Advanced Fragment Condensation Strategy for High-Purity Semaglutide Manufacturing
The pharmaceutical landscape for type II diabetes treatment has been revolutionized by Glucagon-like peptide-1 (GLP-1) analogues, with Semaglutide standing out as a premier long-acting therapeutic agent. As detailed in the recent patent disclosure CN113135991A, the demand for efficient, high-purity synthesis routes for this complex polypeptide is critical for meeting global supply chain requirements. Semaglutide, chemically defined by the CAS number 910463-68-2, presents significant synthetic challenges due to its length (31 amino acids) and the presence of hydrophobic residues that promote aggregation. The patent introduces a sophisticated hybrid strategy combining solid-phase peptide synthesis (SPPS) with fragment condensation, specifically targeting the notorious issues of racemization and difficult purification that plague conventional linear synthesis methods. This technical breakthrough offers a viable pathway for reliable pharmaceutical intermediates supplier networks to enhance their production capabilities while maintaining stringent quality standards required for regulatory approval.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to synthesizing long peptide sequences like Semaglutide typically rely on stepwise amino acid condensation directly on the resin. However, as the peptide chain elongates, particularly with hydrophobic sequences, the resin matrix tends to shrink and the peptide chains aggregate, creating a diffusion barrier that severely hampers reagent access to the reactive sites. This phenomenon leads to incomplete couplings and the formation of deletion sequences that are structurally very similar to the target product, making them extremely difficult to separate. Furthermore, the N-terminal Histidine residue is highly susceptible to base-catalyzed racemization during activation, leading to the formation of the [D-His2] impurity, which possesses different biological activity and must be strictly controlled. Conventional methods also struggle with the formation of +Gly impurities and intramolecular cyclization side reactions, which drastically increase the complexity of the crude product profile and necessitate multiple, low-yield purification steps that drive up manufacturing costs and extend lead times.
The Novel Approach
To overcome these inherent limitations, the disclosed method employs a strategic fragmentation of the synthesis process, isolating the most problematic regions of the peptide sequence for independent optimization. Instead of coupling amino acids one by one from the C-terminus to the N-terminus without interruption, the process utilizes a pre-synthesized, fully protected tetrapeptide fragment corresponding to positions 1-4 (S1-S4). This fragment, specifically designed with N-alkylated glycine protection, acts as a stable building block that can be coupled efficiently to the growing chain. Additionally, the complex side-chain modification at Lysine-20 is prepared as a distinct fragment, Fmoc-Lys(AEEA-AEEA-γ-Glu(OtBu)-octadecanedioic)-OH, ensuring that the bulky hydrophobic tail is introduced with high fidelity. 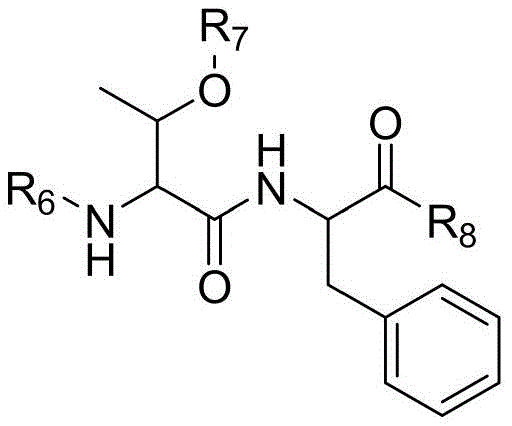
Mechanistic Insights into N-Alkylated Glycine Stabilization
The core innovation of this synthesis route lies in the chemical design of the S1-S4 fragment, which incorporates a specific N-protecting group on the Glycine residue adjacent to the Histidine. In standard peptide coupling, the activation of Histidine can lead to the formation of a 5(4H)-oxazolone intermediate, which allows for the abstraction of the alpha-proton and subsequent racemization to the D-isomer. By introducing an N-alkyl group (such as Hmb, Dmb, or Tmob) onto the nitrogen of the preceding Glycine residue, the formation of this oxazolone ring is sterically hindered and electronically disfavored. This mechanistic intervention effectively "locks" the Histidine configuration, preventing the epimerization that typically occurs during the coupling of the N-terminal residues. 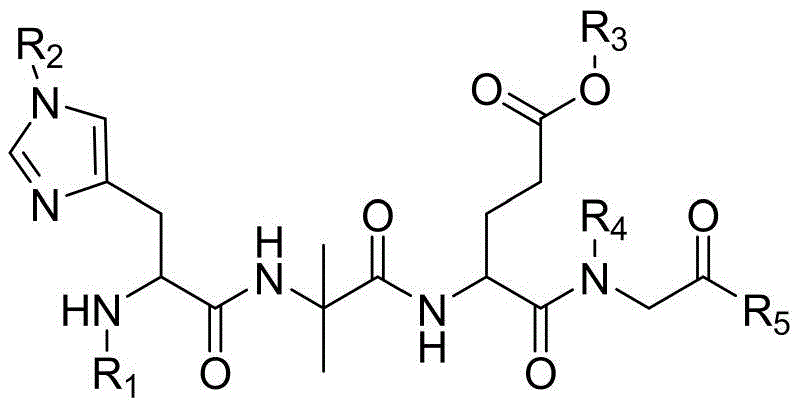
Beyond racemization control, this fragment strategy also addresses the issue of resin swelling and solvation. By coupling a pre-formed tetrapeptide rather than four individual amino acids, the number of repetitive swelling and shrinking cycles the resin undergoes is reduced. This maintains the porosity of the polymer matrix, ensuring that subsequent coupling reagents can penetrate the resin beads effectively. The result is a crude peptide profile with significantly fewer deletion sequences and truncation byproducts. The reduction in closely related impurities means that the final purification via preparative HPLC requires less solvent consumption and fewer runs, directly translating to higher throughput and reduced environmental waste in the manufacturing facility.
