Advanced Synthesis of Substituted Pyrazolopyrimidines for Commercial Pharmaceutical Production
Advanced Synthesis of Substituted Pyrazolopyrimidines for Commercial Pharmaceutical Production
The pharmaceutical landscape for central nervous system (CNS) disorders continuously demands high-purity intermediates capable of supporting large-scale production of active pharmaceutical ingredients (APIs). Patent CN1152033C presents a pivotal advancement in the synthesis of substituted pyrazolopyrimidines, a class of compounds exhibiting potent efficacy as anxiolytics, anticonvulsants, and sedative-hypnotics, particularly for the treatment of insomnia. This intellectual property outlines sophisticated chemical pathways that overcome the limitations of prior art, specifically addressing the inefficiencies associated with multi-step isolations and harsh reaction conditions found in earlier patents like U.S. Patent No. 4,521,422. By leveraging novel cyclization strategies and one-pot methodologies, this technology enables the robust manufacture of complex heterocyclic cores essential for modern drug development.
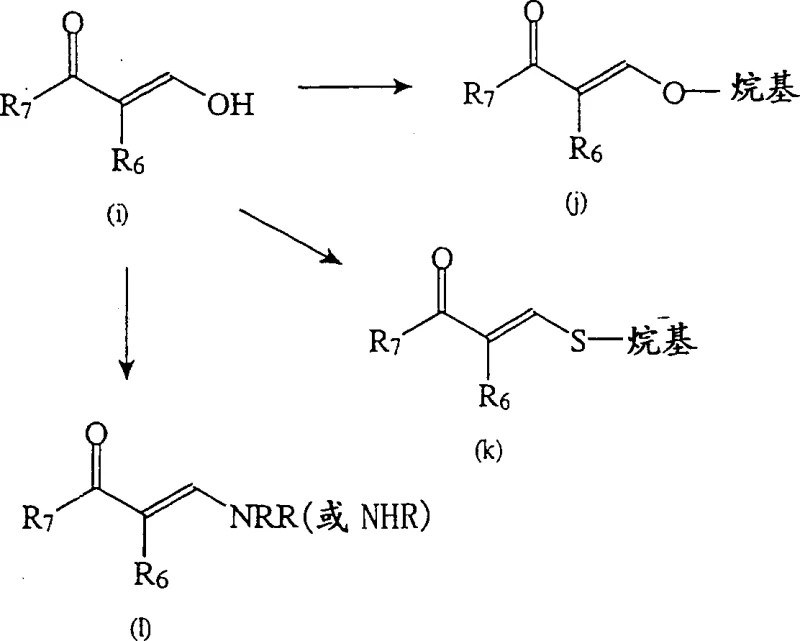
For procurement specialists and supply chain managers, the implications of this patent extend beyond mere chemical novelty; it represents a tangible opportunity for cost reduction in API manufacturing through process intensification. The ability to synthesize these critical scaffolds without isolating unstable intermediates translates directly into reduced operational expenditures and enhanced throughput. As a reliable pharmaceutical intermediate supplier, understanding these mechanistic nuances is crucial for ensuring consistent quality and supply continuity for downstream drug manufacturers who rely on these building blocks for treating sleep disorders and related neurological conditions.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of structure (I) pyrazolopyrimidines, as detailed in foundational patents such as U.S. Patent No. 4,521,422 and 4,900,836, relied heavily on linear sequences that necessitated the isolation of discrete pyrazole intermediates. These conventional routes often involved reacting substituted pyrazoles with 3-dimethylamino-2-propen-1-ones, a process that, while chemically sound, proved relatively time-consuming and economically inefficient for large-scale operations. The requirement to isolate and purify intermediates at multiple stages introduced significant points of potential yield loss and increased the consumption of solvents and energy. Furthermore, the handling of certain intermediates posed stability challenges, complicating inventory management and extending lead times for high-purity pharmaceutical intermediates. These legacy methods lacked the agility required for modern commercial scale-up of complex heterocyclic compounds, often resulting in higher production costs and environmental burdens due to excessive waste generation.
