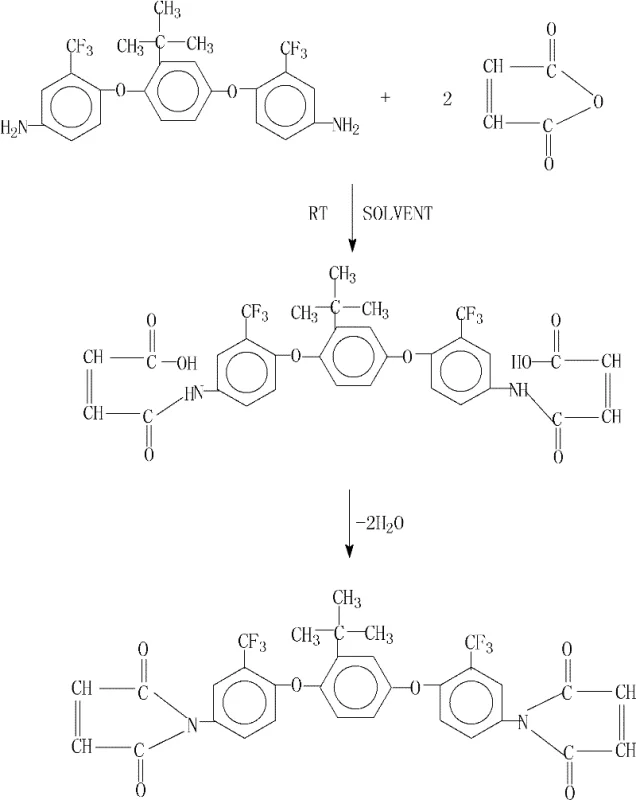
The demand for advanced composite materials in the aerospace and microelectronics sectors has driven an urgent need for resin systems that offer superior thermal stability and mechanical performance without compromising processability. Patent CN101921221B introduces a breakthrough in the synthesis of 2-tert-butyl-1,4-bis(2-trifluoromethyl-4-maleimidophenoxy)benzene, a specialized fluorinated bismaleimide monomer designed to meet these rigorous specifications. Unlike conventional synthetic routes that often suffer from moderate yields and complex purification requirements, this disclosed methodology leverages a unique catalytic dehydration system to achieve near-quantitative conversion rates. For R&D directors and procurement specialists seeking a reliable bismaleimide resin supplier, understanding the nuances of this patented pathway is critical for securing a supply chain capable of delivering high-purity intermediates at a competitive cost structure.
This technical insight report dissects the proprietary two-step synthesis described in the patent, highlighting how the strategic integration of anhydrous calcium chloride and N,N-dimethylaniline transforms the cyclization efficiency. By operating under atmospheric pressure and utilizing recoverable solvent systems, the process addresses key pain points in industrial scale-up, including waste generation and energy consumption. As we transition towards more sustainable manufacturing paradigms, the ability to produce high-performance polyimide precursors with minimal environmental footprint becomes a decisive factor in vendor selection. The following analysis provides a deep dive into the mechanistic advantages and commercial implications of adopting this refined synthetic protocol for the production of advanced thermosetting materials.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis of bismaleimide monomers typically relies on the reaction of diamines with maleic anhydride followed by dehydration cyclization using sodium acetate and acetic anhydride. While historically established, this classical approach presents significant drawbacks when applied to complex, fluorinated substrates. The use of acetic anhydride often generates substantial amounts of acetic acid waste, necessitating expensive neutralization and disposal procedures that inflate the overall cost of goods sold. Furthermore, conventional cyclization conditions frequently result in incomplete conversion, leading to crude products contaminated with amic acid intermediates and polymeric by-products. These impurities are notoriously difficult to remove, often requiring multiple recrystallization steps that further erode the final yield and extend the production cycle time. For supply chain managers, these inefficiencies translate into unpredictable lead times and higher inventory carrying costs, making traditional suppliers less attractive for long-term contracts requiring consistent volume.
