Advanced Thiocarbamate Synthesis: Scalable Solutions for Pharmaceutical and Agrochemical Manufacturing
Advanced Thiocarbamate Synthesis: Scalable Solutions for Pharmaceutical and Agrochemical Manufacturing
The global demand for high-purity heterocyclic intermediates continues to drive innovation in process chemistry, particularly for compounds serving dual roles in pharmaceutical and agrochemical applications. A pivotal advancement in this sector is detailed in patent CN101072756B, which discloses a robust method for producing O-aryl N-(6-alkoxy-2-pyridyl)-N-alkyl thiocarbamate derivatives. These compounds are critical precursors for antifungal agents used in treating conditions like athlete's foot, as well as potent herbicides. The patent addresses long-standing challenges in the industry by replacing hazardous reagents with a safer, more economically viable pathway that utilizes carbon disulfide and halomethanes. For R&D directors and procurement specialists, understanding this shift is essential for optimizing supply chains and ensuring regulatory compliance in the manufacture of complex fine chemicals.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of thiocarbamate derivatives relied heavily on the use of thiophosgene, a reagent notorious for its extreme toxicity and stringent handling requirements. As illustrated in the background art of the patent, traditional routes often involved reacting amines or phenols with thiophosgene to generate reactive chlorothioformates or isothiocyanates. While chemically effective, these processes impose severe burdens on industrial facilities, requiring specialized gas scrubbing systems and rigorous safety protocols that drastically inflate operational costs. Furthermore, alternative methods attempting to avoid thiophosgene, such as those utilizing 2,4-dinitrophenyl leaving groups, introduced significant downstream purification challenges. The removal of byproducts like 2,4-dinitrothiophenol proved difficult, often compromising the purity profile required for pharmaceutical grade intermediates and leading to poor reproducibility in yield.
The Novel Approach
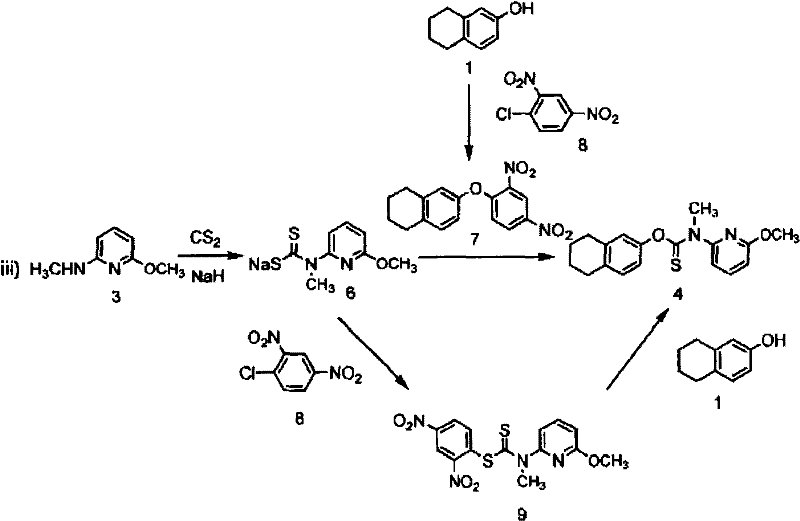
The methodology described in patent CN101072756B represents a paradigm shift by leveraging a halomethane-mediated coupling strategy that bypasses the need for toxic gases entirely. Instead of relying on unstable chloroformates, the process generates a stable alkali metal salt of N-(6-alkoxy-2-pyridyl)-N-alkyldithiocarbamic acid in situ. This intermediate is then coupled with a phenoxide salt in the presence of a dihalomethane, such as dichloromethane or dibromomethane. This approach not only eliminates the safety risks associated with thiophosgene but also streamlines the workflow by avoiding the generation of difficult-to-remove nitro-aromatic impurities. The result is a cleaner reaction profile that facilitates easier isolation of the target thiocarbamate, making it an ideal candidate for reliable pharmaceutical intermediate supplier networks seeking to minimize waste and maximize throughput.
Mechanistic Insights into Halomethane-Mediated Coupling
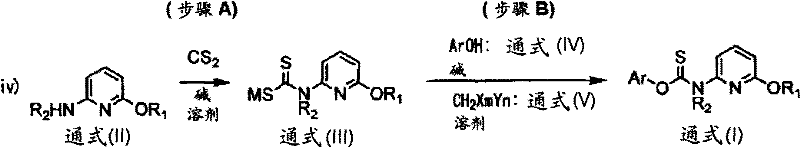
The core innovation of this synthesis lies in the unexpected reactivity enabled by the addition of a halomethane coupling agent. Mechanistically, the reaction begins with the formation of a dithiocarbamate anion via the nucleophilic attack of a secondary amine on carbon disulfide in the presence of a strong base like sodium hydride. Simultaneously, the phenolic substrate is deprotonated to form a phenoxide nucleophile. In the absence of a halomethane, these two anionic species do not react to form the thiocarbamate linkage, even under heating. However, the introduction of a dihalomethane (CH2X2) activates the system, likely through the formation of a transient S-alkylated intermediate or by facilitating a nucleophilic substitution that bridges the sulfur of the dithiocarbamate and the oxygen of the phenol. This unique activation allows the construction of the O-C(=S)-N backbone under remarkably mild conditions, typically between 10°C and 50°C, preserving the integrity of sensitive functional groups.