The Novel Approach
In stark contrast, the methodology disclosed in CN1152033C introduces streamlined synthetic routes that bypass the need for intermediate isolation, thereby drastically simplifying the manufacturing workflow. A prime example is the "one-pot" technique where critical cyclization steps are performed in a single reaction vessel, allowing for the direct conversion of starting materials into the desired pyrazolopyrimidine core. This approach not only enhances efficiency but also mitigates the risks associated with handling reactive species. Additionally, the patent describes versatile functionalization strategies, such as the use of enaminone 8 in acetic acid for cyclization, which offers superior control over regioselectivity and impurity profiles. By integrating these improved techniques, manufacturers can achieve substantial cost savings and operational flexibility, making the production of compounds like N-methyl-N-(3-{3-[2-thienylcarbonyl]-pyrazol-[1,5-alpha]-pyrimidin-7-yl}phenyl)acetamide far more commercially viable.
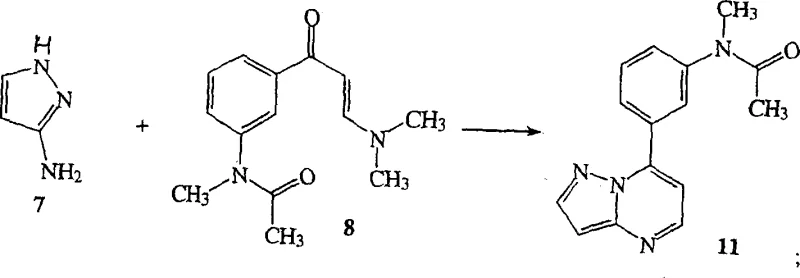
Mechanistic Insights into Pyrazolopyrimidine Core Construction
The chemical architecture of these therapeutic agents relies on the precise construction of the pyrazolo[1,5-a]pyrimidine ring system, a process that demands rigorous control over reaction parameters to ensure high purity. The patent elucidates several mechanistic pathways, including the cyclization of aminopyrazoles with enaminones, which serves as the foundational step for building the heterocyclic template. In one embodiment, the reaction of aminopyrazole 7 with enaminone 8 in refluxing acetic acid facilitates the formation of the pyrazolopyrimidine 11 through a condensation-cyclization sequence. This mechanism is highly advantageous as it tolerates various substituents, allowing for the generation of a diverse library of analogues. Furthermore, the subsequent functionalization of the core can be achieved through robust cross-coupling reactions, such as the Suzuki coupling of halogenated intermediates with boronic acids, or via Grignard additions to nitrile precursors, providing chemists with multiple handles to introduce the requisite thiophene carbonyl moiety.
Impurity control is inherently built into these novel pathways through the minimization of unit operations. By avoiding the isolation of intermediates like the pyrazole species, the process reduces the exposure of reactive compounds to potential degradation pathways that often generate difficult-to-remove byproducts. The use of phase transfer catalysis in alkylation steps, employing agents like tetrabutylammonium sulfate in biphasic systems, further refines the process by ensuring complete conversion of starting materials under mild conditions. This meticulous attention to reaction engineering ensures that the final API intermediate meets stringent purity specifications required by regulatory bodies. For R&D directors, this level of mechanistic clarity provides confidence in the scalability and reproducibility of the synthesis, ensuring that the transition from laboratory bench to commercial reactor proceeds without unforeseen chemical hurdles.
How to Synthesize N-methyl-N-(3-{3-[2-thienylcarbonyl]-pyrazol-[1,5-alpha]-pyrimidin-7-yl}phenyl)acetamide Efficiently
The synthesis of this specific compound, often referred to as Compound 1, exemplifies the efficiency gains possible through the patented methodologies. The process typically initiates with the preparation of key intermediates, such as the enaminone 8, which can be generated via phase transfer alkylation of acetophenone derivatives. Following this, the core assembly involves the condensation of the appropriate aminopyrazole with the enaminone in a polar protic solvent like acetic acid. The detailed standardized synthesis steps see the guide below, which outlines the precise stoichiometry, temperature profiles, and workup procedures necessary to maximize yield. This structured approach ensures that even complex transformations, such as the introduction of the 2-thienylcarbonyl group via Friedel-Crafts acylation or metal-catalyzed coupling, are executed with high fidelity.
- Cyclize aminopyrazole intermediates with enaminone 8 in acetic acid to form the pyrazolopyrimidine core.
- Perform functionalization via Friedel-Crafts acylation with 2-thiophenoyl chloride or Suzuki coupling with boronic acids.
