Advanced Photochemical Synthesis of 25-Hydroxyvitamin D3 for Commercial Scale-Up
The global demand for bioactive forms of Vitamin D has surged as research confirms their superior efficacy over standard cholecalciferol in both human nutrition and animal husbandry. Patent CN103044301A introduces a groundbreaking photochemical synthesis method for 25-Hydroxyvitamin D3, addressing the critical bottleneck of producing this high-value metabolite efficiently. Unlike traditional methods that rely on complex multi-step organic synthesis, this innovation leverages a direct photochemical ring-opening of 25-hydroxy-7-dehydrocholesterol followed by thermal isomerization. This approach not only simplifies the production workflow but also ensures a final product purity exceeding 99% without the need for costly column chromatography. Understanding the biological imperative behind this synthesis is crucial for stakeholders evaluating its market potential. As illustrated in the metabolic pathway, Vitamin D3 must undergo hydroxylation in the liver and kidney to become physiologically active, making the direct supply of 25-hydroxylated forms a significant value-add for the supply chain. 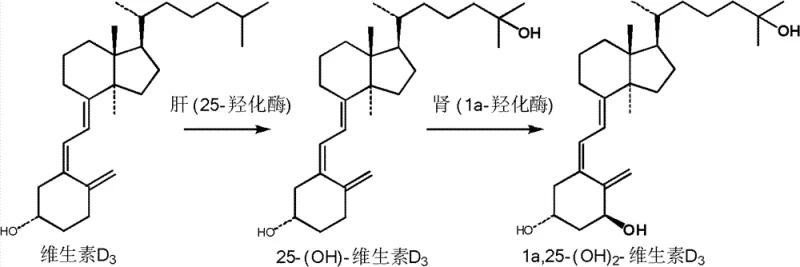
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of 25-Hydroxyvitamin D3 has been plagued by inefficient synthetic routes that hinder large-scale commercialization. Total synthesis methods involve an excessive number of reaction steps, resulting in low overall yields and significant difficulties in separating the target molecule from complex reaction mixtures. Semi-synthetic approaches, which often attempt to modify the side chain of existing Vitamin D structures, typically require breaking the steroid skeleton and rebuilding it, a process that generates numerous by-products and drives up raw material costs substantially. Furthermore, conventional thermochemical reactions often suffer from poor selectivity, yielding less than 10% of the desired product while creating separation challenges that necessitate expensive purification technologies. These inefficiencies create a barrier to entry for many manufacturers, limiting the availability of reliable active vitamin D3 supplier options in the global market and keeping prices prohibitively high for widespread use in feed additives.
The Novel Approach
The patented photochemical method represents a paradigm shift by utilizing a one-step singlet bond breaking mechanism under light irradiation to directly access the target structure. By dissolving 25-hydroxy-7-dehydrocholesterol in a specifically tuned mixed solvent system containing both polar and non-polar components, the reaction kinetics are optimized to favor the formation of the pre-vitamin intermediate. The process incorporates a critical recovery step where unreacted starting material is separated via freezing and recrystallization, allowing it to be recycled back into the reactor. This closed-loop material flow drastically improves raw material utilization efficiency and reduces waste generation. Additionally, the subsequent thermal isomerization step is straightforward and robust, enabling the production of either high-purity crystals or stable oil formulations depending on market requirements, thereby offering flexibility for cost reduction in nutritional ingredients manufacturing.
Mechanistic Insights into Photochemical Ring-Opening and Isomerization
The core of this technology lies in the precise control of photochemical kinetics to prevent the degradation of the sensitive secosteroid structure. When 25-hydroxy-7-dehydrocholesterol is exposed to UV light from a high-pressure mercury lamp, it undergoes a conrotatory electrocyclic ring-opening to form 25-hydroxy-previtamin D3. However, this intermediate is unstable and can easily convert into unwanted by-products such as 25-hydroxy-lumisterol and 25-hydroxy-tachysterol if the irradiation continues unchecked. The patent specifies maintaining the conversion rate of the starting material below 40%, ideally between 30% and 39%, to maximize the ratio of the desired pre-vitamin while minimizing these secondary photoproducts. This kinetic control is achieved by monitoring the reaction via HPLC and adjusting the illumination time, typically around 15 to 30 minutes, ensuring the reaction stops at the optimal point before equilibrium shifts towards degradation products.
Following the photochemical step, the separation mechanism relies on the distinct solubility profiles of the starting material versus the products in polar solvents at low temperatures. By cooling the reaction mixture to between -10°C and -20°C, the unreacted 25-hydroxy-7-dehydrocholesterol crystallizes out due to its significantly reduced solubility, while the product remains in the solution. This physical separation is far more scalable and cost-effective than chromatographic methods. The remaining filtrate, rich in the pre-vitamin, is then subjected to thermal isomerization at 60°C to 70°C under an inert atmosphere. This [1,7]-sigmatropic hydrogen shift converts the pre-vitamin into the stable 25-Hydroxyvitamin D3. The use of antioxidants like 2,6-di-tert-butyl-p-methoxyphenol throughout the process prevents oxidative degradation, ensuring the stringent purity specifications required for high-purity vitamin supplements are met consistently.
