Revolutionizing Optoelectronic Intermediate Production: High-Yield Synthesis of 2,5-Disubstituted Terephthalaldehyde
The global demand for high-performance optoelectronic materials continues to surge, driving the need for robust and scalable synthetic routes for key building blocks like 2,5-disubstituted-1,4-terephthalaldehyde. Patent CN112538007B introduces a groundbreaking preparation method that addresses long-standing inefficiencies in organic synthesis, specifically targeting the production of these vital dialdehyde intermediates. This technology leverages a sophisticated catalytic system involving tert-butyllithium (t-BuLi) and hexamethylphosphoric triamide (HMPA) to transform 2,5-disubstituted-1,4-dibromobenzene precursors into high-purity aldehydes. For R&D directors and procurement specialists in the electronic chemical sector, this innovation represents a paradigm shift from low-yield, cumbersome processes to a streamlined, high-efficiency workflow. The patent details how the synergistic action of t-BuLi and trace HMPA not only accelerates reaction kinetics but also stabilizes critical intermediates, ensuring consistent product quality essential for downstream applications in Covalent Organic Frameworks (COFs) and advanced display technologies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2,5-disubstituted-1,4-terephthalaldehyde has been plagued by significant technical hurdles that hinder commercial viability. Traditional methodologies predominantly rely on the use of n-butyllithium (n-BuLi) for low-temperature halogen extraction, a process that is notoriously finicky and inefficient. As cited in the background art of the patent, literature reports indicate that even when employing excessive equivalents of n-butyllithium, such as four equivalents, the isolated yields remain dismally low, often hovering around merely 13%. Other attempts using varying amounts of n-butyllithium on different alkyl-substituted dibromobenzenes have shown marginal improvements, with yields stagnating between 15% and 32%. These poor conversion rates are attributed to the instability of the lithiated intermediates and the prevalence of competing side reactions that consume the starting material without generating the desired dialdehyde. Furthermore, the post-processing associated with these conventional routes is excessively繁琐 (cumbersome), requiring complex purification steps to separate the target product from a myriad of byproducts, which drastically increases manufacturing costs and waste generation.
The Novel Approach
In stark contrast to these legacy methods, the novel approach disclosed in Patent CN112538007B utilizes tert-butyllithium as the primary lithiating agent, supported by a catalytic amount of HMPA. This strategic substitution fundamentally alters the reaction landscape, resulting in a dramatic enhancement of reaction activity. The steric bulk and higher reactivity of tert-butyllithium facilitate a more rapid and complete halogen-lithium exchange compared to its linear n-butyl counterpart. Crucially, the addition of trace HMPA serves to stabilize the existence form of the carbocation ions generated during the process. This stabilization effect遏制 (curbs) the occurrence of excessive side reactions, allowing the system to maintain high conversion efficiency even under standard laboratory conditions. The result is a robust synthetic route that consistently delivers yields of 78% or higher, effectively more than doubling the output of traditional methods. This leap in efficiency translates directly to reduced raw material consumption and a significantly simplified downstream purification process, making it an ideal candidate for cost reduction in electronic chemical manufacturing.
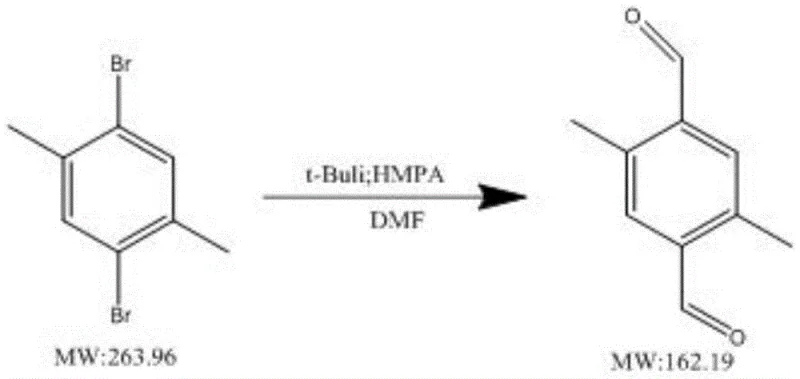
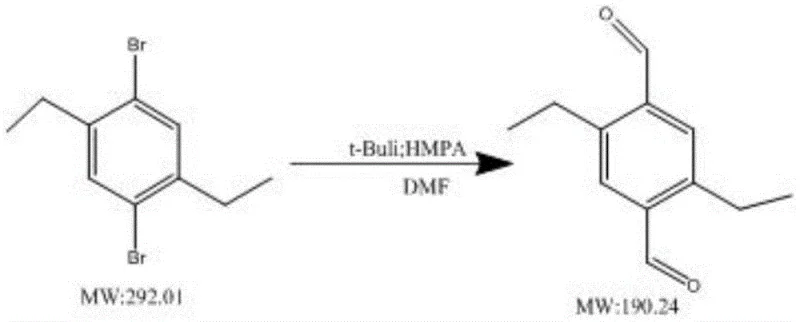
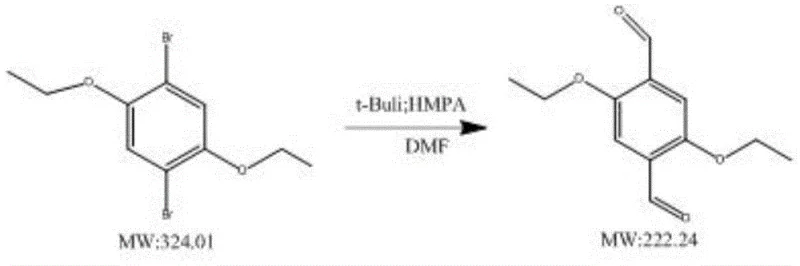
Mechanistic Insights into t-BuLi and HMPA Synergistic Catalysis
