Optimizing Vismodegib Production: A Technical Analysis of Novel Condensation Routes
Introduction to Advanced Vismodegib Manufacturing Technologies
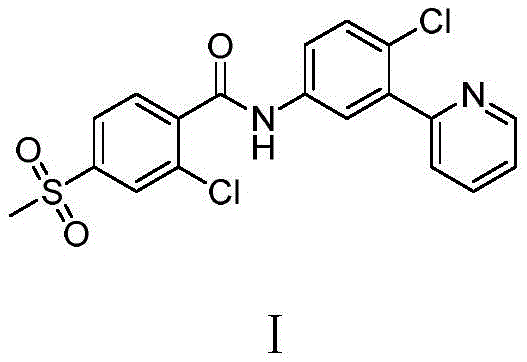
The pharmaceutical landscape for oncology treatments continues to evolve, with Vismodegib standing out as a critical Hedgehog pathway inhibitor for treating basal cell carcinoma. As global demand for this active pharmaceutical ingredient (API) surges, the efficiency of its supply chain becomes paramount. Patent CN111056994B, published in August 2021, introduces a transformative preparation method that addresses longstanding bottlenecks in vismodegib synthesis. This technical disclosure outlines a streamlined three-step process starting from 2-chloro-5-nitroacetophenone, leveraging a Michael addition strategy to construct the pivotal pyridine ring. Unlike legacy methods reliant on precious metal catalysis, this novel approach emphasizes cost-efficiency, environmental sustainability, and operational safety. For R&D directors and procurement strategists, understanding this shift from cross-coupling to condensation chemistry is essential for securing a reliable vismodegib intermediate supplier capable of meeting rigorous purity standards while optimizing manufacturing expenditures.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
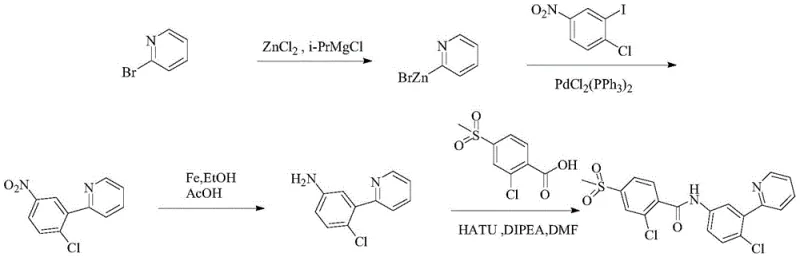
Historically, the industrial synthesis of vismodegib has been plagued by complex multi-step sequences involving expensive reagents and hazardous waste streams. Traditional routes, such as those described in US7888364, rely heavily on Negishi coupling reactions. These methods necessitate the in-situ preparation of 2-pyridylzinc bromide from 2-bromopyridine and grignard reagents, a process fraught with safety risks due to the pyrophoric nature of organozinc compounds. Furthermore, the stability of these intermediates is often poor, leading to inconsistent yields and complicating inventory management. Other prior art, such as CN103910671A, utilizes palladium-catalyzed coupling with pyridine-N-oxides, which not only suffers from low yields (often around 60%) but also generates significant phosphorus-containing wastewater during the deoxygenation step using phosphorus trichloride. These environmental liabilities and the high cost of palladium catalysts create substantial barriers to cost reduction in pharmaceutical intermediates manufacturing.
The Novel Approach
In stark contrast, the methodology disclosed in CN111056994B circumvents these issues by employing a robust Michael addition followed by cyclization. This route initiates with the reaction of 2-chloro-5-nitroacetophenone and acrolein, two commodity chemicals that are significantly cheaper and more stable than organometallic reagents. The process eliminates the need for transition metal catalysts like palladium or nickel in the ring-forming step, thereby removing the risk of heavy metal contamination in the final API. The total yield of this novel pathway is reported to reach approximately 85%, a substantial improvement over the 37.6% to 61.1% yields observed in older synthetic schemes. By simplifying the workflow to just three main chemical transformations, this approach drastically reduces the operational complexity and solvent consumption, positioning it as a superior choice for the commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Michael Addition and Oxidative Cyclization
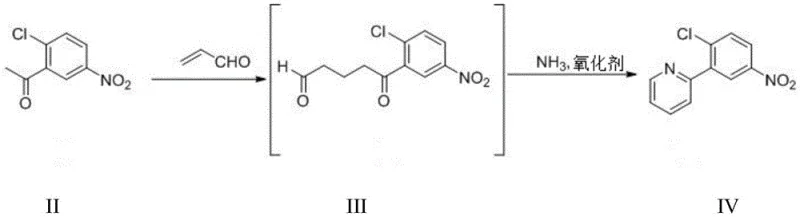
The core innovation of this patent lies in the mechanistic elegance of constructing the 2-arylpyridine motif. The reaction begins with a base-catalyzed Michael addition where the enolate of 2-chloro-5-nitroacetophenone attacks the beta-carbon of acrolein. This forms a 1,5-dicarbonyl intermediate (Compound III), which possesses high stability compared to halogenated analogues used in other methods. Subsequent treatment with ammonia facilitates a condensation reaction to form a dihydropyridine ring. The final aromatization is achieved through an oxidation step using agents like hydrogen peroxide or tert-butyl peroxide. This oxidative dehydrogenation is highly selective, ensuring that the nitro group on the phenyl ring remains intact while the pyridine ring is fully aromatized. The use of non-halogenated acrolein is particularly critical; it avoids the formation of bromine-containing by-products that are difficult to remove and potentially genotoxic, thus enhancing the overall safety profile of the manufacturing process.
