Advanced Hydroformylation Technology for Scalable Production of High-Purity Muguet Aldehyde Intermediates
The global fine fragrance industry continuously demands novel and high-quality olfactory ingredients to differentiate luxury products, with muguet or lily-of-the-valley notes remaining a cornerstone of modern perfumery. Patent CN116018334A introduces a groundbreaking synthetic methodology for producing 3-(cyclohex-1-en-1-yl)propanal derivatives, which are pivotal intermediates for recreating these delicate floral scents. Unlike traditional extraction methods that fail to capture the full spectrum of the flower's aroma, this chemical synthesis allows for the precise construction of the molecular architecture required for the desired aldehyde facet. The invention specifically addresses the long-standing challenges in this field by providing a route that starts from novel vinyl cyclohexyl intermediates, enabling manufacturers to achieve unprecedented levels of purity and yield. This technological leap is particularly significant for R&D teams seeking to stabilize their supply chains against the volatility of natural raw materials while maintaining the highest sensory standards.
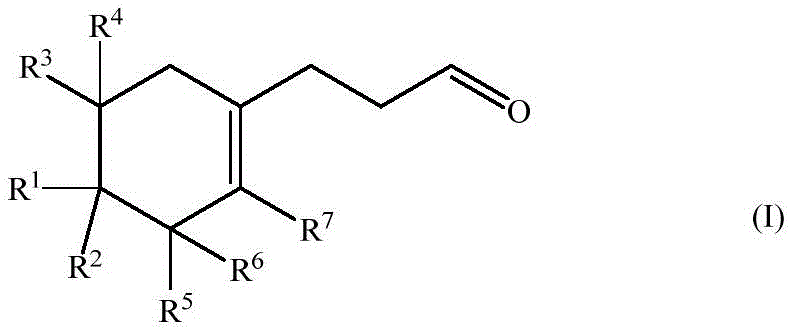
Historically, the synthesis of muguet aldehydes has been plagued by inefficient methodologies that rely on hazardous reagents and offer poor selectivity. Conventional approaches often necessitate the use of Grignard reagents, free radical chemistry, or the thermal decomposition of dienals, all of which suffer from low overall yields and complex impurity profiles that are difficult to remove. These legacy processes not only increase the cost of goods sold due to material loss but also introduce significant safety risks associated with handling reactive organometallic species on a large scale. Furthermore, the inability to effectively control the position of the double bond often results in mixtures of isomers that dilute the olfactory potency of the final product. In stark contrast, the novel approach detailed in the patent utilizes a streamlined sequence involving hydroformylation and elimination, which drastically simplifies the synthetic pathway. By leveraging transition metal catalysis, this new method bypasses the need for stoichiometric hazardous reagents, thereby enhancing both the economic viability and the environmental sustainability of the manufacturing process.
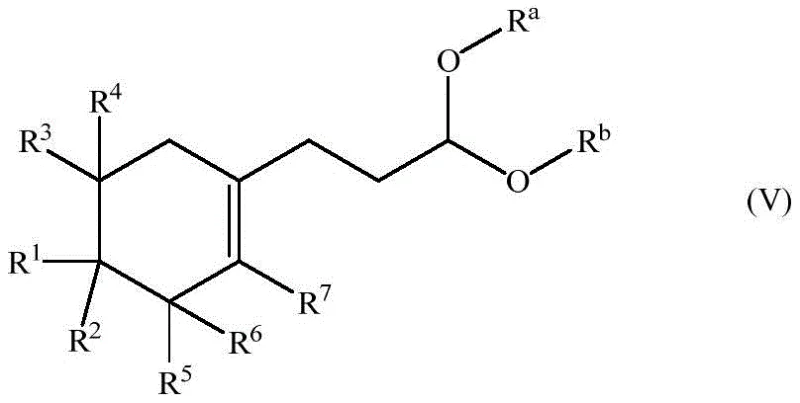
The core of this innovative process lies in the rhodium-catalyzed hydroformylation of specific vinyl cyclohexyl precursors, a reaction that requires precise control over regioselectivity to ensure the formation of the linear aldehyde rather than the branched isomer. The patent highlights the critical role of bulky bidentate phosphorus ligands, such as BIPHEPHOS or Xantphos derivatives, in directing the rhodium catalyst to favor the linear addition of the formyl group to the terminal carbon of the vinyl chain. This selectivity is paramount because the linear aldehyde is the direct precursor to the desired muguet scent, whereas branched isomers contribute little to no olfactory value and act as impurities. Following the hydroformylation, the process incorporates a sophisticated protection strategy where the sensitive aldehyde group is converted into an acetal. This protection step is essential to shield the carbonyl functionality during the subsequent acidic elimination reaction, preventing polymerization or degradation. The final transformation involves an acid-catalyzed elimination and isomerization that not only removes the protecting group but also drives the migration of the double bond from the exocyclic position into the endocyclic position of the cyclohexene ring.
