Scalable Synthesis of 4-Amino-2-Butenoyl Chloride for Advanced Kinase Inhibitor Manufacturing
Introduction to Patent CN1761644A and Kinase Inhibitor Intermediates
The pharmaceutical landscape for oncology treatments continues to evolve rapidly, driven by the demand for highly specific protein kinase inhibitors. Patent CN1761644A presents a significant technological advancement in the synthesis of 3-cyanoquinoline derivatives, which serve as critical intermediates in the production of these potent therapeutic agents. Specifically, the patent discloses novel methods for preparing 4-amino-2-butenoyl chloride compounds and their subsequent application in forming amide bonds with complex quinoline scaffolds. This technology addresses the longstanding challenges associated with scaling up the production of protein-tyrosine kinase inhibitors, which are vital for treating conditions such as cancer, restenosis, and angiogenesis. By providing a robust pathway that transitions from laboratory concepts to pilot workshop feasibility, this intellectual property offers a reliable foundation for pharmaceutical intermediate suppliers aiming to support global drug development pipelines. The strategic importance of this synthesis route lies in its ability to maintain high chemical integrity while managing the reactivity of sensitive acid chloride intermediates.
For R&D directors and procurement specialists, understanding the nuances of this patent is essential for securing a stable supply of high-quality active pharmaceutical ingredient (API) precursors. The described methodology moves beyond theoretical chemistry, offering concrete operational parameters that ensure reproducibility and safety on an industrial scale. The core innovation involves the controlled generation of 4-(dimethylamino)-2-butenoyl chloride and its immediate utilization in coupling reactions without extensive isolation steps that could lead to degradation. This approach not only streamlines the manufacturing workflow but also significantly mitigates the risks associated with handling unstable intermediates. As the industry seeks more efficient routes to complex heterocyclic compounds, the processes outlined in CN1761644A stand out as a benchmark for modern intermediate synthesis, promising enhanced yield consistency and superior impurity profiles compared to legacy methods.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3-cyanoquinoline derivatives, such as those described in earlier patents like WO9843960, has been largely confined to laboratory-scale operations. These conventional methods often suffer from significant scalability issues, primarily due to the instability of the acylating agents and the difficulty in controlling exothermic reactions during the coupling phase. Traditional protocols frequently rely on harsh conditions or inefficient purification techniques that result in variable yields and inconsistent purity levels, making them unsuitable for the rigorous demands of commercial API manufacturing. Furthermore, the isolation of intermediate acid chlorides in prior art often leads to decomposition or polymerization, creating bottlenecks that increase production costs and extend lead times. The lack of defined crystallization parameters in older methods means that removing trace impurities, which can be toxic or affect drug efficacy, becomes a costly and iterative process. Consequently, manufacturers relying on these legacy routes face substantial supply chain vulnerabilities and quality control challenges.
The Novel Approach
In stark contrast, the novel approach detailed in CN1761644A introduces a meticulously engineered process designed specifically for large-scale preparation. This method utilizes a suspension-based technique where the acid chloride is generated in situ or handled as a stable suspension in solvents like acetonitrile, thereby minimizing exposure to degrading conditions. A key differentiator is the precise thermal management strategy, which involves cooling the reaction mixture to 0-10°C during reagent addition and carefully warming it to 25-30°C to drive completion before cooling again for coupling. This thermal cycling ensures that the chlorinating agent, typically oxalyl chloride, is fully consumed, preventing downstream contamination. Moreover, the patent outlines a sophisticated workup procedure involving the dropwise addition of the aniline component until its concentration drops below 5%, followed by precipitation with aqueous base. This controlled addition prevents localized high concentrations that could lead to bis-acylation or other side reactions, ensuring a cleaner reaction profile and facilitating easier downstream processing.