From an impurity control perspective, this mechanism offers distinct advantages. The high reaction selectivity minimizes the formation of regioisomers and oligomeric side products often seen in metal-catalyzed couplings. The intermediate compounds exhibit appropriate stability, allowing for potential isolation or telescoping without significant degradation. Furthermore, the reduction of the nitro group to the amine can be performed using catalytic hydrogenation with Pd/C or Raney Nickel, or alternatively with iron powder. The patent highlights that controlling the temperature and pressure during hydrogenation (e.g., 0.2-1.0 MPa H2) ensures minimal formation of hydroxylamine intermediates, which are known genotoxic impurities. This rigorous control over the reduction step ensures that the final amine precursor meets the stringent purity specifications required for oncology drug substances.
How to Synthesize Vismodegib Efficiently
The synthesis protocol detailed in the patent provides a clear roadmap for laboratory and pilot-scale production. The process is divided into three distinct stages: the formation of the nitro-pyridine intermediate, the reduction to the amine, and the final amidation. Each step is optimized for specific solvents and temperatures to maximize yield and purity. For instance, the initial addition reaction is preferably conducted in solvents like 1,2-dichloroethane or tetrahydrofuran at moderate temperatures (20 to 40°C), ensuring safe handling of acrolein. The subsequent amidation utilizes standard acid-binding agents like potassium carbonate or triethylamine in dichloromethane, a widely available solvent system.
- Perform Michael addition of 2-chloro-5-nitroacetophenone and acrolein in solvent A with an organic base catalyst, followed by reaction with ammonia and an oxidant to form 2-(2-chloro-5-nitrophenyl)pyridine.
- Reduce the nitro group of the pyridine intermediate using hydrogen with Pd/C or Raney Nickel, or alternatively using iron/zinc powder in acidic conditions, to obtain the amine derivative.
- Conduct an amidation reaction between the resulting amine and 2-chloro-4-methylsulfonylbenzoyl chloride in the presence of an acid-binding agent to yield Vismodegib.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthesis route translates into tangible strategic benefits. The primary driver of value is the drastic simplification of the raw material portfolio. By replacing expensive, specialized reagents like 2-pyridylzinc bromide or 2-phenylpyridine with bulk commodities like acrolein and nitroacetophenone, manufacturers can achieve significant cost savings in raw material acquisition. This shift also mitigates supply risk, as commodity chemicals are less susceptible to market volatility compared to custom-synthesized organometallics. Additionally, the elimination of phosphorus trichloride and heavy metal catalysts simplifies waste treatment protocols, reducing the environmental compliance burden and associated disposal costs.
- Cost Reduction in Manufacturing: The economic impact of this process is profound due to the removal of precious metal catalysts and the reduction in step count. Traditional routes often require expensive palladium ligands and rigorous purification steps to remove trace metals, which adds considerable time and expense. By utilizing a metal-free cyclization strategy, the new method eliminates the need for expensive scavengers and chromatography, leading to a leaner cost structure. The high total yield further amplifies these savings by maximizing the output per unit of input material, effectively lowering the cost of goods sold (COGS) for the final API.
- Enhanced Supply Chain Reliability: Supply continuity is critical for life-saving medications. This synthesis route enhances reliability by relying on a robust chemical foundation that is less prone to failure. The intermediates generated are stable and do not require cryogenic storage or inert atmosphere handling to the same extent as organozinc species. This stability allows for more flexible logistics and inventory management, reducing the lead time for high-purity pharmaceutical intermediates. Manufacturers can maintain consistent production schedules without the frequent interruptions caused by reagent instability or catalyst deactivation.
- Scalability and Environmental Compliance: Scaling chemical processes often reveals hidden inefficiencies, but this route is designed with green chemistry principles in mind. The avoidance of halogenated acroleins prevents the generation of bromine-containing wastewater, a significant environmental hazard. Furthermore, the use of catalytic hydrogenation or iron reduction for the nitro group offers scalable options that generate benign by-products like water or iron oxides. This alignment with environmental regulations facilitates smoother regulatory approvals and long-term operational sustainability, making it an ideal candidate for multi-ton commercial production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this vismodegib synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on yield optimization, impurity profiles, and process safety. Understanding these nuances is vital for technical teams evaluating the feasibility of technology transfer.
Q: What are the primary advantages of the Michael addition route for Vismodegib?
A: The Michael addition route utilizes cheap and readily available raw materials like 2-chloro-5-nitroacetophenone and acrolein, avoiding expensive organozinc reagents or palladium catalysts used in traditional cross-coupling methods. It offers a shorter process flow with higher total yields reaching up to 85% and generates less hazardous waste.
Q: How does this method address genotoxic impurity concerns?
A: Unlike routes utilizing halogenated acroleins which can lead to substitution side reactions and halogenated impurities, this method employs non-halogenated acrolein. The high stability of the Michael addition intermediate and the specific oxidation conditions minimize the formation of genotoxic by-products, ensuring high product purity.
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, the process is designed for industrial scalability. It operates under mild reaction conditions (0-60°C for key steps), uses common solvents like dichloroethane or THF, and avoids the handling of unstable organometallic reagents, making it safer and more robust for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Vismodegib Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and compliant manufacturing pathways for oncology therapeutics. Our technical team has extensively analyzed the route disclosed in CN111056994B and validated its potential for industrial application. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to commercial reactor is seamless. Our facilities are equipped with rigorous QC labs and advanced analytical instrumentation to guarantee stringent purity specifications, particularly for genotoxic impurities which are a major concern in vismodegib synthesis.
We invite global partners to collaborate with us to leverage this advanced manufacturing technology. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us to request specific COA data and route feasibility assessments, ensuring that your supply chain is built on a foundation of scientific excellence and commercial reliability.