The core of this technological breakthrough lies in the intricate interplay between the organolithium reagent and the polar aprotic additive. When tert-butyllithium is introduced to the 2,5-disubstituted-1,4-dibromobenzene substrate at cryogenic temperatures (typically -75°C to -80°C), it initiates a rapid halogen-metal exchange. Unlike n-butyllithium, which can suffer from slower kinetics and aggregation issues, t-BuLi provides a highly reactive species that efficiently generates the dilithio-intermediate. However, the true genius of the system is the role of HMPA. As a strong Lewis base, HMPA coordinates strongly with the lithium cations. This coordination breaks up aggregates of the organolithium species, effectively "naked" the carbanions and increasing their nucleophilicity. More importantly, in the context of this specific oxidation reaction with DMF, HMPA stabilizes the transition states and the resulting carbocation-like character of the intermediates. This stabilization is critical because it prevents the premature decomposition or protonation of the reactive species before they can react with the DMF electrophile. By maintaining the integrity of the lithiated intermediate, the system ensures that the subsequent formylation step proceeds with high fidelity.
Furthermore, the mechanism dictates strict control over impurity profiles, which is a primary concern for R&D directors focusing on purity specifications. The suppression of side reactions, such as Wurtz-type coupling or elimination reactions, is directly linked to the stability imparted by the HMPA additive. In the absence of HMPA, as demonstrated in comparative examples within the patent, the yield drops precipitously to around 45%, indicating that the intermediates are too unstable to survive the reaction timeline. The optimized molar ratio of substrate to HMPA to t-BuLi (1:0.02-0.05:2.1-2.5) creates a "sweet spot" where the concentration of active species is maximized without inducing thermal runaway or uncontrolled reactivity. This precise balance allows for the commercial scale-up of complex optoelectronic intermediates with predictable and reproducible outcomes, ensuring that the final product meets the stringent purity requirements necessary for high-end applications like OLEDs and semiconductor processing.
How to Synthesize 2,5-Disubstituted-1,4-terephthalaldehyde Efficiently
The operational protocol for this synthesis is designed to be both rigorous and scalable, ensuring that laboratory success can be translated into industrial reality. The process begins with the precise weighing of the dibromobenzene derivative and the HMPA catalyst, followed by dissolution in an appropriate solvent system within a three-necked flask equipped for inert atmosphere handling. The reaction mixture is subjected to a dry ice-ethanol bath to achieve temperatures between -75°C and -80°C, a critical parameter for controlling the exothermic nature of the lithiation. Once thermal equilibrium is reached, the tert-butyllithium is added dropwise over a controlled period, typically one hour, to manage heat evolution and ensure uniform mixing. After the addition is complete, the system is allowed to warm to room temperature and stir for a defined period to complete the halogen exchange. Subsequently, the mixture is re-cooled, and DMF is introduced as the formylating agent. The detailed standardized synthesis steps, including specific quenching and purification protocols, are outlined below to guide process engineers in replicating this high-yield transformation.
- Prepare the reaction system by weighing 2,5-disubstituted-1,4-dibromobenzene and HMPA, cooling to -78°C in a dry ice ethanol bath, and slowly adding tert-butyllithium.
- Warm the mixture to room temperature for stirring, then re-cool to -78°C before dropwise addition of DMF as the formylating agent.
- Quench the reaction with saturated ammonium chloride, extract with ethyl acetate, and purify the crude white solid via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology offers compelling economic and logistical benefits that extend far beyond simple yield metrics. The primary advantage lies in the drastic simplification of the manufacturing cost structure. By eliminating the need for massive excesses of reagents and reducing the formation of intractable byproducts, the process inherently lowers the cost of goods sold (COGS). The high conversion rate means that less raw material is wasted, and the burden on waste treatment facilities is significantly reduced, aligning with modern environmental compliance standards. Moreover, the robustness of the t-BuLi/HMPA system reduces the risk of batch failures, a common issue with sensitive organolithium chemistry. This reliability enhances supply chain continuity, ensuring that critical intermediates for optoelectronic device production are available without interruption. The ability to source high-purity terephthalaldehyde derivatives from a reliable optoelectronic material intermediate supplier who utilizes this technology translates to greater stability in the downstream production of finished electronic components.
