Advanced Iridium Phosphorescent Emitters: Scaling High-Efficiency OLED Materials for Next-Gen Displays
The rapid evolution of flat-panel display technology has placed organic light-emitting diodes (OLEDs) at the forefront of the optoelectronic industry, driven by the demand for higher resolution, flexibility, and energy efficiency. Central to this technological leap is the development of advanced phosphorescent emitting materials, specifically organometallic complexes capable of harvesting both singlet and triplet excitons to theoretically achieve 100% internal quantum efficiency. Patent CN111253442A introduces a groundbreaking class of organic iridium metal complexes designed to address the persistent challenges of low current efficiency and stability in existing OLED architectures. These novel compounds feature a generalized structural formula where specific heterocyclic ligands are combined with tunable substituents to precisely modulate emission wavelengths and enhance device performance. As a reliable OLED material supplier, understanding the nuances of these molecular designs is crucial for integrating next-generation emitters into commercial production lines. The structural versatility allows for fine-tuning of the Highest Occupied Molecular Orbital (HOMO) and Lowest Unoccupied Molecular Orbital (LUMO) levels, facilitating better charge balance within the emissive layer.
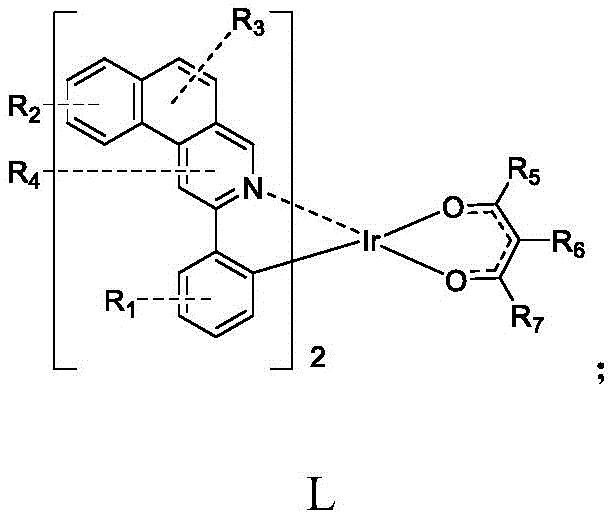
Furthermore, the inclusion of deuterium, halogen, or various alkyl and aryl groups at specific positions on the ligand framework provides a powerful toolkit for chemists to optimize morphological stability and prevent concentration quenching. This level of molecular engineering is essential for extending the operational lifetime of OLED panels, a key metric for consumer electronics manufacturers. The patent highlights that these complexes are not merely theoretical constructs but are synthesized through practical, scalable routes that yield high-purity products suitable for vacuum deposition processes. By leveraging these advanced materials, display manufacturers can achieve superior brightness and color purity, directly translating to enhanced user experiences in smartphones, televisions, and wearable devices. The strategic modification of the ancillary ligands also plays a pivotal role in improving the thermal stability of the complexes, ensuring they withstand the rigors of the evaporation process without decomposition.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the development of phosphorescent OLED materials has been hindered by several critical bottlenecks associated with conventional iridium complexes. Many early-generation emitters suffered from significant efficiency roll-off at high brightness levels, limiting their utility in high-performance display applications where peak luminance is required. Additionally, traditional synthetic routes often involved harsh reaction conditions, expensive catalysts, or multi-step procedures that resulted in low overall yields and difficult purification challenges. The presence of impurities, even at trace levels, can act as quenching sites that drastically reduce the efficiency and lifespan of the final OLED device. Furthermore, conventional ligands often lacked the necessary thermal and morphological stability, leading to crystallization within the thin film over time, which manifests as dark spots and device failure. These limitations necessitated a search for new molecular architectures that could offer robust performance without compromising on manufacturability or cost-effectiveness. The reliance on non-optimized ligand systems often resulted in poor solubility or processability, complicating the solution-processing techniques that are increasingly desired for large-area display fabrication.
The Novel Approach
The innovative strategy presented in this patent overcomes these hurdles by introducing a modular design based on a specific cyclometalated ligand scaffold coupled with tailored beta-diketonate ancillary ligands. This approach allows for the systematic variation of substituents to fine-tune the electronic and steric properties of the complex without altering the core coordination geometry. By selecting specific heterocyclic ligands, the inventors have successfully regulated the emission wavelength, enabling coverage across the visible spectrum with high color purity. The synthesis method described is notably streamlined, utilizing a two-step process that first forms a chloro-bridged iridium dimer and subsequently converts it into the final neutral complex using readily available reagents. This simplification reduces the number of purification steps required, thereby minimizing material loss and lowering the overall production cost. Moreover, the resulting complexes exhibit excellent thermal stability and sublimation properties, making them ideal candidates for high-vacuum thermal evaporation, the industry-standard method for depositing small-molecule OLED layers. The ability to achieve high current efficiency and brightness simultaneously represents a significant leap forward in emitter technology.