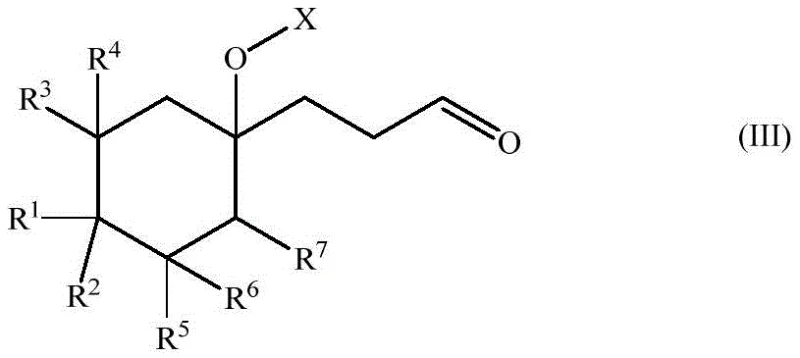
Understanding the mechanistic nuances of the elimination and isomerization steps is crucial for optimizing the final product quality and ensuring the correct sensory profile. The conversion of the protected intermediate into the final unsaturated aldehyde involves a delicate balance of thermodynamic and kinetic factors controlled by the choice of acid catalyst and reaction temperature. The patent discloses that strong Bronsted or Lewis acids can facilitate the elimination of the leaving group, initially forming an exo-isomer where the double bond resides in the alkyl side chain. However, under the reaction conditions, this exo-isomer rapidly isomerizes to the more thermodynamically stable endo-isomer, where the double bond is conjugated within the cyclohexene ring system. This isomerization is vital because the endo-configuration is responsible for the characteristic fresh, floral muguet note, while the exo-form possesses a different and often less desirable odor. The ability to drive this equilibrium almost completely towards the endo-product demonstrates the robustness of the chemical design, ensuring that the final fragrance ingredient meets the rigorous specifications required by top-tier perfumers without extensive chromatographic purification.
How to Synthesize 3-(Cyclohex-1-en-1-yl)propanal Efficiently
The synthesis of these high-value fragrance intermediates follows a logical progression designed to maximize yield while minimizing waste generation. The process begins with the preparation of the vinyl cyclohexyl starting material, which can be derived from readily available cyclic ketones via ethynylation and selective hydrogenation. Once the vinyl precursor is secured, it undergoes the key hydroformylation step in a pressurized reactor using syngas and a specialized rhodium catalyst system. The reaction conditions are carefully tuned, typically operating at moderate temperatures and pressures to maintain catalyst stability and activity. Following the formation of the linear aldehyde, the crude mixture is subjected to acetalization using a diol and an acid catalyst, often in a Dean-Stark apparatus to remove water and drive the equilibrium forward. The resulting protected intermediate is then treated with an acid to effect elimination and isomerization, finally yielding the target aldehyde after a mild deprotection step.
- Perform rhodium-catalyzed hydroformylation on a formula (II) vinyl cyclohexyl derivative using syngas and a bidentate phosphorus ligand like BIPHEPHOS to generate the linear aldehyde intermediate.
- Protect the resulting aldehyde group as an acetal (formula IV) using a diol and acid catalyst to prevent side reactions during the subsequent elimination step.
- Execute an acid-catalyzed elimination and isomerization reaction to remove the protecting group and shift the double bond into the ring, yielding the final formula (I) endo-isomer.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this hydroformylation-based technology represents a strategic opportunity to optimize costs and secure long-term supply continuity. The shift away from Grignard reagents and radical chemistries eliminates the need for expensive, moisture-sensitive raw materials that require specialized storage and handling infrastructure. Instead, the process relies on commodity chemicals like syngas, acetic anhydride, and standard diols, which are widely available in the global market and less susceptible to price volatility. This transition to more robust feedstocks significantly reduces the risk of production stoppages caused by raw material shortages, thereby enhancing the reliability of the supply chain. Furthermore, the improved selectivity of the rhodium-catalyzed step means that less starting material is wasted on unwanted byproducts, directly translating to a lower cost per kilogram of the final active ingredient. The reduction in waste generation also aligns with increasingly stringent environmental regulations, potentially lowering disposal costs and improving the overall sustainability profile of the manufacturing site.
