Advanced Asymmetric Copper Catalysis for Scalable Production of Chiral Cyclopropane Intermediates
Advanced Asymmetric Copper Catalysis for Scalable Production of Chiral Cyclopropane Intermediates
The landscape of chiral synthesis for agrochemical and pharmaceutical intermediates is constantly evolving, driven by the need for higher enantiomeric purity and more sustainable catalytic processes. A pivotal advancement in this field is documented in patent CN1968958A, which discloses a highly efficient method for producing optically active cyclopropanecarboxylic ester compounds. These compounds serve as critical building blocks for synthetic pyrethroid insecticides and various pharmaceutical agents, where stereochemistry dictates biological activity. The core innovation lies in the generation of a novel asymmetric copper complex formed by the precise combination of a copper source, a chiral bisoxazoline ligand, and a specific boron compound activator. This tripartite catalyst system addresses long-standing challenges in cyclopropanation reactions, specifically regarding the control of diastereoselectivity and enantioselectivity. For R&D directors and procurement specialists seeking a reliable agrochemical intermediate supplier, understanding the mechanistic advantages of this patented route is essential for securing supply chains that demand rigorous quality specifications and cost-effective manufacturing protocols.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of optically active cyclopropane derivatives has relied heavily on rhodium-based catalysts or simpler copper systems that often suffer from suboptimal stereoselectivity and lower turnover numbers. Conventional copper-bisoxazoline complexes, while effective to a degree, frequently struggle to maintain high enantiomeric excess (e.e.) across a broad range of olefin substrates, particularly when scaling up from laboratory to commercial production. Without specific activators, these traditional catalysts may yield mixtures of cis and trans isomers that require expensive and wasteful downstream purification steps, such as repeated recrystallization or preparative chromatography, to isolate the desired bioactive isomer. Furthermore, the instability of certain diazo compounds in the presence of less optimized catalytic environments can lead to safety hazards and reduced overall process efficiency. These limitations translate directly into higher operational expenditures and longer lead times for high-purity intermediates, creating bottlenecks for manufacturers aiming to reduce costs in pharmaceutical manufacturing and agrochemical production.
The Novel Approach
The methodology outlined in the patent introduces a transformative improvement by incorporating a boron compound into the standard copper-bisoxazoline framework. This modification creates a highly active cationic copper species that exhibits superior control over the approach of the diazo compound to the olefin double bond. 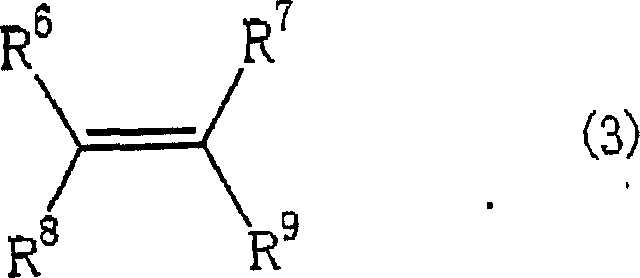 As demonstrated in the experimental data, the inclusion of activators like trityl tetrakis(pentafluorophenyl)borate boosts reaction yields significantly, reaching levels around 80% compared to roughly 60% in non-activated comparative examples. Moreover, the trans-to-cis isomer ratio improves markedly, favoring the thermodynamically stable and often biologically preferred trans-isomer. This novel approach not only enhances the chemical efficiency but also simplifies the purification workflow, making it an attractive option for the commercial scale-up of complex polymer additives and fine chemical intermediates. The ability to tune the catalyst by selecting specific bisoxazoline ligands allows for further optimization tailored to specific substrate requirements.
As demonstrated in the experimental data, the inclusion of activators like trityl tetrakis(pentafluorophenyl)borate boosts reaction yields significantly, reaching levels around 80% compared to roughly 60% in non-activated comparative examples. Moreover, the trans-to-cis isomer ratio improves markedly, favoring the thermodynamically stable and often biologically preferred trans-isomer. This novel approach not only enhances the chemical efficiency but also simplifies the purification workflow, making it an attractive option for the commercial scale-up of complex polymer additives and fine chemical intermediates. The ability to tune the catalyst by selecting specific bisoxazoline ligands allows for further optimization tailored to specific substrate requirements.
