Advanced Lithium-Catalyzed Synthesis of Optically Active Bisamido Alcohols for Commercial Scale-Up
Introduction to Patent CN1898193A Technology
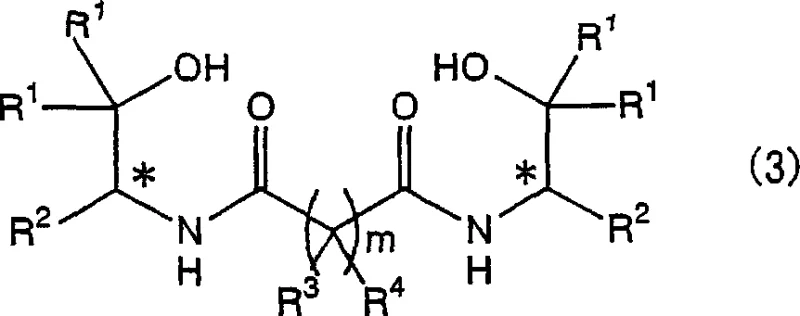
The chemical industry constantly seeks more efficient pathways for synthesizing complex chiral intermediates, particularly those serving as ligands for asymmetric catalysis. Patent CN1898193A introduces a robust methodology for producing optically active bisamido alcohol compounds, represented generally by Formula (3), which are critical precursors for bisoxazoline ligands. Unlike traditional methods that rely on harsh acylating agents, this invention utilizes a direct amidation strategy between optically active amino alcohols and diester compounds in the presence of a lithium compound. This technological shift addresses significant pain points in fine chemical manufacturing, specifically regarding safety, waste generation, and operational simplicity. By leveraging mild lithium catalysis, the process achieves high conversion rates while maintaining the stereochemical integrity of the chiral centers, a non-negotiable requirement for pharmaceutical and agrochemical applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of bisamido alcohols has relied heavily on the use of acyl chlorides, such as malonyl dichloride, reacting with amino alcohols in the presence of a base. This conventional approach presents severe drawbacks for large-scale manufacturing, primarily due to the corrosive nature of acyl chlorides and the generation of stoichiometric amounts of salt byproducts. Furthermore, alternative solvent-free thermal methods involving dimethyl malonate often require excessively high temperatures and lack precise control over reaction kinetics, leading to potential racemization or decomposition of sensitive functional groups. The handling of acid chlorides also necessitates specialized equipment resistant to corrosion and rigorous safety protocols to manage hydrogen chloride gas evolution, thereby inflating capital expenditure and operational complexity for production facilities aiming for commercial scale-up of complex pharmaceutical intermediates.
The Novel Approach
The novel approach detailed in CN1898193A circumvents these issues by employing diester compounds directly as the acyl source, activated by a catalytic amount of a lithium compound. This method effectively replaces the hazardous acyl chloride step with a cleaner transesterification-amidation sequence that proceeds under milder thermal conditions, typically ranging from 20°C to 150°C. The use of lithium alkoxides or halides as promoters facilitates the nucleophilic attack of the amine on the ester carbonyl without the need for strong external bases that might compromise chiral stability. This results in a significantly simplified workup procedure where the primary byproduct is a simple alcohol that can be easily removed by distillation, driving the equilibrium towards the desired bisamido product. Consequently, this route offers a greener, safer, and more economically viable pathway for producing high-purity intermediates essential for the fine chemicals sector.
Mechanistic Insights into Lithium-Catalyzed Direct Amidation
The core mechanism of this transformation involves the activation of the diester carbonyl group by the lithium species, which increases the electrophilicity of the carbon atom, making it more susceptible to nucleophilic attack by the amino group of the chiral amino alcohol. The lithium cation likely coordinates with the carbonyl oxygen, stabilizing the transition state and lowering the activation energy required for the tetrahedral intermediate formation. Following the collapse of this intermediate, the alkoxide leaving group is expelled, forming the amide bond. Crucially, the reaction conditions are sufficiently mild to prevent epimerization at the alpha-carbon adjacent to the nitrogen, ensuring that the optical purity of the starting material, such as the structures shown in Formula (1), is faithfully transferred to the final bisamido product. This preservation of chirality is paramount, as any loss of enantiomeric excess would render the subsequent bisoxazoline ligands ineffective for asymmetric synthesis applications.
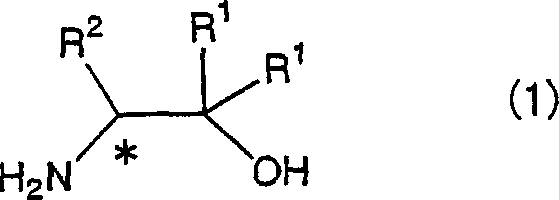
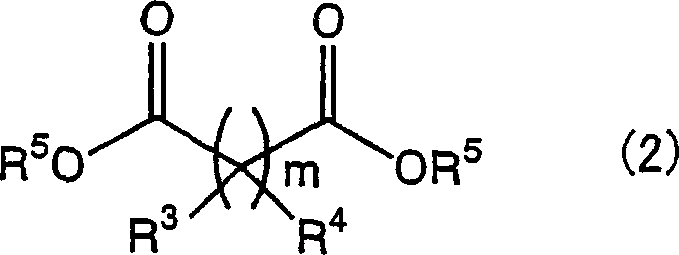
Furthermore, the versatility of the lithium catalyst allows for compatibility with a broad spectrum of substrates, including those with bulky steric hindrance or electron-withdrawing groups. The reaction tolerates various diester structures, from malonates to succinates and oxalates, as depicted in Formula (2), providing chemists with the flexibility to tune the backbone of the final ligand system. The mechanism also benefits from the removal of the generated alcohol byproduct, often achieved by heating the reaction mixture above the boiling point of the alcohol or applying reduced pressure. This Le Chatelier-driven shift ensures near-quantitative conversion without requiring a large excess of reagents, which is a common inefficiency in equilibrium-controlled amidation reactions. Such mechanistic efficiency translates directly into higher space-time yields and reduced raw material consumption in an industrial setting.
