Advanced Rhodium-Catalyzed Synthesis of Spiro Dihydrophthalazines for Commercial Pharmaceutical Applications
Advanced Rhodium-Catalyzed Synthesis of Spiro Dihydrophthalazines for Commercial Pharmaceutical Applications
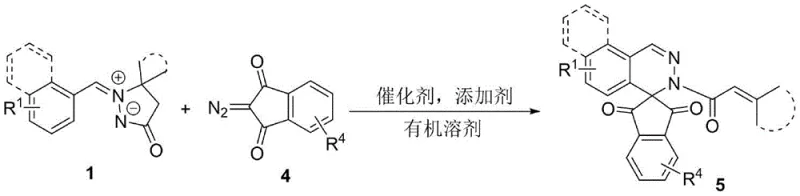
The pharmaceutical industry continuously seeks novel scaffolds that offer enhanced biological activity and improved physicochemical properties, and the recent disclosure in patent CN115197228A presents a significant breakthrough in this domain. This patent details a highly efficient synthetic methodology for constructing pyrazolone[spiro]dihydrophthalazine and 1,3-indanedione[spiro]dihydrophthalazine compounds, which are increasingly recognized as valuable pharmacophores in modern drug discovery programs. The core innovation lies in the utilization of a rhodium-catalyzed tandem reaction between aryl azomethine imines and diazo compounds, enabling the rapid assembly of complex spiro-fused architectures with remarkable precision. By leveraging this advanced catalytic system, manufacturers can access structurally diverse intermediates that were previously difficult to synthesize using conventional stepwise approaches. This technological advancement not only streamlines the synthetic route but also aligns with the growing demand for sustainable and atom-economical processes in the production of high-value fine chemicals. For R&D teams and procurement strategists, understanding the implications of this patent is crucial for securing a competitive edge in the development of next-generation therapeutic agents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the construction of dihydrophthalazine skeletons bearing spiro-cyclic units has been fraught with significant synthetic challenges that hinder both research scalability and commercial viability. Conventional routes often rely on multi-step sequences involving harsh reaction conditions, expensive protecting group strategies, and stoichiometric amounts of toxic reagents that complicate downstream purification. These legacy methods frequently suffer from poor atom economy, generating substantial chemical waste that increases disposal costs and environmental burdens for manufacturing facilities. Furthermore, the rigidity of traditional synthetic pathways often limits the scope of compatible functional groups, restricting the chemical diversity available for medicinal chemistry optimization campaigns. The reliance on unstable intermediates in older protocols can also lead to inconsistent batch-to-batch quality, posing risks to supply chain continuity for critical pharmaceutical ingredients. Consequently, there has been a persistent need for a more robust and direct approach that can overcome these inefficiencies while maintaining high standards of purity and yield.
The Novel Approach
The methodology described in CN115197228A represents a paradigm shift by introducing a direct tandem reaction strategy that efficiently constructs the target spiro-fused frameworks in a single operational step. This novel approach utilizes readily available aryl azomethine imines and diazo precursors, which react seamlessly in the presence of a specialized rhodium catalyst system to form the desired products with high selectivity. The reaction conditions are notably mild, typically proceeding at moderate temperatures between 60-120°C in common organic solvents such as acetonitrile or toluene, which simplifies process engineering requirements. By avoiding the need for pre-functionalized substrates or extreme conditions, this method significantly reduces the operational complexity and safety hazards associated with large-scale chemical manufacturing. The broad substrate scope demonstrated in the patent indicates that a wide variety of electronic and steric environments are tolerated, allowing for the rapid generation of diverse compound libraries for biological screening. This efficiency translates directly into reduced production timelines and lower overall manufacturing costs for complex pharmaceutical intermediates.

Mechanistic Insights into Rhodium-Catalyzed Tandem Cyclization
The success of this synthetic transformation is underpinned by a sophisticated catalytic cycle involving rhodium(III) species that facilitate the activation and coupling of the reactants with high fidelity. The mechanism likely initiates with the coordination of the rhodium catalyst to the diazo compound, generating a reactive metal-carbenoid intermediate that is poised for subsequent bond-forming events. This electrophilic species then engages with the aryl azomethine imine through a concerted or stepwise cycloaddition pathway, establishing the critical carbon-carbon and carbon-nitrogen bonds that define the spiro center. The precise control exerted by the pentamethylcyclopentadienyl ligand environment on the rhodium center ensures that the reaction proceeds with excellent regioselectivity, minimizing the formation of unwanted isomeric byproducts. Understanding this mechanistic nuance is vital for process chemists aiming to optimize reaction parameters for maximum throughput and minimal impurity profiles in a commercial setting. The ability to tune the electronic properties of the catalyst through ligand modification further enhances the versatility of this platform for adapting to specific substrate challenges.
Impurity control is a paramount concern in the synthesis of pharmaceutical intermediates, and this catalytic system offers inherent advantages in managing side reactions that typically plague spiro-cycle formation. The high chemoselectivity of the rhodium catalyst ensures that sensitive functional groups present on the starting materials remain intact throughout the transformation, preserving the integrity of the molecular architecture. By operating under mild thermal conditions, the risk of thermal decomposition or polymerization of reactive intermediates is significantly mitigated, leading to cleaner crude reaction mixtures. This reduction in side-product formation simplifies the purification process, often allowing for straightforward crystallization or chromatography to achieve the stringent purity specifications required for GMP manufacturing. Furthermore, the use of silver additives helps to scavenge halides or stabilize the active catalytic species, contributing to the overall robustness and reproducibility of the process. For quality assurance teams, this level of control provides confidence in the consistency of the final product across different production batches.