Mechanistic Insights into Boron-Activated Asymmetric Copper Catalysis
The heart of this technological breakthrough is the formation of a well-defined chiral environment around the copper center, facilitated by the unique interaction between the metal, the ligand, and the boron activator. The optically active bisoxazoline ligand, represented by Formula (1) in the patent, acts as a bidentate chelator that locks the copper atom into a rigid geometry. 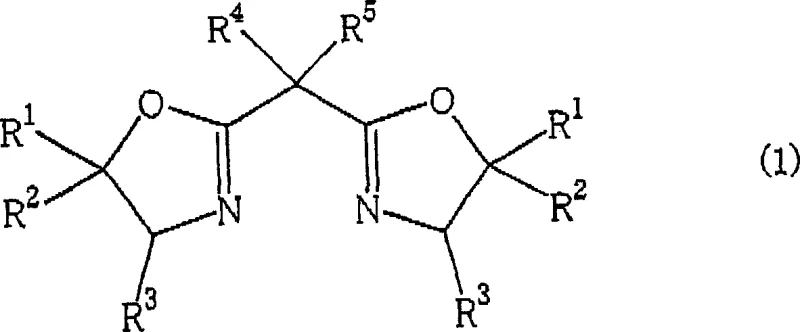 When combined with a boron compound possessing a weakly coordinating anion, such as tetraarylborates, the copper complex becomes cationic. This cationic nature increases the electrophilicity of the copper-carbenoid intermediate formed upon reaction with the diazoacetate. The increased electrophilicity accelerates the rate of cyclopropanation, thereby minimizing side reactions such as dimerization of the diazo compound or non-selective background reactions. The steric bulk of the substituents on the bisoxazoline ring, such as tert-butyl or phenyl groups at the 4-position, creates a chiral pocket that discriminates between the two faces of the approaching olefin. This discrimination is the fundamental driver of the high enantiomeric excess observed, often exceeding 95% e.e. for the major trans-isomer. Understanding this mechanism allows process chemists to predict catalyst performance and select the optimal ligand-copper-boron combination for new substrate classes.
When combined with a boron compound possessing a weakly coordinating anion, such as tetraarylborates, the copper complex becomes cationic. This cationic nature increases the electrophilicity of the copper-carbenoid intermediate formed upon reaction with the diazoacetate. The increased electrophilicity accelerates the rate of cyclopropanation, thereby minimizing side reactions such as dimerization of the diazo compound or non-selective background reactions. The steric bulk of the substituents on the bisoxazoline ring, such as tert-butyl or phenyl groups at the 4-position, creates a chiral pocket that discriminates between the two faces of the approaching olefin. This discrimination is the fundamental driver of the high enantiomeric excess observed, often exceeding 95% e.e. for the major trans-isomer. Understanding this mechanism allows process chemists to predict catalyst performance and select the optimal ligand-copper-boron combination for new substrate classes.
Impurity control is another critical aspect managed by this catalytic system. In traditional methods, the formation of homocoupling products from the diazo reagent is a common impurity that complicates isolation. The rapid and selective transfer of the carbene unit to the olefin in this boron-activated system suppresses these parasitic pathways. Additionally, the high diastereoselectivity (favoring the trans-isomer) reduces the burden on downstream separation units. Since the cis-isomer often possesses different physical properties and potentially different biological activities, its minimization is crucial for meeting stringent regulatory specifications for pesticide and drug intermediates. The robustness of the catalyst system, which tolerates various functional groups on the olefin such as esters, ethers, and halides, ensures that the impurity profile remains consistent and manageable even when switching between different batches of raw materials. This consistency is vital for maintaining supply chain reliability and ensuring that every batch meets the required purity standards without extensive reprocessing.
How to Synthesize Optically Active Cyclopropanecarboxylate Efficiently
Implementing this synthesis route requires careful attention to the preparation of the catalyst and the addition protocol of the reagents to maximize safety and yield. The process begins with the in situ generation of the active catalyst species under an inert atmosphere to prevent oxidation or moisture deactivation. The specific order of mixing—typically combining the copper salt and ligand first, followed by the boron activator—ensures the correct coordination geometry is established before the reactive diazo species is introduced. Once the catalyst is formed, the olefin substrate is added, and the diazoacetate is introduced slowly to control the exotherm and maintain high selectivity. The detailed standardized synthesis steps see the guide below.
- Prepare the asymmetric copper complex by mixing a monovalent or divalent copper compound, an optically active bisoxazoline ligand, and a specific boron compound in a suitable solvent under inert atmosphere.