- Cost Reduction in Manufacturing: The implementation of this catalytic route offers substantial economic benefits by improving atom economy and reducing the number of purification steps required. Traditional methods often generate significant amounts of salt waste and require energy-intensive distillations to separate close-boiling isomers, whereas the high selectivity of the new process minimizes these downstream burdens. By utilizing highly efficient rhodium catalysts that can operate at low loadings, the consumption of precious metals is optimized, further driving down the variable costs of production. Additionally, the ability to perform multiple transformations, such as elimination and isomerization, in a single pot or sequential telescoped manner reduces solvent usage and reactor occupancy time. These efficiencies collectively contribute to a leaner manufacturing operation that can offer competitive pricing without compromising on the quality of the fragrance ingredient.
- Enhanced Supply Chain Reliability: Securing a stable supply of critical fragrance intermediates is paramount for maintaining brand consistency in the luxury goods sector. This new synthetic route enhances supply chain resilience by diversifying the source of raw materials away from niche suppliers of hazardous reagents towards bulk chemical providers. The robustness of the catalytic system allows for flexible production scheduling, as the reaction is less sensitive to minor fluctuations in operating conditions compared to sensitive organometallic processes. This operational flexibility enables manufacturers to respond more quickly to changes in market demand, ensuring that customers receive their orders on time even during periods of high volatility. Moreover, the scalability of the hydroformylation process means that production capacity can be easily expanded to meet growing global demand for muguet notes in fine fragrances and functional perfumery applications.
- Scalability and Environmental Compliance: As the chemical industry moves towards greener manufacturing practices, this process offers a clear advantage in terms of environmental compliance and ease of scale-up. The avoidance of stoichiometric metal reagents and the reduction of organic waste streams simplify the wastewater treatment requirements, making it easier for facilities to meet local discharge limits. The use of continuous flow technology for the hydroformylation step is also highly feasible, allowing for safer handling of syngas and better heat management compared to large batch reactors. This adaptability to continuous processing not only improves safety but also facilitates the transition from pilot plant to commercial scale with minimal re-engineering. Consequently, companies adopting this technology can future-proof their operations against tightening environmental regulations while demonstrating a commitment to sustainable chemistry to their stakeholders.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these advanced fragrance intermediates. Understanding these details helps stakeholders evaluate the feasibility of integrating this technology into their existing supply chains. The answers are derived directly from the experimental data and embodiments disclosed in the patent documentation, ensuring accuracy and relevance for industrial application.
Q: What are the key advantages of the hydroformylation route over traditional Grignard methods for muguet aldehydes?
A: The hydroformylation route described in CN116018334A offers significantly higher atom economy and avoids the use of hazardous Grignard reagents. It provides superior regioselectivity for the linear aldehyde when using specific ligands like BIPHEPHOS, leading to reduced impurity profiles and simplified downstream purification compared to older radical or thermolysis methods.
Q: How does the process ensure the correct endo-isomer formation for the desired olfactory profile?
A: The process utilizes a specific elimination and isomerization step under acidic conditions. This step converts the initial exo-isomers (double bond in the alkyl chain) into the thermodynamically more stable endo-isomers (double bond within the cyclohexene ring), which are critical for the characteristic muguet scent note.
Q: Can this synthesis be scaled for industrial production of fragrance ingredients?
A: Yes, the patent explicitly describes conditions suitable for batch and continuous processing. The use of robust rhodium catalysts and standard industrial reagents like syngas and acetic anhydride supports scalability from kilogram to multi-ton annual production capacities with consistent quality control.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-(Cyclohex-1-en-1-yl)propanal Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-purity intermediates in defining the olfactory character of premium fragrances. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated hydroformylation and isomerization techniques described in CN116018334A are executed with precision. We maintain stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of 3-(cyclohex-1-en-1-yl)propanal derivatives meets the exacting standards required by the global perfumery industry. Our commitment to quality assurance means that we can consistently deliver materials with the correct endo/exo isomer ratio, preserving the integrity of the muguet scent profile for our clients' formulations.
We invite procurement leaders and R&D directors to collaborate with us to optimize their fragrance supply chains through a Customized Cost-Saving Analysis. By leveraging our technical expertise, we can help you evaluate the feasibility of transitioning to this more efficient synthetic route, potentially unlocking significant value in your bill of materials. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments tailored to your volume requirements. Together, we can drive innovation in fine fragrance manufacturing while achieving your strategic sourcing and sustainability goals.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
