Scaling High-Performance Fluorinated Bismaleimide Production for Aerospace and Electronics
The development of advanced composite materials for aerospace and high-performance electronics relies heavily on the availability of specialized resin monomers that can withstand extreme thermal and mechanical stress. Patent CN101921222B introduces a groundbreaking preparation method for 3,3',5,5'-tetramethyl-4,4'-bis(4-maleimido-2-trifluoromethylphenoxy)biphenyl, a sophisticated bismaleimide (BMI) monomer designed to address the limitations of traditional epoxy systems. This specific fluorinated biphenyl derivative offers superior heat resistance and chemical reactivity, enabling the creation of next-generation prepregs and insulation materials. As a reliable high-performance resin intermediate supplier, understanding the nuances of this synthesis is critical for R&D teams aiming to enhance the thermal stability of their final polymer products without compromising processability.
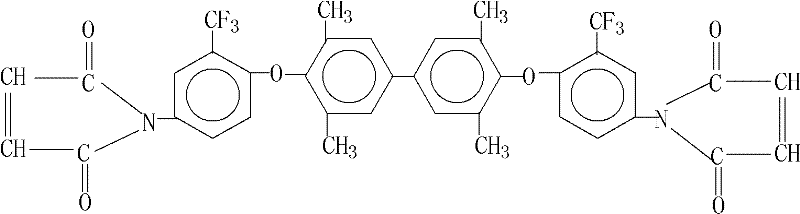
The strategic value of this patent lies in its ability to bridge the gap between laboratory-scale innovation and industrial feasibility. Historically, the synthesis of complex bismaleimides has been plagued by issues such as low purity, difficult purification steps, and the generation of hazardous waste. However, the methodology outlined in CN101921222B utilizes a streamlined two-step reaction sequence that maximizes atom economy and minimizes environmental impact. For procurement managers and supply chain heads, this translates to a more robust sourcing strategy for high-purity bismaleimide monomer, ensuring consistent quality for critical applications in printed circuit boards and aircraft structural components.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis routes for bismaleimide resins often suffer from significant inefficiencies that hinder their widespread adoption in cost-sensitive industries. Conventional methods frequently require harsh reaction conditions, including high pressures or the use of corrosive catalysts that complicate reactor maintenance and increase capital expenditure. Furthermore, older techniques often struggle with incomplete imidization, leading to products contaminated with maleamic acid intermediates which can detrimentally affect the curing profile and final thermal properties of the resin. The lack of effective dehydration strategies in legacy processes also results in lower yields and necessitates energy-intensive purification steps, driving up the overall cost reduction in advanced composite material manufacturing. These technical bottlenecks have historically limited the availability of high-quality BMI monomers, creating supply chain vulnerabilities for manufacturers of aerospace-grade composites.
The Novel Approach
The innovative process described in the patent overcomes these historical barriers through a meticulously optimized reaction environment that favors high conversion rates and product purity. By employing a specific combination of anhydrous calcium chloride and N,N-dimethylaniline during the cyclodehydration phase, the method effectively drives the equilibrium towards the desired imide formation while facilitating the removal of water via azeotropic distillation. This approach allows the reaction to proceed smoothly at atmospheric pressure, eliminating the need for expensive high-pressure vessels and enhancing operational safety. The result is a highly efficient synthesis that not only simplifies the workflow but also ensures that the final 3,3',5,5'-tetramethyl-4,4'-bis(4-maleimido-2-trifluoromethylphenoxy)biphenyl product meets stringent purity specifications required for electronic and aerospace applications.
Mechanistic Insights into Thermal Imidization and Cyclodehydration
The chemical transformation detailed in this patent involves a precise two-stage mechanism that begins with the formation of a bismaleamic acid intermediate followed by a catalytic cyclodehydration. In the first stage, the diamine precursor reacts with maleic anhydride in a polar aprotic solvent at room temperature, forming the amic acid linkage without generating excessive heat or side products. This mild initial step is crucial for maintaining the integrity of the sensitive fluorinated biphenyl backbone. The subsequent imidization step is where the true innovation lies; the addition of calcium chloride acts as a Lewis acid and dehydrating agent, coordinating with the carbonyl oxygen to activate the carboxylic acid group for nucleophilic attack by the amide nitrogen. Simultaneously, N,N-dimethylaniline serves as a basic catalyst, facilitating proton transfer and accelerating the ring-closure reaction to form the stable five-membered imide ring.
