Advanced Synthesis of Lesinurad Intermediates: A Safer, Scalable Route for Global Supply
Advanced Synthesis of Lesinurad Intermediates: A Safer, Scalable Route for Global Supply
The pharmaceutical landscape for gout therapeutics has evolved significantly with the development of uricosuric agents like Lesinurad (RDEA594). As demand for high-purity active pharmaceutical ingredients (APIs) grows, the efficiency and safety of the supply chain become paramount. Patent CN103524440A introduces a groundbreaking preparation method for Lesinurad and its key intermediates, addressing critical bottlenecks in traditional manufacturing. This technical insight report analyzes the novel synthetic pathway, highlighting its potential to redefine cost structures and supply reliability for global procurement and R&D teams seeking a reliable pharmaceutical intermediates supplier.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Lesinurad and its precursors has relied on pathways that present significant operational and safety challenges. Early developmental routes, such as those reported by Valeant Pharmaceuticals, often necessitated the use of thiophosgene, a reagent known for its extreme toxicity and difficult handling requirements in an industrial setting. Furthermore, conventional strategies typically involved constructing the core structure and then performing a complex transformation of an amino group into a bromine atom at a late stage. This specific halogenation step is not only operationally complicated but also relies on expensive raw materials, creating a substantial burden on the cost of goods sold (COGS). The reliance on hazardous gases and multi-step functional group interconversions inherently increases the risk profile and extends the lead time for high-purity pharmaceutical intermediates.
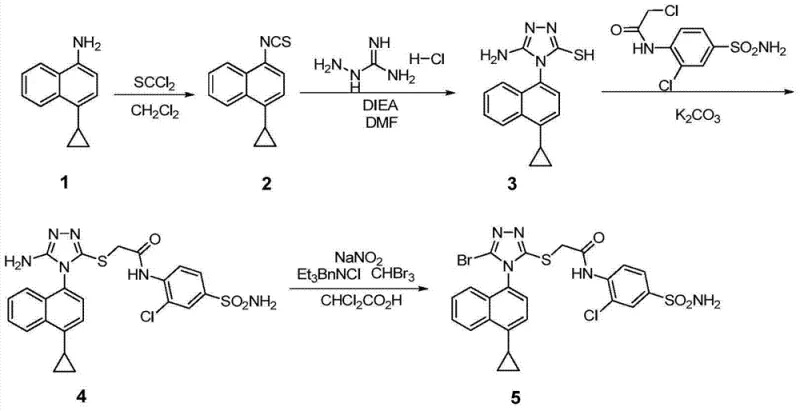
The Novel Approach
In stark contrast, the methodology disclosed in CN103524440A offers a streamlined, economically efficient, and environmentally friendly alternative. The core innovation lies in the construction of a novel triazole-based intermediate that bypasses the need for thiophosgene entirely. Instead, the process utilizes 4-cyclopropyl-naphthylamine as a starting material, reacting it with carbon disulfide and hydrazine hydrate to form a stable dithiocarbazate precursor. This is subsequently cyclized to form the triazole ring. Crucially, the bromination is achieved directly on the triazole moiety rather than the naphthalene ring, utilizing accessible brominating agents. This strategic shift simplifies the purification process, as intermediates and final products can be easily separated, thereby enhancing overall throughput and reducing waste generation significantly.
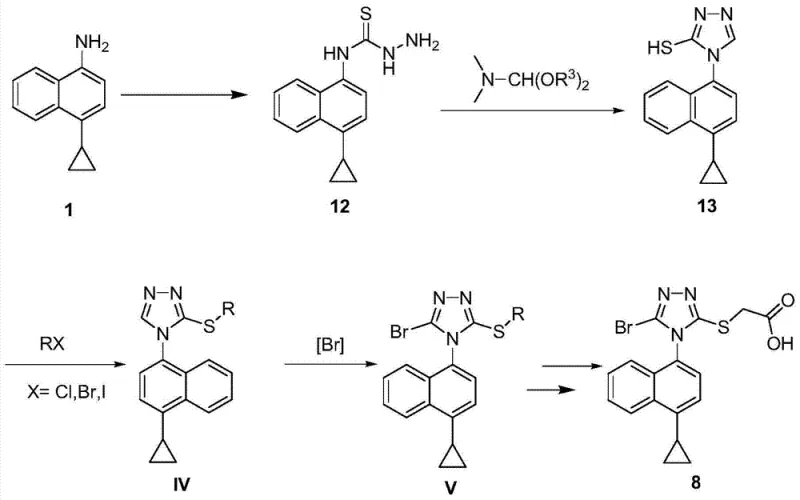
Mechanistic Insights into Triazole Cyclization and Selective Bromination
The chemical elegance of this new route is rooted in the efficient formation of the 1,2,4-triazole ring system. The reaction begins with the nucleophilic attack of the amine on carbon disulfide in the presence of a base, forming a dithiocarbamate salt. Subsequent treatment with hydrazine hydrate under reflux conditions facilitates the cyclization to yield the hydrazino-thione intermediate (Compound 12). The closure of the triazole ring is then achieved using DMF dialkyl acetals, such as DMF dimethylacetal or diethyl acetal. This cyclization step is highly robust, proceeding in solvents like 1,4-dioxane at elevated temperatures to ensure complete conversion. The resulting thiol-triazole (Compound 13) serves as a versatile pivot point for further functionalization, demonstrating high stability and ease of isolation.
