Advanced Chiral Copper Catalysis for Scalable Agrochemical Intermediate Manufacturing
The pharmaceutical and agrochemical industries are constantly seeking more efficient pathways to synthesize chiral building blocks, and patent CN1798609A presents a significant breakthrough in this domain. This intellectual property discloses a novel optically active copper catalyst composition that fundamentally improves the asymmetric cyclopropanation process, a critical step in producing pyrethroid insecticides and other bioactive molecules. Unlike traditional catalysts that often struggle with activity at lower temperatures or leave high levels of unreacted hazardous diazo compounds, this invention introduces a ternary system comprising a specific salicylideneaminoalcohol ligand, a copper source, and a crucial Lewis acidic or lithium additive. For R&D directors and process chemists, this represents a tangible opportunity to enhance reaction profiles, achieving yields as high as 96% with superior enantiomeric excess. The ability to operate effectively at temperatures ranging from -20°C to 30°C not only improves safety by controlling exothermic risks but also offers substantial energy savings in large-scale manufacturing environments.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of optically active cyclopropane compounds, which serve as vital intermediates for pyrethroids, has relied on copper complexes that often exhibit suboptimal performance under mild conditions. Conventional methods frequently require higher reaction temperatures to achieve acceptable conversion rates, which can lead to thermal decomposition of sensitive diazoacetate reagents and the formation of unwanted by-products. Furthermore, without specific additives, these older catalytic systems often suffer from poor atom economy, leaving significant amounts of unreacted diazo compounds in the final mixture, necessitating complex and costly purification steps. The lack of stereocontrol in some prior art methods also results in lower optical purity, forcing manufacturers to employ additional resolution steps that drastically reduce overall yield and increase waste generation. These inefficiencies create bottlenecks in the supply chain, increasing the cost of goods sold and extending lead times for high-purity agrochemical intermediates.
The Novel Approach
The methodology described in CN1798609A overcomes these historical hurdles by integrating a Lewis acidic compound or a lithium compound into the copper-ligand matrix. This strategic modification activates the catalyst system, enabling it to maintain high turnover frequencies even at reduced temperatures where side reactions are suppressed. By utilizing specific optically active salicylideneaminoalcohols defined by Formula (1), the catalyst creates a highly defined chiral environment around the copper center. 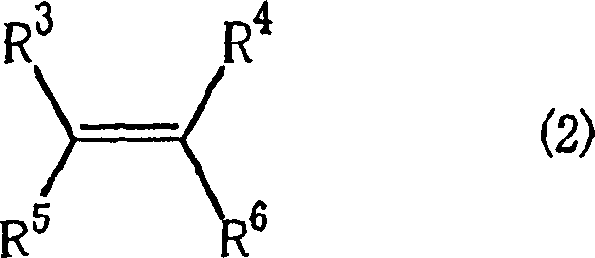 This structural precision ensures that when prochiral olefins, such as the general structure shown above, react with diazoacetates, the formation of the cyclopropane ring occurs with exceptional stereoselectivity. The result is a process that delivers high trans/cis ratios and optical purities exceeding 90% e.e. in optimized examples, directly addressing the purity concerns of regulatory bodies and reducing the burden on downstream processing teams.
This structural precision ensures that when prochiral olefins, such as the general structure shown above, react with diazoacetates, the formation of the cyclopropane ring occurs with exceptional stereoselectivity. The result is a process that delivers high trans/cis ratios and optical purities exceeding 90% e.e. in optimized examples, directly addressing the purity concerns of regulatory bodies and reducing the burden on downstream processing teams.