How to Synthesize Optically Active Bisamido Alcohol Efficiently
To implement this synthesis effectively, one must carefully select the lithium catalyst and solvent system to match the solubility profile of the specific amino alcohol and diester being used. The patent outlines a generalized protocol where the reactants are mixed in a hydrocarbon or ether solvent, followed by the addition of the lithium promoter, typically lithium methoxide or lithium chloride. The reaction is then heated to reflux, allowing the byproduct alcohol to distill off or remain in solution depending on the specific boiling points involved. Detailed standard operating procedures regarding stoichiometry, mixing orders, and purification steps such as filtration or recrystallization are critical for reproducibility. For a comprehensive guide on executing this reaction with specific substrates like tert-leucinol or phenylglycinol, please refer to the standardized synthesis steps provided below.
- Mix optically active amino alcohol (Formula 1) and diester compound (Formula 2) in a suitable solvent such as n-heptane or toluene.
- Add a catalytic amount of a lithium compound, such as lithium methoxide or lithium chloride, to the reaction mixture.
- Heat the mixture to a temperature between 20°C and 150°C, preferably removing the byproduct alcohol to drive the reaction to completion.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this lithium-catalyzed technology offers substantial strategic benefits over legacy acyl chloride routes. The elimination of acyl chlorides removes a major bottleneck in raw material sourcing, as diesters like dimethyl malonate are commodity chemicals with stable global supply chains and significantly lower price volatility compared to specialized acid chlorides. Additionally, the reduction in hazardous waste generation simplifies environmental compliance and lowers the cost of waste disposal, which is a growing concern for chemical manufacturers facing stricter regulatory scrutiny. The ability to run the reaction in common solvents like n-heptane or toluene further enhances supply chain reliability, as these solvents are readily available in bulk quantities worldwide, reducing lead time for high-purity pharmaceutical intermediates manufacturing.
- Cost Reduction in Manufacturing: The process eliminates the need for expensive and hazardous acyl chloride reagents, replacing them with cost-effective diesters and catalytic amounts of lithium salts. This substitution drastically reduces raw material costs and removes the necessity for expensive corrosion-resistant reactor linings and acid gas scrubbing systems. Furthermore, the high yields reported in the patent examples, often exceeding 90%, minimize material loss and maximize the throughput of existing production assets, leading to significant overall cost savings in fine chemical manufacturing operations.
- Enhanced Supply Chain Reliability: By relying on widely available diester feedstocks and simple lithium catalysts, manufacturers can mitigate risks associated with the supply of specialized acylating agents which may have limited suppliers. The robustness of the reaction conditions allows for flexible scheduling and easier scale-up from pilot to commercial production without extensive re-optimization. This stability ensures consistent delivery schedules for downstream customers who rely on these chiral intermediates for time-sensitive drug development projects, thereby strengthening the supplier-client relationship through dependable service.
- Scalability and Environmental Compliance: The absence of corrosive byproducts like hydrogen chloride gas makes this process inherently safer and easier to scale to multi-ton quantities. The simplified workup, often involving just filtration or solvent removal, reduces the number of unit operations required, lowering energy consumption and water usage. This aligns perfectly with modern green chemistry principles and helps companies meet their sustainability goals by reducing the carbon footprint associated with the production of complex chiral building blocks for the pharmaceutical industry.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These answers are derived directly from the experimental data and claims presented in CN1898193A, providing clarity on reaction scope, safety, and product quality. Understanding these details is essential for R&D teams evaluating this technology for process integration and for procurement officers assessing the viability of sourcing these intermediates from new suppliers.
Q: What are the advantages of using lithium catalysts over acyl chlorides for bisamido alcohol synthesis?
A: Using lithium catalysts with diesters eliminates the need for corrosive and hazardous acyl chlorides (like malonyl dichloride), significantly improving process safety and reducing waste treatment costs associated with acid gas scrubbing.
Q: Can this method preserve the optical purity of the starting amino alcohol?
A: Yes, the patent data confirms that the configuration of the asymmetric center in the product remains identical to that of the starting optically active amino alcohol, ensuring high enantiomeric excess for downstream ligand synthesis.
Q: What solvents are compatible with this lithium-catalyzed amidation process?
A: The process is versatile and supports a wide range of solvents including aromatic hydrocarbons like toluene, aliphatic hydrocarbons like n-heptane, and ethers like THF, allowing for flexible optimization based on solubility and boiling point requirements.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Bisamido Alcohol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality chiral intermediates play in the development of next-generation therapeutics and advanced materials. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the lithium-catalyzed synthesis of bisamido alcohols can be transferred seamlessly from the laboratory to full-scale manufacturing. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch meets the exacting standards required for asymmetric catalyst ligand production, providing our partners with the confidence they need to advance their own R&D pipelines.
We invite you to collaborate with us to leverage this innovative technology for your specific project needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our optimized manufacturing processes can enhance your supply chain efficiency and reduce your overall cost of goods sold.