How to Synthesize 25-Hydroxyvitamin D3 Efficiently
Implementing this synthesis route requires careful attention to solvent selection and environmental controls to ensure reproducibility and safety. The process begins with preparing a reaction solution where the concentration of the starting steroid is maintained between 3% and 5% by weight in a mixed solvent system, such as ethanol and n-pentane. It is critical to operate under an inert gas blanket, typically nitrogen or argon, to exclude oxygen which could compromise the stability of the conjugated triene system. The detailed standardized synthesis steps, including specific solvent ratios, temperature gradients, and filtration protocols, are outlined in the technical guide below for process engineers to follow.
- Dissolve 25-hydroxy-7-dehydrocholesterol in a mixed solvent system of polar and non-polar solvents with an antioxidant.
- Perform photochemical reaction under inert gas protection using a high-pressure mercury lamp, controlling conversion below 40%.
- Recover unreacted starting material via freezing filtration, then thermally isomerize the filtrate to obtain crystals or oil.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, this patented process offers tangible benefits that translate directly into improved margins and operational resilience. By eliminating the need for column chromatography, the method removes one of the most expensive and time-consuming unit operations from the production line. This simplification not only reduces capital expenditure on purification equipment but also significantly lowers the consumption of silica gel and solvents associated with traditional purification. The ability to recover and recycle unreacted starting material further enhances the economic viability of the process, ensuring that raw material costs are minimized even when dealing with high-value steroid precursors. These efficiencies collectively contribute to substantial cost savings in the final product pricing.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts and complex chromatographic purification steps removes significant cost centers from the production budget. Traditional methods often require expensive heavy metal removal processes to meet regulatory standards, whereas this photochemical route relies on physical separation techniques that are inherently cleaner. By recycling the unreacted 25-hydroxy-7-dehydrocholesterol, the effective consumption of the most expensive raw material is drastically reduced. This logical deduction of cost drivers suggests a much lower cost of goods sold (COGS) compared to semi-synthetic alternatives, allowing for more competitive pricing strategies in the bulk chemical market.
- Enhanced Supply Chain Reliability: The reliance on common industrial solvents like ethanol, hexane, and petroleum ether ensures that the supply chain is not vulnerable to shortages of exotic reagents. The process conditions, such as room temperature for the photochemical step and moderate heating for isomerization, are easy to maintain in standard chemical manufacturing facilities without requiring specialized high-pressure or cryogenic equipment. This accessibility means that production can be scaled up rapidly to meet sudden spikes in demand for animal feed additives or pharmaceutical intermediates. Reducing lead time for high-purity feed additives becomes feasible because the batch cycle time is shortened by the simplified workup procedures.
- Scalability and Environmental Compliance: The process is designed with industrial scalability in mind, utilizing continuous or large-batch photochemical reactors that are well-understood in the fine chemical industry. The reduction in solvent usage and the absence of toxic heavy metal waste streams simplify wastewater treatment and environmental compliance efforts. The ability to produce both crystalline and oil forms allows manufacturers to tailor the product to specific downstream applications, whether it be for solid dosage forms in human supplements or liquid premixes in agriculture. This versatility supports the commercial scale-up of complex steroid derivatives without the typical environmental liabilities associated with older synthetic routes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this photochemical synthesis technology. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on process capabilities and product quality. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: How is high purity achieved without column chromatography?
A: The process utilizes precise solubility differences at low temperatures to crystallize and separate unreacted starting materials, eliminating the need for expensive chromatographic purification while maintaining purity above 99%.
Q: What is the advantage of controlling conversion rates below 40%?
A: Limiting the photochemical conversion to below 40% prevents the formation of secondary by-products like tachysterol, ensuring a higher ratio of the desired pre-vitamin intermediate and simplifying downstream purification.
Q: Can this method be scaled for industrial feed additive production?
A: Yes, the method uses common solvents and standard photochemical reactors, avoiding complex total synthesis steps, which makes it highly suitable for large-scale manufacturing of cost-effective feed additives.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 25-Hydroxyvitamin D3 Supplier
The photochemical synthesis of 25-Hydroxyvitamin D3 represents a significant technological advancement that aligns perfectly with the capabilities of modern CDMO partners. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that this innovative laboratory method can be successfully translated into robust industrial manufacturing. Our facility is equipped with state-of-the-art photochemical reactors and rigorous QC labs capable of verifying the stringent purity specifications required for this sensitive molecule. We understand the nuances of handling steroid intermediates and maintaining the inert conditions necessary to prevent degradation, guaranteeing a consistent supply of high-quality material for your formulation needs.
We invite global partners to collaborate with us to optimize their supply chains for active vitamin ingredients. By leveraging this efficient synthesis route, we can offer a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage potential clients to contact our technical procurement team to request specific COA data and route feasibility assessments. Whether you are developing a new premium animal feed additive or a high-potency human supplement, our expertise ensures that you receive a reliable 25-Hydroxyvitamin D3 supplier partnership that drives value and innovation in your product lines.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