Mechanistic Insights into Cu-Catalyzed Asymmetric Cyclopropanation
The core of this technological advancement lies in the synergistic interaction between the copper center and the multifunctional ligand system. The optically active salicylideneaminoalcohol acts not merely as a spectator ligand but as a dynamic participant in stabilizing the reactive copper-carbene intermediate formed from the diazoacetate. 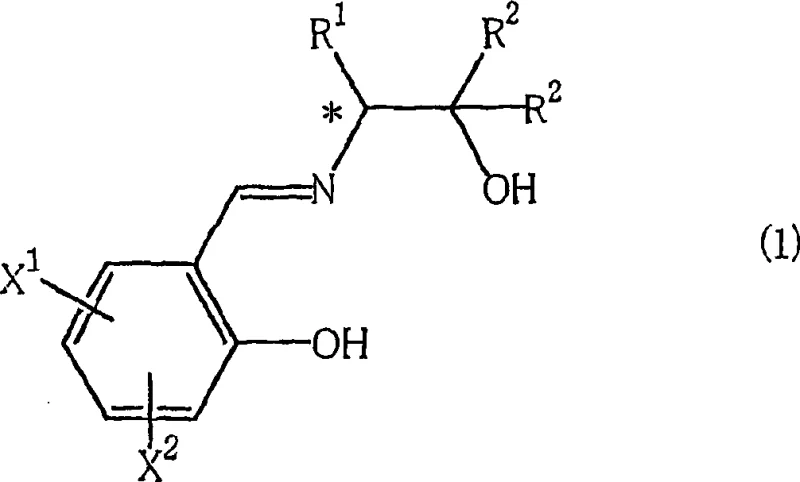 As illustrated in the ligand structure above, the presence of specific substituents (R1, R2) and aromatic modifications (X1, X2) allows for fine-tuning of the steric and electronic properties of the catalyst. The introduction of the Lewis acid component, such as titanium tetraisopropoxide or triethoxyaluminum, is believed to coordinate with the oxygen atoms of the ligand or the incoming substrate, further rigidifying the transition state. This rigidity is crucial for discriminating between the enantiotopic faces of the olefin substrate, thereby driving the high enantioselectivity observed in the experimental data. Understanding this mechanism allows process chemists to select the optimal ligand-metal-additive combination for specific substrate classes, ensuring robust performance across different batches.
As illustrated in the ligand structure above, the presence of specific substituents (R1, R2) and aromatic modifications (X1, X2) allows for fine-tuning of the steric and electronic properties of the catalyst. The introduction of the Lewis acid component, such as titanium tetraisopropoxide or triethoxyaluminum, is believed to coordinate with the oxygen atoms of the ligand or the incoming substrate, further rigidifying the transition state. This rigidity is crucial for discriminating between the enantiotopic faces of the olefin substrate, thereby driving the high enantioselectivity observed in the experimental data. Understanding this mechanism allows process chemists to select the optimal ligand-metal-additive combination for specific substrate classes, ensuring robust performance across different batches.
Furthermore, the impurity profile of the reaction is significantly improved through this mechanistic control. In conventional systems, the decomposition of diazo compounds often leads to dimerization or polymerization by-products that are difficult to remove. However, the enhanced activity of this novel catalyst ensures rapid consumption of the diazo reagent, with residual levels dropping to as low as 0.1% in successful embodiments. This drastic reduction in hazardous residuals simplifies the work-up procedure, often allowing for direct concentration or simple distillation rather than extensive chromatographic purification. For quality control teams, this means a cleaner crude product with a more predictable impurity spectrum, facilitating faster release testing and reducing the risk of batch failures due to out-of-specification impurities. The ability to suppress dimerization while promoting the desired cyclopropanation pathway is a testament to the precise electronic modulation achieved by the ternary catalyst system.
How to Synthesize Optically Active Cyclopropane Compounds Efficiently
Implementing this catalytic technology requires careful attention to the preparation of the active species and the control of reaction parameters. The patent outlines a straightforward protocol where the ligand and copper salt are first mixed in a solvent like ethyl acetate or toluene, followed by the addition of the Lewis acid promoter. This pre-formation step is critical to ensure the complete assembly of the active catalytic complex before the introduction of the reactive diazo species. Once the catalyst is ready, the prochiral olefin is added, and the mixture is cooled to the desired reaction temperature, typically between 0°C and 20°C for optimal balance of rate and selectivity. The detailed standardized synthesis steps see below guide.
- Prepare the catalyst by mixing an optically active salicylideneaminoalcohol ligand with a copper salt and a Lewis acidic additive (e.g., titanium or aluminum compound) in an organic solvent.
- Combine the catalyst solution with a prochiral olefin substrate under an inert atmosphere, optionally adding a reducing agent if using divalent copper.