From an impurity control perspective, this mechanism offers distinct advantages over prior art. Traditional routes often generated stoichiometric amounts of corrosive HCl or toxic thiophenols that required extensive washing and neutralization steps. In contrast, the halomethane-mediated pathway produces inorganic salts and minor organic byproducts that are readily removed via standard aqueous workups or crystallization. The patent data indicates that using solvents like DMF or THF further enhances the solubility of the ionic intermediates, ensuring homogeneous reaction conditions that minimize side reactions. For quality assurance teams, this translates to a more consistent impurity profile, reducing the risk of batch failures and ensuring that the final high-purity OLED material or API intermediate meets stringent specifications without requiring complex chromatographic purification.
How to Synthesize O-aryl N-(6-alkoxy-2-pyridyl)-N-alkyl thiocarbamate Efficiently
Implementing this synthesis requires precise control over stoichiometry and temperature to maximize yield and safety. The process is generally divided into two stages: the formation of the dithiocarbamate salt and the subsequent coupling with the phenol. Operators must ensure that the molar ratio of carbon disulfide and base to the amine substrate is maintained between 1.0 and 1.1 equivalents to prevent excess reagent accumulation while driving the reaction to completion. The choice of solvent is also critical; while ethers like THF are suitable for the initial salt formation, polar aprotic solvents like DMF are preferred for the coupling step to solubilize the phenoxide salt. Detailed standard operating procedures regarding the specific addition rates of halomethanes and quenching protocols are essential for safe scale-up.
- React 6-alkoxy-2-alkylaminopyridine with carbon disulfide and a base (e.g., NaH) in a solvent like THF to form the alkali metal salt of N-(6-alkoxy-2-pyridyl)-N-alkyldithiocarbamic acid.
- Treat the desired phenol (Ar-OH) with a base (e.g., NaOH) in a polar solvent like DMF to generate the phenoxide salt.
- Combine the dithiocarbamate salt and phenoxide salt, then add a halomethane (e.g., CH2Br2) at 10-50°C to effect the coupling and form the final thiocarbamate ester.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this thiophosgene-free methodology offers substantial strategic benefits beyond mere chemical efficiency. The primary advantage lies in the drastic simplification of logistics and hazard management. By eliminating the need to transport and store compressed toxic gases, facilities can significantly reduce their insurance premiums and regulatory compliance overhead. Furthermore, the raw materials required—carbon disulfide, common phenols, and dihalomethanes—are commodity chemicals with robust global supply chains, ensuring continuity of supply even during market fluctuations. This stability is crucial for maintaining production schedules for critical agrochemical intermediates and pharmaceutical ingredients.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the elimination of expensive safety infrastructure and waste treatment costs associated with thiophosgene. Additionally, the higher selectivity of the reaction reduces the consumption of solvents and energy required for purification. By avoiding the formation of tenacious impurities like dinitrothiophenols, the downstream processing time is shortened, leading to lower labor costs and increased plant capacity utilization. The use of inexpensive bases like sodium hydroxide or sodium hydride further contributes to a favorable cost of goods sold (COGS) profile.
- Enhanced Supply Chain Reliability: Relying on commodity reagents rather than specialized, regulated gases mitigates the risk of supply disruptions. The process operates under mild thermal conditions (10-50°C), which reduces the energy load on manufacturing plants and allows for the use of standard glass-lined or stainless steel reactors without the need for exotic metallurgy. This flexibility enables manufacturers to diversify their production sites, reducing lead time for high-purity pharmaceutical intermediates and ensuring that customers receive their orders on schedule regardless of regional constraints.
- Scalability and Environmental Compliance: The environmental footprint of this synthesis is markedly lower than conventional routes. The absence of sulfur-containing toxic gases and the reduced generation of hazardous organic waste align with modern green chemistry principles. This makes the process highly scalable, as environmental permitting for expansion is less cumbersome compared to facilities handling thiophosgene. For companies aiming to meet corporate sustainability goals, adopting this technology demonstrates a commitment to safer, cleaner manufacturing practices while maintaining commercial viability.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this thiocarbamate synthesis technology. These insights are derived directly from the experimental data and claims within patent CN101072756B, providing a factual basis for evaluating the feasibility of this route for your specific application needs. Understanding these nuances is vital for technical teams assessing the compatibility of this method with existing production lines.
Q: Why is the halomethane activation step critical in this synthesis?
A: Without the addition of a halomethane such as dichloromethane or dibromomethane, the reaction between the phenoxide salt and the dithiocarbamate salt does not proceed effectively, even at elevated temperatures. The halomethane acts as a crucial coupling mediator that enables the formation of the thiocarbamate bond under mild conditions.
Q: What are the safety advantages of this method over conventional thiophosgene routes?
A: Conventional methods rely on thiophosgene, a highly toxic gas with strict transport and handling regulations. This patented process utilizes carbon disulfide and stable alkali metal salts, significantly reducing operational hazards and eliminating the need for specialized gas handling infrastructure.
Q: Can this process be scaled for industrial production of antifungal intermediates?
A: Yes, the process is designed for industrial scalability. It operates at mild temperatures (10-50°C) using common solvents like DMF and THF, and avoids the purification difficulties associated with nitro-thiophenol byproducts found in older CS2-based methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Thiocarbamate Derivative Supplier
The technological advancements outlined in patent CN101072756B underscore the potential for safer, more efficient production of critical thiocarbamate intermediates. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory processes are successfully translated into robust industrial realities. Our facility is equipped with rigorous QC labs and adheres to stringent purity specifications, guaranteeing that every batch of pharmaceutical or agrochemical intermediate meets the highest international standards. We understand the complexities of managing reactive sulfur chemistry and have the expertise to handle these processes safely and effectively.
We invite you to collaborate with us to leverage this advanced synthesis for your product pipeline. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this thiophosgene-free route can optimize your budget. Please contact us to request specific COA data and route feasibility assessments, and let us support your journey toward more sustainable and cost-effective manufacturing.