How to Synthesize Semaglutide Efficiently
The implementation of this fragment-based strategy requires precise control over reaction conditions and protecting group orthogonality. The process begins with the preparation of the key S1-S4 fragment on a suitable resin, followed by cleavage to yield the fully protected tetrapeptide acid. This fragment is then activated and coupled to the resin-bound peptide chain corresponding to positions 5-31. The detailed operational parameters, including specific solvent ratios, activation times, and temperature controls necessary to maximize yield and minimize side reactions, are critical for successful replication.
- Prepare the key S1-S4 protected tetrapeptide fragment (e.g., Boc-His(Trt)-Aib-Glu(OtBu)-N(Hmb)-Gly-OH) using solid-phase synthesis with N-alkylated glycine to prevent racemization.
- Synthesize the complex Lys20 side-chain fragment (Fmoc-Lys(AEEA-AEEA-gamma-Glu(OtBu)-octadecanedioic)-OH) separately in solution or on resin.
- Assemble the full peptide sequence on resin using stepwise coupling for the main chain, incorporating the pre-formed S1-S4 fragment and Lys20 fragment, followed by cleavage and purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this fragment condensation methodology offers substantial strategic benefits beyond mere technical feasibility. The primary advantage lies in the drastic simplification of the downstream processing workflow. By significantly reducing the load of difficult-to-separate impurities like D-His racemates and +Gly byproducts in the crude material, the burden on the purification department is alleviated. This reduction in purification complexity allows for larger batch sizes to be processed per cycle without overwhelming the chromatography columns, effectively increasing the production capacity of existing facilities without the need for capital-intensive equipment upgrades.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts or complex scavenging steps often required in alternative synthesis routes leads to direct material cost savings. Furthermore, the improved crude purity reduces the consumption of high-grade organic solvents and stationary phases during the final polishing steps. Since solvent recovery and disposal constitute a major portion of operating expenses in peptide manufacturing, minimizing the volume of waste solvent generated through higher reaction efficiency results in significant operational expenditure (OpEx) reductions. The ability to achieve higher yields per gram of resin also lowers the cost of goods sold (COGS) for the final active pharmaceutical ingredient.
- Enhanced Supply Chain Reliability: The reliance on robust, pre-validated peptide fragments mitigates the risk of batch failures that are common in long linear syntheses. When a synthesis fails at step 30 of a 31-step linear process, the entire batch is lost; however, in a fragment-based approach, the failure of a single fragment synthesis does not necessarily compromise the entire campaign, as stockpiles of validated fragments can be utilized. This modularity enhances supply continuity, ensuring that delivery schedules for critical diabetes medications are met consistently. It also allows for parallel processing, where different fragments are synthesized simultaneously, drastically shortening the overall manufacturing lead time.
- Scalability and Environmental Compliance: The process is designed with industrial scalability in mind, utilizing standard solid-phase reactors and common organic solvents that are easily sourced and managed. The reduction in side reactions means fewer hazardous byproducts are generated, simplifying wastewater treatment and aligning with increasingly stringent environmental regulations. The method supports the commercial scale-up of complex pharmaceutical intermediates from pilot kilogram scales to multi-ton annual production volumes, providing a stable foundation for long-term supply agreements with global pharmaceutical partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. Understanding these details is essential for R&D teams evaluating the feasibility of technology transfer and for procurement specialists assessing the long-term viability of the supply source.
Q: How does this method reduce D-His racemic impurities in Semaglutide?
A: The method utilizes a pre-formed S1-S4 fragment containing an N-alkylated glycine residue (using protecting groups like Hmb, Dmb, or Tmob). This N-alkylation sterically hinders the formation of oxazolone intermediates during the coupling of Histidine, which is the primary pathway for racemization, thereby significantly suppressing the generation of the [D-His2]-impurity.
Q: What are the advantages of using fragment condensation over linear SPPS for long peptides?
A: Linear solid-phase peptide synthesis (SPPS) for long, hydrophobic sequences like Semaglutide often suffers from resin shrinkage and peptide aggregation, leading to incomplete couplings and difficult-to-remove deletion sequences. Fragment condensation breaks the synthesis into manageable, high-purity blocks, improving overall coupling efficiency and simplifying the final purification profile.
Q: Is this synthesis method suitable for industrial scale-up?
A: Yes, the patent explicitly states that the method facilitates industrial mass production. By reducing the burden of crude product purification and improving the yield of the target peptide through better impurity control (specifically reducing +Gly and cyclization byproducts), the process becomes more robust and cost-effective for large-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Semaglutide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust synthesis pathways in the competitive landscape of GLP-1 therapeutics. Our technical team has extensively analyzed the fragment condensation strategies outlined in patent CN113135991A and possesses the expertise to implement these advanced protocols effectively. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our facilities are equipped with rigorous QC labs capable of detecting trace impurities down to the ppm level, guaranteeing that every batch meets stringent purity specifications required for regulatory filings.
We invite you to collaborate with us to optimize your supply chain for Semaglutide and related peptide intermediates. By leveraging our technical capabilities, you can secure a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage potential partners to contact our technical procurement team to request specific COA data and route feasibility assessments, allowing us to demonstrate how our advanced manufacturing processes can drive value and reliability for your organization.