Mechanistic Insights into Iridium Cyclometalation and Coordination
The core of this technology lies in the precise coordination chemistry between the iridium center and the organic ligands, a process that dictates the photophysical properties of the final emitter. The synthesis begins with the cyclometalation of the main ligand precursor (Formula A) with iridium trichloride, forming a stable chloro-bridged dimer intermediate (Formula B). This step is critical as it establishes the strong Ir-C and Ir-N bonds that define the kinetic stability of the complex. The reaction typically proceeds in a mixed solvent system of ethylene glycol ethyl ether and water under reflux, conditions that facilitate the cleavage of the C-H bond and its replacement with the metal-carbon bond. The formation of the dimer is a thermodynamically favorable process that locks the ligand into a rigid conformation, reducing non-radiative decay pathways and enhancing phosphorescence quantum yield. Understanding this mechanistic step is vital for scaling, as incomplete cyclometalation can lead to heterogeneous mixtures that are difficult to separate and detrimental to device performance.
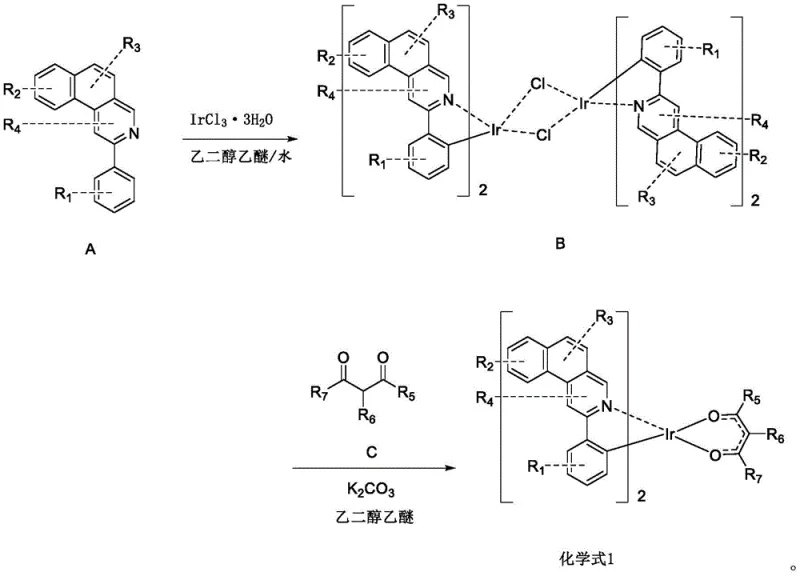
Following the formation of the dimer, the second stage involves the displacement of the bridging chloride atoms by the anionic oxygen donors of the ancillary ligand (Formula C), typically a substituted acetylacetone derivative. This substitution reaction is facilitated by the presence of a base, such as potassium carbonate, which neutralizes the hydrochloric acid byproduct and drives the equilibrium towards the formation of the neutral bis-cyclometalated complex. The choice of the ancillary ligand is not merely structural; it significantly influences the electron density at the metal center, thereby tuning the energy gap between the ground and excited states. For instance, the introduction of electron-withdrawing groups like fluorine or trifluoromethyl on the ancillary ligand can blue-shift the emission, while electron-donating alkyl groups can red-shift it. This mechanistic flexibility allows for the creation of a library of emitters covering the entire RGB spectrum. Furthermore, the rigid chelation provided by the bidentate ancillary ligand prevents ligand dissociation under thermal stress, ensuring the integrity of the molecule during the high-temperature evaporation process used in OLED fabrication.
How to Synthesize Organic Iridium Metal Complexes Efficiently
The synthesis of these high-performance emitters follows a robust and reproducible protocol that balances yield with purity, essential for commercial viability. The process leverages standard laboratory equipment and common chemical reagents, making it accessible for scale-up without requiring specialized high-pressure reactors or exotic catalysts. The initial cyclometalation step is performed under an inert atmosphere to prevent oxidation of the sensitive iridium species, ensuring consistent batch-to-batch quality. Detailed standardized synthesis steps for specific derivatives like L001, L020, and L053 are outlined in the patent examples, demonstrating the versatility of the general method across different substituent patterns. For a comprehensive guide on executing this synthesis with optimal parameters, please refer to the structured protocol below.
- React the cyclometalated ligand precursor (Formula A) with Iridium Trichloride (IrCl3·3H2O) in a mixture of ethylene glycol ethyl ether and water under reflux to form the chloro-bridged dimer intermediate (Formula B).
- Purify the intermediate dimer by filtration and washing with water, ethanol, and petroleum ether to remove unreacted starting materials and salts.
