Scalable Synthesis of 2,7-Dihydroxy-9-fluorenone: Technical Breakthroughs for Global Supply Chains
Scalable Synthesis of 2,7-Dihydroxy-9-fluorenone: Technical Breakthroughs for Global Supply Chains
The pharmaceutical and agrochemical industries constantly demand high-purity intermediates that can be manufactured reliably at scale. Patent CN102424651A introduces a significant advancement in the synthesis of 2,7-diacetoxy-9-fluorenone and its hydrolyzed derivative, 2,7-dihydroxy-9-fluorenone. These fluorenone derivatives serve as critical building blocks for anticancer agents, sympathetic nerve inhibitors, and advanced polymer stabilizers. The disclosed methodology addresses long-standing challenges in organic synthesis, specifically targeting the inefficiencies associated with traditional multi-step oxidation processes. By leveraging a streamlined oxidative protocol followed by controlled hydrolysis, this technology offers a robust pathway for producing complex aromatic ketones. For R&D directors and procurement specialists, understanding the nuances of this patent is essential for securing a reliable pharmaceutical intermediate supplier capable of meeting stringent quality specifications.
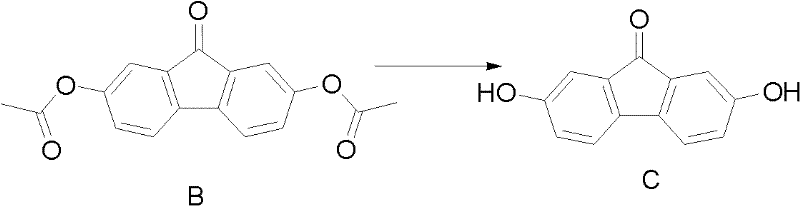
The structural integrity of these compounds is paramount for their downstream applications in dye synthesis and photosensitive materials. As illustrated in the provided chemical diagrams, the transformation preserves the rigid fluorenone core while precisely functionalizing the 2 and 7 positions. This level of regioselectivity is often difficult to achieve without expensive protecting group strategies or harsh conditions that degrade the molecular scaffold. The patent details a method that not only achieves high conversion rates but also ensures that the final product profile is clean, with minimal impurities that could complicate subsequent coupling reactions. For companies seeking cost reduction in fine chemical manufacturing, adopting such efficient synthetic routes is a strategic imperative to maintain competitiveness in the global market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of 2,7-dihydroxy-9-fluorenone and its acetylated precursors has been plagued by operational complexities and safety hazards. Conventional literature describes multi-step oxidation sequences that often require cryogenic temperatures, excessive solvent volumes, and dangerous reagents. These traditional approaches frequently necessitate tedious purification steps, such as column chromatography, which are notoriously difficult to translate from the laboratory bench to industrial reactors. The reliance on low-temperature conditions increases energy consumption significantly, while the use of large solvent quantities creates substantial waste disposal burdens. Furthermore, the variability in yield and purity associated with these older methods poses a severe risk to supply chain continuity. For a procurement manager, these factors translate into unpredictable lead times and inflated raw material costs, making the conventional synthesis routes economically unsustainable for large-volume production.
The Novel Approach
In stark contrast, the methodology outlined in CN102424651A presents a simplified and robust alternative that eliminates many of these bottlenecks. The novel approach utilizes a direct oxidative conversion of Compound A into Compound B within an organic acid medium, followed by a straightforward hydrolysis step to yield Compound C. This process operates under relatively mild thermal conditions, typically ranging from 0°C to 80°C, which significantly reduces the energy footprint of the manufacturing process. Crucially, the workup procedure relies on simple phase separation, distillation, and filtration, completely bypassing the need for column chromatography. 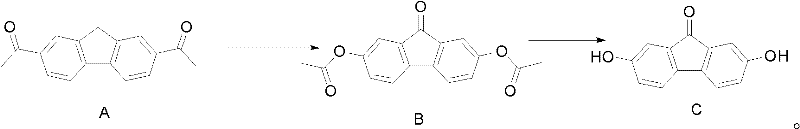
This streamlining of the purification workflow is a game-changer for commercial scale-up of complex organic intermediates. By avoiding chromatographic separation, the process becomes inherently more scalable, allowing for the processing of hundreds of kilograms of material without a proportional increase in labor or silica gel costs. The ability to isolate the product via crystallization ensures that the final material meets high-purity standards (>99% by HPLC) directly from the reactor. This efficiency not only accelerates production cycles but also enhances the overall safety profile of the operation by minimizing operator exposure to hazardous solvents and stationary phases.
Mechanistic Insights into Oxidative Functionalization and Hydrolysis
The core of this synthetic strategy lies in the precise control of oxidative conditions to functionalize the fluorene skeleton. The reaction begins with the treatment of Compound A with a selected oxidant in the presence of an organic acid, such as acetic acid or trifluoroacetic acid. This environment facilitates the formation of the 9-fluorenone carbonyl group while simultaneously installing or preserving the acetoxy functionalities at the 2 and 7 positions. The choice of oxidant is flexible, with the patent listing options ranging from peracids like trifluoroperoxyacetic acid to inorganic oxidants like sodium dichromate. This versatility allows manufacturers to optimize the reaction based on reagent availability and cost, providing a buffer against supply chain disruptions for specific chemicals. The reaction kinetics are monitored via HPLC or LC-MS to ensure complete conversion of the starting material before proceeding to the workup phase.