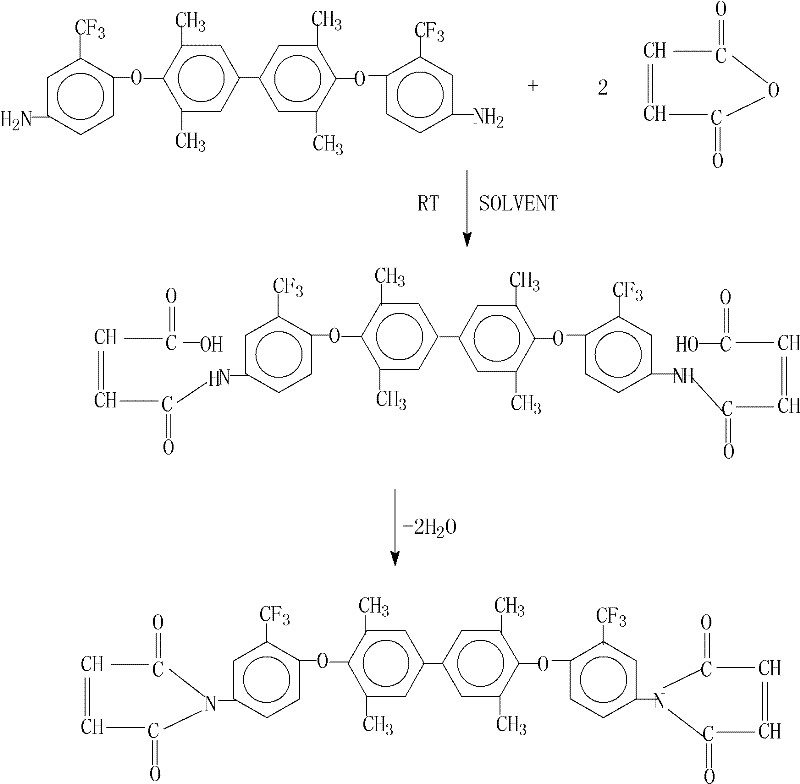
Controlling impurities in this synthesis is paramount, particularly the removal of unreacted maleic anhydride and the maleamic acid intermediate, which can act as plasticizers or degrade thermal performance. The patent describes a clever workup procedure where the azeotropic dehydrating agent, such as toluene or xylene, is distilled off and recycled, concentrating the reaction mixture before precipitation. The addition of ethanol as an anti-solvent induces the crystallization of the target bismaleimide while keeping soluble impurities in the mother liquor. This crystallization-driven purification mechanism ensures that the final powder possesses a sharp melting point and consistent molecular weight distribution, which are critical parameters for the commercial scale-up of complex polymer additives. The absence of corrosive byproducts further simplifies the downstream processing, allowing for the use of standard stainless steel equipment without specialized linings.
How to Synthesize 3,3',5,5'-Tetramethyl-4,4'-bis(4-maleimido-2-trifluoromethylphenoxy)biphenyl Efficiently
Executing this synthesis requires careful attention to solvent selection and stoichiometric ratios to replicate the high yields reported in the patent data. The process begins by dissolving the fluorinated diamine in a solvent like N-methyl-2-pyrrolidone (NMP) and adding maleic anhydride under controlled temperature conditions to prevent premature polymerization. Following the formation of the intermediate solution, the introduction of the catalytic dehydration system must be timed precisely to initiate the reflux process effectively. Operators should monitor the water separation closely to determine the endpoint of the reaction, ensuring complete conversion before proceeding to the isolation phase. The detailed standardized synthesis steps see the guide below for specific operational parameters and safety considerations.
- React 3,3',5,5'-tetramethyl-4,4'-bis(4-amino-2-trifluoromethylphenoxy)biphenyl with maleic anhydride in a polar aprotic solvent at room temperature to form the bismaleamic acid intermediate.
- Add anhydrous calcium chloride, an azeotropic dehydrating agent like toluene, and N,N-dimethylaniline, then heat to reflux to facilitate cyclodehydration and water removal.