The Novel Approach
In stark contrast, the method disclosed in CN101921221B utilizes a sophisticated catalytic system comprising anhydrous calcium chloride and N,N-dimethylaniline to drive the dehydration cyclization. This novel approach eliminates the need for corrosive acetic anhydride, thereby reducing equipment maintenance costs and enhancing operator safety. The patent data demonstrates a dramatic improvement in process efficiency, with yields soaring from a mediocre 46.9% in comparative examples lacking the catalyst to an exceptional 99.7% in the optimized process. This leap in efficiency is not merely a laboratory curiosity; it represents a fundamental shift in manufacturing economics, allowing for the production of high-purity OLED material or aerospace intermediates with minimal raw material loss. By conducting the reaction in a polar aprotic solvent followed by azeotropic dehydration with toluene or xylene, the process ensures a homogeneous reaction environment that facilitates rapid kinetics and superior product quality.
Mechanistic Insights into Catalytic Dehydration Cyclization
The core innovation of this synthesis lies in the synergistic interaction between the Lewis acid character of calcium chloride and the basic properties of N,N-dimethylaniline during the cyclization step. Initially, the diamine reacts with maleic anhydride at room temperature to form the bis-maleamic acid intermediate, a reversible exothermic reaction that proceeds rapidly in solvents like N,N-dimethylacetamide or N-methyl-2-pyrrolidone. The subsequent cyclization to form the imide ring is the rate-determining step, traditionally hindered by the stability of the amic acid bond. In this patented system, calcium chloride acts as a dehydrating agent that coordinates with the carbonyl oxygen, activating the carboxylic acid group for nucleophilic attack by the amide nitrogen. Simultaneously, N,N-dimethylaniline serves as a proton scavenger, neutralizing the water molecule released during ring closure and shifting the equilibrium decisively towards the imide product. This dual-activation mechanism lowers the activation energy required for cyclization, allowing the reaction to proceed smoothly at reflux temperatures between 100°C and 160°C.
From an impurity control perspective, this mechanism offers distinct advantages over acid-catalyzed methods. The mild basicity of the amine catalyst prevents the polymerization of the maleimide double bonds, a common side reaction that can occur under strongly acidic or high-thermal stress conditions. The patent specifies that the resulting product exhibits a sharp melting point at 212.2°C and a clean exothermic peak at 304.2°C, indicative of a highly crystalline and chemically pure substance. For R&D teams focused on the commercial scale-up of complex polymer additives, this level of purity is essential to ensure consistent curing behavior in the final composite application. The absence of residual amic acid or oligomeric species guarantees that the thermal and mechanical properties of the cured resin matrix remain within tight specification limits, reducing the risk of batch-to-batch variability in downstream manufacturing processes.
How to Synthesize 2-tert-butyl-1,4-bis(2-trifluoromethyl-4-maleimidophenoxy)benzene Efficiently
Implementing this synthesis route requires precise control over stoichiometry and temperature profiles to maximize the benefits of the catalytic system. The process begins with the dissolution of the fluorinated diamine in a polar aprotic solvent, followed by the controlled addition of maleic anhydride to manage the exotherm. Once the amic acid intermediate is formed, the introduction of the solid calcium chloride and the liquid amine catalyst must be timed correctly to initiate the dehydration phase without causing premature precipitation. The detailed standardized synthesis steps below outline the critical parameters for achieving the reported 99%+ yields, ensuring that technical teams can replicate the patent's success in a pilot or production setting. Adhering to these guidelines is crucial for maintaining the structural integrity of the trifluoromethyl groups and preventing thermal degradation of the sensitive maleimide functionality.
- React 2-tert-butyl-1,4-bis(2-trifluoromethyl-4-aminophenoxy)benzene with maleic anhydride (1: 2.0-2.1 molar ratio) in a polar aprotic solvent at room temperature for 1 hour to form the amic acid intermediate.
- Add anhydrous calcium chloride powder, an azeotropic dehydrating agent (e.g., toluene), and N,N-dimethylaniline to the solution.