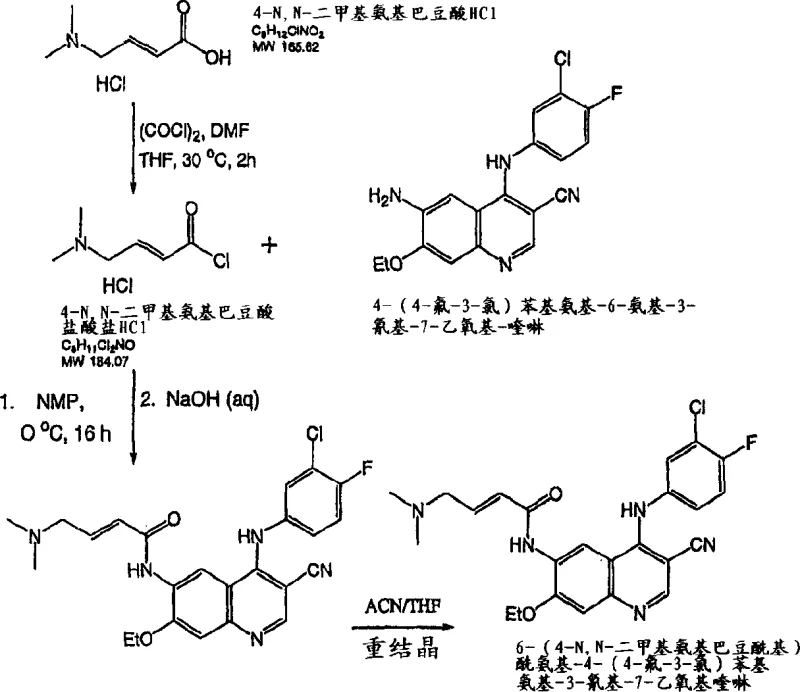
Mechanistic Insights into Acylation and Crystallization Control
The chemical mechanism underpinning this synthesis relies on the nucleophilic attack of the aniline nitrogen on the carbonyl carbon of the activated acid chloride. However, the true sophistication of this patent lies in the kinetic control of this reaction. By maintaining the reaction temperature between 0°C and 10°C during the addition of the acid chloride to the aniline solution in N-methylpyrrolidone (NMP), the process suppresses competing hydrolysis reactions and minimizes thermal stress on the sensitive quinoline ring system. The use of catalytic amounts of dimethylformamide (DMF) during the formation of the acid chloride enhances the electrophilicity of the carbonyl group via the Vilsmeier-Haack intermediate, ensuring rapid and complete conversion of the carboxylic acid precursor. This mechanistic efficiency is crucial for reducing the residence time of reactive species, thereby limiting the formation of oligomeric impurities that are difficult to remove later. The subsequent quenching with aqueous sodium bicarbonate or sodium hydroxide serves a dual purpose: neutralizing excess acid and inducing precipitation of the product in a form that is amenable to filtration.
Impurity control is further enhanced through a rigorous recrystallization protocol that leverages the solubility differences between the target molecule and its byproducts. The patent specifies a solvent system comprising tetrahydrofuran (THF) and acetonitrile (CH3CN), which is heated to reflux to dissolve the crude solid and then slowly cooled to induce crystallization. This thermal gradient allows for the selective exclusion of impurities from the crystal lattice, resulting in a product with purity exceeding 98% after a second recrystallization step. The mechanism of impurity rejection here is thermodynamic; as the solution cools, the saturation point of the desired compound is reached first, causing it to precipitate while impurities remain in the mother liquor due to their higher solubility or lower concentration. This level of purification is critical for pharmaceutical intermediates, where strict regulatory limits on genotoxic impurities and heavy metals must be met. The ability to achieve such high purity without chromatography makes this process economically viable for ton-scale production.