- React the purified dimer with the ancillary ligand (Formula C, typically a substituted acetylacetone) and potassium carbonate in ethylene glycol ethyl ether at 110-120°C to yield the final neutral iridium complex.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this novel iridium complex technology offers substantial strategic benefits that extend beyond mere performance metrics. The streamlined synthetic route significantly reduces the complexity of the manufacturing process, which directly correlates to lower operational expenditures and reduced risk of production delays. By eliminating the need for multiple complex purification stages or rare catalytic systems, the overall cost of goods sold can be optimized, allowing for more competitive pricing in the volatile display materials market. The use of common solvents like ethylene glycol ethyl ether and potassium carbonate ensures that raw material sourcing is stable and not subject to the geopolitical fluctuations often associated with specialty reagents. This reliability is paramount for supply chain heads who must guarantee continuous production lines for major display panel manufacturers. Furthermore, the high purity achievable through the described precipitation and chromatography methods reduces the risk of downstream device failure, minimizing warranty claims and protecting brand reputation.
- Cost Reduction in Manufacturing: The synthetic pathway described eliminates the need for expensive transition metal catalysts often required in cross-coupling reactions for ligand synthesis, relying instead on direct cyclometalation with inexpensive iridium chloride. This fundamental shift in chemistry removes the costly step of removing trace heavy metal impurities, which typically requires specialized scavenging resins or additional recrystallization cycles. Consequently, the overall process mass intensity is improved, leading to less waste generation and lower disposal costs. The high yields reported in the examples suggest that raw material utilization is efficient, further driving down the unit cost of the final emitter. These cumulative efficiencies translate into significant cost savings that can be passed down the supply chain or reinvested into further R&D.
- Enhanced Supply Chain Reliability: The precursors required for this synthesis, such as substituted phenylisoquinolines and acetylacetone derivatives, are commodity chemicals or easily synthesized intermediates with established global supply networks. This reduces the dependency on single-source suppliers for exotic starting materials, thereby mitigating the risk of supply disruptions. The robustness of the reaction conditions, which tolerate standard heating and stirring without requiring cryogenic temperatures or ultra-high vacuum, means that production can be easily transferred between different manufacturing sites if necessary. This flexibility is a critical asset for supply chain resilience, ensuring that production targets can be met even in the face of regional logistical challenges. The ability to source materials locally in major chemical hubs further shortens lead times and reduces transportation carbon footprints.
- Scalability and Environmental Compliance: The process is inherently scalable, moving seamlessly from gram-scale laboratory optimization to kilogram and metric-ton commercial production without fundamental changes to the reaction engineering. The use of aqueous workups and standard organic solvents simplifies waste stream management, allowing for easier compliance with increasingly stringent environmental regulations regarding volatile organic compound (VOC) emissions. The precipitation of the intermediate dimer allows for easy isolation without the need for energy-intensive distillation steps, reducing the overall energy consumption of the plant. Additionally, the high purity of the final product minimizes the generation of off-spec material that would otherwise require reprocessing or disposal. This alignment with green chemistry principles not only satisfies regulatory requirements but also appeals to eco-conscious consumers and corporate sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of these iridium complexes in OLED manufacturing. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for decision-makers. Understanding these details is essential for evaluating the feasibility of integrating these materials into existing production workflows. For more specific technical data or custom synthesis requests, our team is available to provide detailed consultations.
Q: What is the primary advantage of the iridium complexes disclosed in CN111253442A?
A: The primary advantage is the significant improvement in current efficiency and brightness when used as dopants in organic electroluminescent devices. By selecting specific heterocyclic ligands and tuning substituents (R groups), the emission wavelength and charge transport properties are optimized compared to conventional emitters.
Q: How is the purity of the final iridium complex ensured during synthesis?
A: The patent describes a robust purification protocol involving precipitation, sequential washing with water and organic solvents, and final purification via silica gel column chromatography. This multi-step process ensures HPLC purity levels exceeding 99.5%, which is critical for preventing device quenching and ensuring long operational lifetimes.
Q: Can these materials be scaled for industrial OLED panel production?
A: Yes, the synthesis utilizes standard reflux conditions and commercially available solvents like ethylene glycol ethyl ether. The reaction steps are straightforward and do not require exotic catalysts or extreme pressure, making the process highly amenable to scale-up from laboratory grams to metric ton quantities required for commercial display manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Organic Iridium Metal Complex Supplier
The technical potential of the iridium complexes disclosed in CN111253442A represents a significant opportunity for advancing OLED display performance, but realizing this potential requires a manufacturing partner with deep expertise in organometallic chemistry. NINGBO INNO PHARMCHEM stands as a premier CDMO partner, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the sensitive nature of iridium chemistry, ensuring that stringent purity specifications are met consistently through our rigorous QC labs. We understand that the transition from patent to product involves navigating complex regulatory and quality assurance landscapes, and our dedicated teams are prepared to support you through every stage of the product lifecycle. Our commitment to excellence ensures that the high efficiency and brightness promised by the patent are delivered in every batch we produce.
To explore how these advanced materials can optimize your display portfolio, we invite you to initiate a dialogue with our technical procurement team. We encourage you to request a Customized Cost-Saving Analysis tailored to your specific volume requirements and device architecture. Our experts are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities align with your strategic goals. By partnering with us, you gain access to a secure, high-quality supply chain that empowers your innovation in the competitive display market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