- Distill off the dehydrating agent for recycling, cool the mixture, precipitate the product with ethanol, and filter to obtain the pure bismaleimide powder.
Commercial Advantages for Procurement and Supply Chain Teams
For organizations managing the procurement of specialty chemicals, the economic implications of this patented process extend far beyond the immediate reaction yield. The ability to operate at atmospheric pressure significantly reduces the capital investment required for reactor infrastructure, allowing for flexible manufacturing setups that can be scaled according to demand. Moreover, the protocol emphasizes the recovery and recycling of organic solvents such as NMP and toluene, which represents a substantial cost saving opportunity by minimizing raw material consumption and waste disposal fees. This closed-loop solvent management system aligns with modern environmental compliance standards, reducing the regulatory burden on manufacturing facilities and enhancing the overall sustainability profile of the supply chain.
- Cost Reduction in Manufacturing: The elimination of high-pressure equipment and corrosive reagents drastically lowers both the initial capital expenditure and ongoing maintenance costs for production facilities. By utilizing readily available starting materials and a catalyst system that promotes near-quantitative conversion, the process minimizes raw material waste and reduces the need for expensive purification chromatography. The efficient solvent recovery loop further contributes to financial efficiency, ensuring that the cost per kilogram of the final monomer remains competitive even at smaller production scales. This economic model supports a robust business case for domestic production of high-value electronic chemicals.
- Enhanced Supply Chain Reliability: The reliance on common industrial solvents and non-proprietary catalysts means that the supply chain is less vulnerable to disruptions caused by the scarcity of exotic reagents. The simplicity of the reaction conditions allows for production in a wider range of chemical plants, diversifying the potential supplier base and reducing lead time for high-purity electronic chemicals. Additionally, the stability of the intermediate and final products facilitates easier storage and transportation, mitigating risks associated with logistics and inventory management. This resilience is critical for maintaining continuous production schedules in the fast-paced aerospace and electronics sectors.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, with reaction parameters that translate linearly from laboratory benchtop to multi-ton industrial reactors. The absence of hazardous corrosive byproducts simplifies wastewater treatment and reduces the environmental footprint of the manufacturing process. By adhering to green chemistry principles through solvent recycling and high atom economy, manufacturers can meet increasingly strict environmental regulations without sacrificing productivity. This forward-thinking approach ensures long-term operational viability and positions the supply chain as a leader in sustainable chemical manufacturing practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this specialized bismaleimide monomer. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear picture of the technology's capabilities. Understanding these details is essential for technical teams evaluating the feasibility of integrating this material into their existing resin formulations or composite manufacturing lines.
Q: What are the key thermal properties of this fluorinated bismaleimide monomer?
A: The synthesized monomer exhibits a sharp melting point around 311.9°C and demonstrates excellent thermal stability, making it ideal for high-temperature thermosetting polyimide materials and epoxy modifications.
Q: How does the patented method improve production yield compared to conventional routes?
A: By utilizing a specific catalytic system comprising anhydrous calcium chloride and N,N-dimethylaniline during the imidization step, the process achieves yields exceeding 99%, significantly reducing raw material waste.
Q: Is this synthesis process suitable for large-scale industrial manufacturing?
A: Yes, the process operates at atmospheric pressure without requiring high-pressure equipment, uses recoverable solvents, and avoids corrosive substances, ensuring it is environmentally friendly and scalable.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,3',5,5'-Tetramethyl-4,4'-bis(4-maleimido-2-trifluoromethylphenoxy)biphenyl Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-performance monomers play in the development of advanced materials for the aerospace and electronics industries. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from R&D to full-scale manufacturing is seamless and efficient. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, guaranteeing the consistency and reliability required for mission-critical applications. Our expertise in handling fluorinated compounds and complex imidization reactions positions us as an ideal partner for your specialty chemical needs.
We invite you to collaborate with us to optimize your supply chain and reduce your overall material costs through our Customized Cost-Saving Analysis. Our technical procurement team is ready to provide specific COA data and route feasibility assessments tailored to your unique project requirements. By leveraging our manufacturing capabilities and deep technical knowledge, we can help you secure a stable supply of high-quality intermediates that drive innovation in your final products. Contact us today to discuss how we can support your next breakthrough in composite material technology.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