How to Synthesize 4-Amino-2-Butenoyl Chloride Efficiently
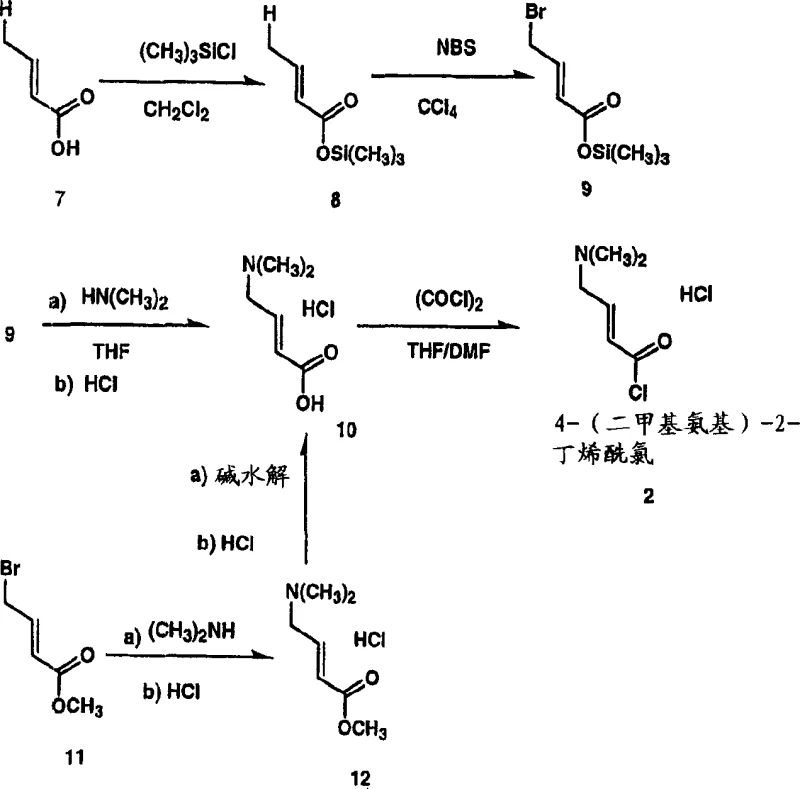
The synthesis of the key intermediate, 4-(dimethylamino)-2-butenoyl chloride hydrochloride, is a pivotal step that determines the success of the entire manufacturing campaign. The patent provides a detailed roadmap for converting 4-dimethylaminocrotonic acid into its corresponding acid chloride using oxalyl chloride in a polar aprotic solvent. This transformation must be executed with precision to avoid the formation of side products that could complicate the subsequent coupling reaction. The process begins with the suspension of the acid salt in acetonitrile, followed by the controlled addition of the chlorinating agent at low temperatures. Operators must monitor the reaction progress closely, typically using HPLC, to confirm the complete consumption of oxalyl chloride before proceeding. Once the acid chloride is formed, it is not isolated as a dry solid but rather used directly as a suspension or solution to maintain stability. This "telescoped" approach reduces handling risks and improves overall process efficiency. For a comprehensive guide on the specific operational parameters, stoichiometry, and safety precautions required for this synthesis, please refer to the standardized protocol below.
- Preparation of Acid Chloride: React 4-dimethylaminocrotonic acid hydrochloride with oxalyl chloride in acetonitrile at 0-10°C, warming to 25-30°C until consumption is complete.
- Coupling Reaction: Add the acid chloride suspension to a cooled solution of the quinoline aniline derivative in NMP, maintaining temperature below 10°C to control exotherm.
- Purification: Precipitate the product using aqueous base, followed by rigorous recrystallization from acetonitrile/THF mixtures to achieve >98% purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of the technology described in CN1761644A offers substantial strategic advantages over traditional sourcing models. The primary benefit lies in the inherent scalability of the process, which has been validated for pilot workshop and large-scale manufacturing environments. Unlike laboratory curiosities that fail upon scale-up, this method utilizes common, commercially available solvents such as acetonitrile, THF, and NMP, which simplifies logistics and reduces raw material costs. The robust nature of the crystallization steps ensures consistent batch-to-batch quality, reducing the risk of production delays caused by out-of-specification results. Furthermore, the elimination of complex purification techniques like column chromatography in favor of crystallization significantly lowers the cost of goods sold (COGS). This cost reduction in pharmaceutical intermediate manufacturing is achieved not by cutting corners, but by optimizing the physical chemistry of the process to maximize yield and minimize waste. The streamlined workflow also shortens the overall production cycle time, allowing suppliers to respond more agilely to fluctuating market demands.