- Combine the prepared catalyst solution with the target olefin substrate, ensuring the reaction mixture is maintained at a controlled temperature between -20°C and 80°C.
- Slowly add the diazoacetic acid ester to the reaction mixture over several hours, then stir to complete the cyclopropanation reaction before isolating the product via distillation or chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this boron-activated copper catalysis offers tangible benefits that extend beyond simple chemical yield. The primary advantage lies in the drastic simplification of the purification process. By achieving high diastereoselectivity and enantioselectivity directly in the reactor, the need for multiple crystallization steps or expensive chiral chromatography is significantly reduced. This reduction in downstream processing translates directly into lower manufacturing costs and shorter production cycles. Furthermore, the use of copper, an abundant and inexpensive base metal, compared to precious metals like rhodium or palladium, represents a substantial cost saving in raw material expenditure. The catalyst loading can be kept relatively low while maintaining high activity, further enhancing the economic viability of the process for large-scale production runs.
- Cost Reduction in Manufacturing: The elimination of expensive precious metal catalysts and the reduction in solvent usage due to fewer purification steps create a leaner manufacturing process. The high yield obtained minimizes waste generation, aligning with green chemistry principles and reducing disposal costs. Additionally, the ability to use commercially available copper salts and ligands without requiring exotic custom synthesis lowers the barrier to entry for production. The qualitative improvement in process efficiency means that facilities can produce more product per unit of time and resource, effectively lowering the cost of goods sold (COGS) for these high-value intermediates.
- Enhanced Supply Chain Reliability: The robustness of the catalyst system against variations in reaction conditions ensures consistent output quality, which is critical for maintaining long-term supply contracts. The use of stable and readily available reagents reduces the risk of supply disruptions associated with scarce or geopolitically sensitive materials. By optimizing the reaction to proceed at moderate temperatures and pressures, the process becomes safer and easier to operate in standard multipurpose reactors, increasing the flexibility of manufacturing sites to switch between different products as market demand fluctuates. This flexibility is a key asset in managing inventory levels and responding quickly to customer needs.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing solvents and conditions that are compatible with standard industrial equipment. The high atom economy of the cyclopropanation reaction, combined with the high selectivity, results in less chemical waste, simplifying compliance with increasingly strict environmental regulations. The ability to recycle solvents and the potential for catalyst recovery further enhance the sustainability profile of the manufacturing route. For companies aiming to reduce their carbon footprint and meet corporate sustainability goals, adopting this efficient catalytic method provides a clear pathway to greener production of essential agrochemical and pharmaceutical ingredients.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this asymmetric synthesis technology. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing a reliable basis for process development decisions. Understanding these nuances helps technical teams evaluate the feasibility of integrating this method into existing production lines.
Q: What is the primary advantage of adding a boron compound to the copper catalyst system?
A: According to patent CN1968958A, the addition of a boron compound, such as trityl tetrakis(pentafluorophenyl)borate, significantly enhances both the chemical yield and the stereoselectivity (trans/cis ratio and enantiomeric excess) compared to conventional copper-bisoxazoline systems lacking this activator.
Q: What types of substrates are compatible with this asymmetric cyclopropanation method?
A: The process demonstrates broad substrate scope, successfully reacting various olefins including 3-methyl-2-butenyl acetate and (3-methyl-2-butenyl) benzyl ether with diazoacetates to produce high-value chiral intermediates for pyrethroids and pharmaceuticals.
Q: Can divalent copper compounds be used instead of monovalent copper salts?
A: Yes, the patent explicitly states that divalent copper compounds like copper(II) chloride can be utilized, often in conjunction with a reducing agent such as phenylhydrazine, to generate the active monovalent copper species in situ without compromising optical purity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Optically Active Cyclopropanecarboxylate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality chiral intermediates play in the development of next-generation agrochemicals and pharmaceuticals. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that utilize advanced analytical techniques to verify enantiomeric excess and isomer ratios. Our facility is equipped to handle the specific requirements of air- and moisture-sensitive catalytic reactions, guaranteeing the consistency and reliability that global supply chains demand.
We invite you to collaborate with us to leverage this advanced catalytic technology for your specific project needs. Whether you require a Customized Cost-Saving Analysis for your current supply chain or need to validate the feasibility of a new route, our experts are ready to assist. Please contact our technical procurement team to request specific COA data and route feasibility assessments tailored to your volume requirements. Let us be your partner in driving innovation and efficiency in the production of complex fine chemical intermediates.