How to Synthesize Pyrazolone Spiro Dihydrophthalazine Efficiently
Implementing this synthesis in a practical setting requires careful attention to the preparation of reagents and the maintenance of optimal reaction conditions to ensure high yields and reproducibility. The process begins with the precise weighing of aryl azomethine imine and diazo pyrazolone precursors, which are then dissolved in a dry organic solvent such as acetonitrile to create a homogeneous reaction mixture. Following this, the rhodium catalyst and silver additive are introduced, and the system is heated to the specified temperature range while monitoring the progress via thin-layer chromatography or HPLC analysis. Detailed standardized synthesis steps see the guide below.
- Prepare the reaction mixture by combining aryl azomethine imine and diazo pyrazolone compounds in an organic solvent such as acetonitrile.
- Add the rhodium catalyst system, specifically Cp*Rh(MeCN)3(SbF6)2, along with a silver acetate additive to facilitate the tandem reaction.
- Heat the reaction mixture to temperatures between 60-120°C under air or inert atmosphere, followed by purification via silica gel chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, the adoption of this rhodium-catalyzed technology offers substantial benefits that extend beyond mere technical feasibility to impact the bottom line of chemical manufacturing operations. The streamlined nature of the tandem reaction eliminates several unit operations typically required in multi-step syntheses, thereby reducing labor costs and equipment occupancy time in production facilities. By utilizing commercially available starting materials and common solvents, supply chain managers can mitigate the risks associated with sourcing exotic or regulated reagents that often cause bottlenecks in global procurement networks. The high atom economy of the process means that a greater proportion of raw material mass is converted into the final product, inherently lowering the cost of goods sold and minimizing waste disposal fees. These factors collectively contribute to a more resilient and cost-effective supply chain capable of meeting the demanding schedules of pharmaceutical development pipelines.
- Cost Reduction in Manufacturing: The elimination of multiple isolation and purification steps inherent in traditional routes leads to significant operational savings and reduced solvent consumption volumes. By avoiding the use of expensive protecting groups and harsh reagents, the overall material cost per kilogram of the final intermediate is drastically optimized for commercial production. The mild reaction conditions also translate to lower energy requirements for heating and cooling, further enhancing the economic viability of the process on an industrial scale. Additionally, the high selectivity reduces the burden on quality control laboratories, allowing for faster release times and reduced analytical overhead costs.
- Enhanced Supply Chain Reliability: The reliance on stable and readily accessible raw materials ensures that production schedules are not disrupted by the volatility of specialty chemical markets. The robustness of the catalytic system allows for flexibility in manufacturing locations, enabling companies to diversify their supplier base and reduce geopolitical risks associated with single-source dependencies. Furthermore, the scalability of the reaction from gram to kilogram scales has been demonstrated, providing confidence that supply can be ramped up quickly to meet sudden increases in demand from clinical trial phases. This reliability is critical for maintaining continuous manufacturing flows and avoiding costly delays in drug development timelines.
- Scalability and Environmental Compliance: The process generates minimal hazardous waste due to its high atom efficiency, aligning with increasingly stringent environmental regulations and corporate sustainability goals. The use of less toxic solvents and the avoidance of heavy metal contamination in the final product simplify the regulatory filing process and reduce the environmental footprint of the manufacturing site. Scaling this reaction to multi-ton quantities is feasible without requiring specialized high-pressure equipment, making it accessible to a wider range of contract manufacturing organizations. This ease of scale-up ensures that the technology remains viable from early-stage development through to commercial launch without the need for disruptive process redesigns.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology in industrial settings. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation to provide accurate guidance for decision-makers. Understanding these details helps stakeholders evaluate the feasibility of integrating this method into their existing manufacturing portfolios. Comprehensive responses cover aspects ranging from catalyst recovery to substrate compatibility.
Q: What are the primary advantages of this rhodium-catalyzed method over conventional synthesis?
A: This method offers high atom economy and operates under mild conditions, eliminating the need for harsh reagents often required in traditional multi-step constructions of spiro scaffolds.
Q: Is this synthesis scalable for industrial production of pharmaceutical intermediates?
A: Yes, the use of readily available raw materials and robust reaction conditions supports commercial scale-up, ensuring consistent supply chain reliability for complex heterocycles.
Q: What is the functional group tolerance of this catalytic system?
A: The protocol demonstrates excellent tolerance for various substituents including halogens, alkyl groups, and electron-withdrawing groups, allowing for diverse derivative synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyrazolone Spiro Dihydrophthalazine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this rhodium-catalyzed synthesis route for producing high-value pharmaceutical intermediates with exceptional efficiency. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from laboratory concept to market reality. Our state-of-the-art facilities are equipped to handle sensitive organometallic chemistry with stringent purity specifications and rigorous QC labs to guarantee product quality. We are committed to delivering reliable supply solutions that empower your drug discovery teams to focus on innovation while we manage the complexities of chemical manufacturing.
We invite you to collaborate with us to leverage this advanced technology for your specific pipeline needs and achieve a competitive advantage in the marketplace. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume requirements and timeline constraints. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our capabilities align with your strategic goals. Let us help you optimize your supply chain and accelerate the development of your next-generation therapeutics through expert chemical synthesis.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