- Cost Reduction in Manufacturing: The implementation of this catalytic system drives down manufacturing expenses through multiple mechanisms. Firstly, the substantial increase in yield—from roughly 30% in conventional methods to over 78%—means that the effective cost per kilogram of the final product is less than half of previous benchmarks, assuming similar raw material costs. Secondly, the reaction conditions allow for the use of stoichiometric amounts of t-BuLi rather than the large excesses often required to force n-BuLi reactions to completion, further reducing reagent costs. Thirdly, the simplified workup procedure, which involves standard aqueous quenching and extraction rather than complex chromatographic separations required for dirty reaction mixtures, reduces labor hours and solvent consumption. These factors combine to create a leaner, more cost-effective production model that maximizes margin potential for high-volume buyers.
- Enhanced Supply Chain Reliability: Supply chain resilience is heavily dependent on the predictability of the manufacturing process. Conventional low-yield syntheses are prone to variability; small deviations in temperature or addition rates can lead to catastrophic drops in yield, causing delivery delays. The t-BuLi/HMPA method described in the patent demonstrates remarkable tolerance and consistency across different substrates, including methyl, ethyl, and ethoxy derivatives. This consistency allows suppliers to provide accurate lead times and maintain safety stock levels with confidence. For buyers, this means reducing lead time for high-purity optoelectronic intermediates becomes a tangible reality, as the risk of production bottlenecks caused by failed batches is minimized. The availability of stable, high-quality feedstock supports just-in-time manufacturing strategies for electronics producers.
- Scalability and Environmental Compliance: Scaling organolithium reactions is often fraught with safety and environmental challenges due to pyrophoric reagents and hazardous waste. However, the efficiency of this new method mitigates some of these risks by shortening reaction times and reducing the total volume of chemical waste generated per unit of product. The high atom economy implies that fewer hazardous byproducts enter the waste stream, simplifying effluent treatment and lowering disposal costs. Additionally, the process operates at manageable temperatures (-78°C to RT) that are achievable with standard industrial cooling systems, avoiding the need for extreme cryogenic infrastructure. This ease of scale-up facilitates the commercial production of complex polymer additives and electronic chemicals, ensuring that supply can meet the growing global demand for advanced materials without compromising on safety or regulatory adherence.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and beneficial effects reported in Patent CN112538007B, providing a transparent view of the process capabilities. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this intermediate into their own supply chains. The data confirms that the method is not only theoretically sound but practically validated across multiple examples with consistent results.
Q: Why is tert-butyllithium preferred over n-butyllithium for this synthesis?
A: According to Patent CN112538007B, tert-butyllithium significantly improves reaction activity compared to n-butyllithium. While n-butyllithium methods historically yield only 13-32%, the t-BuLi system achieves yields exceeding 78% by facilitating more efficient halogen-lithium exchange and reducing side reactions.
Q: What is the specific role of HMPA in this catalytic system?
A: HMPA (hexamethylphosphoric triamide) acts as a crucial co-catalyst that stabilizes the carbocation intermediates formed during the reaction. Its presence prevents the decomposition of reactive species, thereby drastically increasing the conversion rate and overall product quantification efficiency.
Q: What represents the typical yield improvement of this new method?
A: The patented process consistently delivers yields of 78% or higher across various substrates, including methyl, ethyl, and ethoxy substituted derivatives. This is a substantial improvement over conventional methods which often struggle to reach 30% yield.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,5-Disubstituted-1,4-terephthalaldehyde Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent literature to commercial reality requires deep technical expertise and state-of-the-art infrastructure. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising yields seen in the lab are maintained at the plant level. Our commitment to quality is unwavering; we adhere to stringent purity specifications and utilize rigorous QC labs to verify every batch of 2,5-disubstituted-1,4-terephthalaldehyde against the highest industry standards. Whether your application involves the fabrication of next-generation OLED displays or the synthesis of advanced Covalent Organic Frameworks, our team is equipped to deliver the high-purity electronic chemical intermediates you need to drive innovation.
We invite you to collaborate with us to optimize your supply chain and reduce your overall material costs. By leveraging our mastery of the t-BuLi/HMPA catalytic system, we can offer a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage potential partners to contact our technical procurement team to request specific COA data and route feasibility assessments. Let us demonstrate how our advanced synthesis capabilities can become a cornerstone of your sourcing strategy, delivering both value and reliability in a competitive market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