- Slowly add a diazoacetate reagent at controlled low temperatures (-20°C to 30°C) to facilitate the asymmetric cyclopropanation reaction with high enantioselectivity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this catalyst technology translates into tangible operational efficiencies and risk mitigation. The primary value driver is the significant reduction in raw material costs associated with higher reaction yields. By minimizing the formation of by-products and ensuring near-complete conversion of the expensive diazoacetate reagent, manufacturers can maximize the output from every kilogram of input material. This efficiency gain is compounded by the elimination of complex purification steps; the cleaner reaction profile means less solvent consumption for chromatography and reduced waste disposal costs, contributing to a greener and more cost-effective manufacturing process. Additionally, the ability to run reactions at lower temperatures reduces the energy load on cooling systems, further lowering the utility costs associated with production.
- Cost Reduction in Manufacturing: The implementation of this high-efficiency catalyst system directly impacts the bottom line by improving the overall mass balance of the synthesis. Since the catalyst promotes high conversion rates with minimal residual starting materials, the need for recycling unreacted reagents is diminished, streamlining the production flow. Moreover, the suppression of side reactions means that the yield of the desired optical isomer is maximized, reducing the amount of waste that must be treated or discarded. This qualitative improvement in process efficiency allows for a more competitive pricing structure for the final agrochemical intermediate, providing a strategic advantage in price-sensitive markets without compromising on quality standards.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by processes that are sensitive to minor variations in conditions or raw material quality. This robust catalyst system demonstrates tolerance to a range of operating conditions and utilizes readily available copper salts and common Lewis acids, reducing dependency on exotic or single-source reagents. The simplified work-up procedure also shortens the batch cycle time, allowing facilities to turn over production campaigns more quickly. This agility enables suppliers to respond faster to market demand fluctuations, ensuring that customers receive their orders of high-purity agrochemical intermediates without the delays typically associated with complex chiral syntheses.
- Scalability and Environmental Compliance: Scaling chiral processes from the lab to the plant is notoriously difficult, but the straightforward nature of this catalyst preparation facilitates smooth technology transfer. The use of common organic solvents and the absence of highly toxic heavy metals beyond standard copper levels simplify environmental compliance and waste management. The reduction in hazardous diazo residues also lowers the safety risks associated with large-scale handling of energetic materials. Consequently, manufacturing partners can scale up production volumes with greater confidence, knowing that the process adheres to strict safety and environmental regulations while maintaining the high stereochemical integrity required for biological activity.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this catalytic technology. These answers are derived directly from the experimental data and claims within the patent documentation, providing clarity on reaction conditions and substrate compatibility. Understanding these details is essential for process engineers evaluating the feasibility of integrating this method into existing production lines.
Q: What is the primary advantage of the Lewis acid additive in this catalyst system?
A: The addition of Lewis acidic compounds (such as titanium or aluminum derivatives) significantly enhances catalytic activity, allowing the reaction to proceed efficiently at lower temperatures while minimizing residual diazoacetate.
Q: Can this catalyst system be used for large-scale pyrethroid production?
A: Yes, the patent demonstrates high yields (up to 96%) and excellent optical purity, making it highly suitable for the commercial scale-up of complex agrochemical intermediates like chrysanthemic acid derivatives.
Q: What types of substrates are compatible with this copper catalyst?
A: The system is versatile and accommodates various prochiral olefins, including substituted dienes and alkenes, reacting them with diazoacetates to form diverse optically active cyclopropane structures.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Optically Active Copper Catalyst Supplier
The technological potential demonstrated in CN1798609A underscores the importance of having a manufacturing partner capable of executing complex asymmetric syntheses with precision. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of this advanced catalyst system can be realized at an industrial level. Our facility is equipped with stringent purity specifications and rigorous QC labs designed to handle sensitive chiral intermediates, guaranteeing that every batch meets the exacting standards required for global agrochemical and pharmaceutical applications. We understand that consistency is key, and our process controls are designed to maintain the high enantiomeric excess and yield profiles described in the patent literature.
We invite you to discuss how this innovative catalytic approach can optimize your supply chain for pyrethroid intermediates. Our technical team is prepared to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Please contact our technical procurement team to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise in chiral catalysis can drive value and reliability for your organization.