- Utilize one-pot techniques to avoid isolation of unstable pyrazole intermediates, enhancing overall yield and purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of the synthetic routes described in CN1152033C offers compelling economic and logistical benefits that directly impact the bottom line. The shift from multi-step isolation processes to streamlined one-pot reactions fundamentally alters the cost structure of manufacturing these high-value intermediates. By reducing the number of discrete processing steps, facilities can lower their consumption of utilities, solvents, and labor hours, leading to significant operational efficiencies. Moreover, the robustness of these chemical pathways ensures a more predictable supply timeline, mitigating the risks of production delays that often plague complex organic syntheses. This reliability is paramount for maintaining continuous API production schedules and meeting the demanding delivery windows of global pharmaceutical clients.
- Cost Reduction in Manufacturing: The elimination of intermediate isolation steps serves as a primary driver for cost optimization in this synthesis. Traditional methods requiring the purification of pyrazole intermediates incur substantial costs related to chromatography, crystallization, and drying operations, all of which consume time and resources. By performing these transformations in a telescoped manner, the process minimizes material loss and reduces the volume of waste solvents generated, aligning with green chemistry principles. Additionally, the use of commercially available reagents and standard catalysts avoids the need for exotic or prohibitively expensive proprietary materials, further driving down the raw material costs associated with producing high-purity pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: The versatility of the synthetic routes provides a strategic buffer against supply chain disruptions. Since the patent discloses multiple pathways to the same target molecule—such as using either Grignard reagents or Suzuki coupling for the thiophene introduction—manufacturers are not locked into a single dependency. If a specific reagent faces global shortages, the process can be adapted to utilize an alternative chemical route without compromising the final product quality. This flexibility ensures a steady flow of materials to downstream customers, reinforcing the supplier's reputation for dependability. Furthermore, the simplified processing requirements allow for faster batch turnover, effectively reducing lead time for high-purity pharmaceutical intermediates and enabling quicker response to market demand fluctuations.
- Scalability and Environmental Compliance: Scaling complex heterocyclic syntheses often presents challenges regarding heat transfer and mixing, but the disclosed methods utilize standard reaction conditions that are readily adaptable to large-scale reactors. The avoidance of hazardous intermediates and the reduction in solvent usage contribute to a safer working environment and simplify waste treatment protocols. This alignment with environmental compliance standards reduces the regulatory burden on manufacturing sites and lowers the costs associated with waste disposal. Consequently, the commercial scale-up of complex pharmaceutical intermediates becomes a more sustainable and economically attractive endeavor, supporting long-term production viability without compromising on safety or environmental stewardship.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of these pyrazolopyrimidine derivatives. The answers are derived directly from the technical specifications and experimental data provided within the patent documentation, ensuring accuracy and relevance for industry professionals. Understanding these details is essential for evaluating the feasibility of integrating this technology into existing manufacturing portfolios.
Q: What are the primary advantages of the one-pot synthesis method described in CN1152033C?
A: The one-pot technique eliminates the need to isolate reactive pyrazole intermediates, significantly reducing processing time, solvent usage, and material loss during purification steps.
Q: Which coupling strategies are viable for introducing the thiophene moiety?
A: The patent discloses multiple robust strategies including Friedel-Crafts acylation using Lewis acids, Grignard addition to nitrile intermediates, and Palladium-catalyzed Suzuki coupling with boronic acids.
Q: How does phase transfer catalysis improve the alkylation steps?
A: Using phase transfer catalysts like tetrabutylammonium sulfate allows efficient N-alkylation in biphasic systems, improving reaction kinetics and simplifying workup procedures compared to traditional anhydrous methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-methyl-N-(3-{3-[2-thienylcarbonyl]-pyrazol-[1,5-alpha]-pyrimidin-7-yl}phenyl)acetamide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development of CNS therapeutics. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated chemistry described in CN1152033C can be translated into reliable industrial reality. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of N-methyl-N-(3-{3-[2-thienylcarbonyl]-pyrazol-[1,5-alpha]-pyrimidin-7-yl}phenyl)acetamide meets the exacting standards required for clinical and commercial applications. Our commitment to process excellence means that we can deliver consistent quality while optimizing the cost structure for our partners.
We invite pharmaceutical companies and research institutions to collaborate with us to leverage these advanced synthetic capabilities. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and project timelines. We encourage you to reach out today to obtain specific COA data and route feasibility assessments that demonstrate how our optimized manufacturing processes can accelerate your drug development programs. Let us be your partner in bringing effective insomnia treatments to the market through superior chemical innovation and supply chain reliability.