Following the formation of the triazole core, the process employs a selective alkylation with haloacetates (e.g., ethyl bromoacetate) to install the side chain, yielding Compound 15. The subsequent bromination step is particularly noteworthy for its selectivity and mild conditions. Using bromine or N-bromo-succinimide (NBS) in solvents like acetonitrile or dichloromethane, the reaction introduces the bromine atom specifically at the 5-position of the triazole ring. This direct halogenation avoids the harsh conditions typically required for aromatic substitution on the naphthalene core. The mechanism ensures that the cyclopropyl group and the ester side chain remain intact, preserving the structural integrity required for the final coupling steps to form Lesinurad. This precision minimizes the formation of regio-isomers and other impurities, directly contributing to a cleaner impurity profile.
How to Synthesize Lesinurad Intermediate Efficiently
The synthesis of the key brominated intermediate (Compound 16) involves a sequence of well-defined unit operations that are amenable to standard chemical processing equipment. The process initiates with the preparation of the thiol-triazole scaffold, followed by S-alkylation and finally, electrophilic bromination. Each step has been optimized to maximize yield while minimizing solvent usage and reaction time. For R&D teams looking to replicate or adapt this chemistry, the detailed standardized synthesis steps see the guide below.
- React 4-cyclopropyl-naphthylamine with carbon disulfide and hydrazine hydrate to form the dithiocarbazate intermediate (Compound 12).
- Cyclize Compound 12 using DMF dialkyl acetal to generate the triazole thiol derivative (Compound 13).
- Alkylate Compound 13 with ethyl bromoacetate to form the thioester (Compound 15), followed by selective bromination to yield the key brominated intermediate (Compound 16).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this novel synthetic route represents a strategic opportunity to optimize the supply of gout therapeutics intermediates. By fundamentally altering the reagent profile and reaction sequence, the process addresses several pain points associated with the legacy manufacturing of complex heterocyclic APIs. The elimination of high-risk reagents and the simplification of downstream processing translate directly into tangible business benefits regarding cost, reliability, and compliance.
- Cost Reduction in Manufacturing: The economic advantages of this route are driven primarily by the substitution of expensive and hazardous reagents with commodity chemicals. Thiophosgene, required in prior art, commands a high price due to its specialized handling and transport requirements; replacing it with carbon disulfide and hydrazine hydrate drastically lowers raw material costs. Furthermore, the avoidance of late-stage, low-yield halogenation steps on the naphthalene ring reduces the consumption of precious starting materials. The simplified workup procedures, which often involve simple filtration or extraction rather than complex chromatography, significantly reduce labor and solvent costs, leading to substantial cost savings in pharmaceutical intermediates manufacturing.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the regulatory restrictions surrounding toxic gases like thiophosgene. By removing this bottleneck, the new route mitigates the risk of production stoppages due to safety audits or transportation bans. The raw materials employed, such as 4-cyclopropyl-naphthylamine and ethyl bromoacetate, are widely available from multiple global suppliers, ensuring a robust and diversified supply base. This redundancy protects against single-source failures and price volatility, ensuring a steady flow of high-purity pharmaceutical intermediates to downstream API manufacturers.
- Scalability and Environmental Compliance: As regulatory pressure mounts regarding environmental, social, and governance (ESG) criteria, this process offers a distinct advantage. It operates without heavy metal catalysts and avoids the generation of sulfur-containing waste streams associated with thiophosgene degradation. The reactions are conducted in common organic solvents that can be readily recovered and recycled. The ease of scaling from laboratory to commercial production is evidenced by the straightforward reaction conditions (ambient to moderate temperatures) and the high purity of the crude products, which minimizes the load on wastewater treatment facilities and aligns with green chemistry principles.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel Lesinurad synthesis pathway. These insights are derived directly from the experimental data and beneficial effects reported in the patent literature, providing clarity for technical decision-makers.
Q: Why is the new synthesis route for Lesinurad considered safer than prior art?
A: The novel method described in CN103524440A completely eliminates the use of thiophosgene, a highly toxic and hazardous gas used in conventional routes. It replaces dangerous reagents with safer alternatives like carbon disulfide and hydrazine hydrate under controlled conditions.
Q: How does this process improve cost efficiency for procurement teams?
A: By utilizing readily available and inexpensive raw materials such as 4-cyclopropyl-naphthylamine and avoiding expensive, specialized halogenation reagents required for late-stage functionalization, the overall material cost is significantly reduced. Additionally, simplified purification steps lower processing expenses.
Q: Is this synthetic route suitable for large-scale industrial production?
A: Yes, the process is designed for scalability. It avoids heavy metal catalysts and environmentally harmful solvents, features easy separation and purification of intermediates, and achieves yields comparable to or higher than existing methods, making it ideal for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Lesinurad Intermediate Supplier
The technical advancements detailed in patent CN103524440A underscore the potential for more efficient and sustainable production of gout therapeutics. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory processes are successfully translated into robust industrial realities. Our commitment to quality is unwavering, with stringent purity specifications and rigorous QC labs dedicated to verifying every batch of Lesinurad intermediate against the highest international standards.
We invite global partners to leverage our expertise for their next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to support your supply chain optimization goals.