- Heat the mixture to reflux (100-160°C) for 2-3 hours to effect cyclization, then distill off the solvent and precipitate the product with ethanol.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis route offers compelling economic and operational benefits that extend beyond simple yield improvements. The elimination of corrosive reagents like acetic anhydride reduces the burden on waste treatment facilities and lowers the total cost of ownership for reaction vessels and piping. Moreover, the high selectivity of the reaction minimizes the formation of by-products, which simplifies the downstream purification process and reduces the consumption of auxiliary solvents and energy. These factors collectively contribute to a more robust and resilient supply chain, capable of meeting the demanding delivery schedules of the aerospace and electronics industries. By partnering with a manufacturer who utilizes this advanced technology, buyers can secure a stable source of high-purity intermediates that support their own product quality goals.
- Cost Reduction in Manufacturing: The most significant financial advantage stems from the drastic increase in reaction yield, which effectively doubles the output per unit of raw material compared to older methods. This near-quantitative conversion means that expensive fluorinated starting materials are utilized with maximum efficiency, directly lowering the variable cost per kilogram of the final product. Additionally, the patent highlights the recovery and recycling of the azeotropic dehydrating agent and the mother liquor, which further diminishes solvent procurement costs and waste disposal fees. Over the lifecycle of a large-scale production campaign, these cumulative savings can be substantial, allowing for more competitive pricing strategies without sacrificing margin.
- Enhanced Supply Chain Reliability: The simplicity of the operational protocol, which avoids high-pressure equipment and hazardous reagents, reduces the likelihood of unplanned shutdowns or safety incidents that could disrupt supply. The use of readily available catalysts and solvents ensures that the manufacturing process is not vulnerable to shortages of niche chemicals. Furthermore, the ability to complete the entire synthesis in a single reactor vessel streamlines the production workflow, shortening the cycle time from raw material intake to finished goods. This operational agility enables suppliers to respond more quickly to fluctuations in market demand, providing customers with greater confidence in continuity of supply for their critical composite material programs.
- Scalability and Environmental Compliance: As regulatory pressures regarding industrial emissions and waste generation intensify, this environmentally friendly process positions manufacturers favorably for future compliance. The absence of corrosive waste streams and the implementation of solvent recycling loops align with green chemistry principles, reducing the environmental footprint of the manufacturing site. This sustainability profile is increasingly important for multinational corporations seeking to minimize Scope 3 emissions in their supply chains. The process is inherently scalable, having been designed with industrial production in mind, ensuring that quality and efficiency are maintained as volumes increase from pilot batches to multi-ton annual production capacities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this specialized bismaleimide resin. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a transparent view of the technology's capabilities. Understanding these details helps stakeholders make informed decisions about integrating this material into their existing formulations or supply networks. We encourage technical teams to review these points carefully to assess the fit for their specific application requirements.
Q: What is the key advantage of the CaCl2/N,N-Dimethylaniline catalytic system?
A: This specific catalytic combination drastically improves the cyclization yield from approximately 47% in traditional methods to over 99%, minimizing raw material waste and purification costs.
Q: Is this bismaleimide resin suitable for high-temperature applications?
A: Yes, the presence of trifluoromethyl groups and the rigid imide structure provides excellent thermal stability, with a melting point around 212°C and decomposition occurring well above 300°C.
Q: Can the solvents used in this process be recycled?
A: Yes, the patent explicitly describes the recovery and recycling of the azeotropic dehydrating agent and the mother liquor, which significantly enhances the environmental profile and cost-efficiency of the process.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-tert-butyl-1,4-bis(2-trifluoromethyl-4-maleimidophenoxy)benzene Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to industrial reality requires a partner with deep technical expertise and robust manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the high yields and purity demonstrated in patent literature are faithfully reproduced at scale. We operate stringent purity specifications and maintain rigorous QC labs equipped to verify the structural integrity and thermal properties of every batch, guaranteeing that the material meets the exacting standards required for aerospace and electronic applications. Our commitment to quality assurance means that you receive a product that performs consistently, batch after batch, supporting the reliability of your final composite structures.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can optimize your supply chain and reduce your overall material costs. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits specific to your volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate our capability to serve as your long-term strategic partner. Let us help you leverage this cutting-edge technology to drive innovation and efficiency in your next-generation material development projects.