- Cost Reduction in Manufacturing: The process achieves significant cost optimization by utilizing inexpensive chlorinating agents like oxalyl chloride and avoiding the need for expensive transition metal catalysts or specialized reagents. The high recovery rates reported, often exceeding 80% even after multiple purification steps, mean that less raw material is required to produce the same amount of final product. Additionally, the ability to recycle mother liquors from the crystallization steps further enhances material efficiency. By designing a process that relies on standard unit operations like filtration and distillation, capital expenditure for new equipment is minimized, allowing existing facilities to be repurposed for this production. These factors combine to create a highly competitive cost structure that can be passed down the supply chain.
- Enhanced Supply Chain Reliability: Supply continuity is bolstered by the use of stable intermediates and robust reaction conditions that are less sensitive to minor variations in operating parameters. The method's tolerance for standard industrial grade solvents reduces dependency on high-purity reagents that might have long lead times. Moreover, the clear definition of critical process parameters (CPPs), such as temperature ranges and addition rates, ensures that the process can be reliably transferred between different manufacturing sites without loss of quality. This transferability is crucial for building a resilient supply chain that can withstand regional disruptions. The high purity of the final intermediate also reduces the burden on downstream API manufacturers, who can proceed with confidence knowing that the input material meets stringent quality standards.
- Scalability and Environmental Compliance: The environmental footprint of this synthesis is managed through the use of aqueous workups and the potential for solvent recovery. The precipitation step using aqueous base generates a solid product that is easily filtered, reducing the volume of liquid waste compared to extraction-heavy processes. The solvents used, while requiring proper handling, are well-understood in terms of waste treatment and recycling. The process avoids the generation of heavy metal waste, which is a significant regulatory hurdle in many jurisdictions. By adhering to green chemistry principles where possible, such as atom economy in the coupling step, the process aligns with increasingly strict environmental regulations. This compliance ensures long-term viability and reduces the risk of regulatory shutdowns or fines.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and supply of 4-amino-2-butenoyl chloride derivatives. These answers are derived directly from the experimental data and claims within patent CN1761644A, providing a factual basis for decision-making. Understanding these details helps stakeholders assess the feasibility of integrating this intermediate into their own production workflows. The clarity of the patent disclosure allows for a high degree of confidence in the reproducibility of the results. For more specific technical data or custom synthesis requests, direct engagement with the technical team is recommended.
Q: What are the critical temperature controls for the acylation step?
A: The process requires strict temperature management, initially cooling the suspension to 0-10°C during chlorinating agent addition to prevent side reactions, then warming to 25-30°C to ensure complete consumption of the reagent before cooling again for the coupling phase.
Q: How is high purity (>98%) achieved in the final product?
A: High purity is secured through a multi-stage recrystallization protocol using tetrahydrofuran (THF) and acetonitrile (CH3CN) solvent systems, which effectively remove organic impurities and residual starting materials.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the patent explicitly describes methods suitable for pilot workshop and large-scale manufacture, utilizing common industrial solvents like acetonitrile and NMP with standard filtration and drying equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Amino-2-Butenoyl Chloride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of life-saving oncology therapies. Our expertise in process chemistry allows us to replicate and optimize the advanced synthesis routes described in patents like CN1761644A, ensuring that our clients receive materials that meet the highest standards of purity and consistency. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, leveraging our state-of-the-art facilities to deliver ton-scale quantities reliably. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that utilize advanced analytical techniques to verify every batch. Whether you require custom synthesis of complex quinoline derivatives or bulk supply of key acylating agents, our team is equipped to handle your most challenging requirements with precision and speed.
We invite you to collaborate with us to explore how our manufacturing capabilities can enhance your supply chain efficiency and reduce your overall project costs. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume needs and quality targets. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments for your upcoming projects. Let us be your trusted partner in navigating the complexities of pharmaceutical intermediate manufacturing, delivering value through science and operational excellence.