Following the isolation of Compound B, the second critical stage involves alkaline hydrolysis to generate the final dihydroxy product. 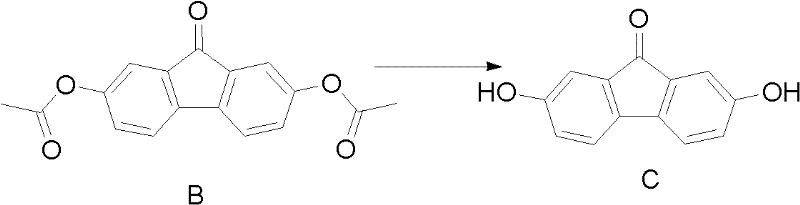
This step is conducted under an inert gas atmosphere to prevent unwanted side reactions or degradation of the sensitive phenolic groups. The use of bases such as sodium hydroxide or potassium carbonate in a biphasic system (organic solvent and aqueous base) ensures efficient cleavage of the acetate esters. The temperature is carefully maintained between 0°C and 60°C to balance reaction rate with product stability. Once the hydrolysis is complete, acidification precipitates the target molecule, which can then be easily filtered and washed. This mechanistic clarity allows process chemists to fine-tune parameters for maximum yield, ensuring that the impurity profile remains within acceptable limits for pharmaceutical applications. The rigorous control over these reaction variables is what enables the consistent production of high-purity fluorenone derivatives required by discerning clients.
How to Synthesize 2,7-Dihydroxy-9-fluorenone Efficiently
Implementing this synthesis route requires careful attention to reagent stoichiometry and thermal management to replicate the high yields reported in the patent data. The process is designed to be modular, allowing for the optimization of the oxidation step independently from the hydrolysis step. Operators must ensure that the organic acid solution is prepared correctly and that the oxidant is added at a controlled rate to manage exotherms. Detailed standard operating procedures (SOPs) should be established to monitor the reaction progress using analytical tools like LC-MS, ensuring that the endpoint is reached before quenching.
- Oxidize Compound A in an organic acid solution with an oxidant at 0-80°C, followed by alkaline wash and filtration to isolate Compound B.
- React Compound B in an organic solvent with an alkaline liquid under inert gas protection to effect hydrolysis.
- Acidify the reaction mixture to precipitate the final product, Compound C, followed by filtration and washing.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain heads and procurement managers, the adoption of this patented methodology offers tangible benefits that extend beyond mere chemical yield. The primary advantage lies in the drastic simplification of the downstream processing units. By replacing column chromatography with filtration and crystallization, the manufacturing facility can significantly reduce its dependency on specialized consumables and skilled labor for purification. This shift translates directly into cost reduction in fine chemical manufacturing, as the operational expenditure per kilogram of product is lowered. Furthermore, the reduced solvent usage and waste generation align with modern environmental compliance standards, mitigating the risk of regulatory penalties and enhancing the sustainability profile of the supply chain.
- Cost Reduction in Manufacturing: The elimination of chromatographic purification is a major driver for cost efficiency. Column chromatography is resource-intensive, requiring vast amounts of silica gel and solvents, which are costly to purchase and dispose of. By utilizing a precipitation-based isolation method, the process minimizes these variable costs. Additionally, the ability to use a wide range of commercially available oxidants allows procurement teams to source materials based on market pricing fluctuations, further optimizing the bill of materials. The high yield reported (65-83%) ensures that raw material utilization is maximized, reducing the cost of goods sold (COGS) and improving margin potential for the final API or intermediate.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route contributes to greater supply chain resilience. Traditional methods that rely on specific, hard-to-source reagents or complex equipment are prone to bottlenecks. In contrast, this method uses common industrial solvents like dichloromethane, toluene, and ethyl acetate, which are readily available from multiple global suppliers. The mild reaction conditions also reduce the risk of equipment failure or safety incidents that could halt production. For a reliable pharmaceutical intermediate supplier, this reliability is crucial for maintaining consistent delivery schedules to downstream partners, ensuring that their own production lines remain uninterrupted.
- Scalability and Environmental Compliance: Scaling a chemical process often introduces new challenges, but this methodology is inherently designed for expansion. The unit operations involved—stirring, heating, filtering, and drying—are standard in almost any chemical plant, facilitating easy technology transfer from pilot scale to full commercial production. Moreover, the reduction in solvent volume and the avoidance of heavy metal-laden silica waste simplify wastewater treatment and solid waste disposal. This environmental friendliness is increasingly important for multinational corporations aiming to meet strict ESG (Environmental, Social, and Governance) goals. The process demonstrates that high efficiency and environmental responsibility can go hand in hand in modern chemical manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of 2,7-dihydroxy-9-fluorenone. These insights are derived directly from the technical specifications and experimental data provided in the patent documentation. Understanding these details helps stakeholders make informed decisions about integrating this intermediate into their supply networks.
Q: What is the purity level achievable with this synthesis method?
A: According to patent CN102424651A, the method yields products with HPLC purity greater than 99% after recrystallization, eliminating the need for complex column chromatography.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process utilizes simple unit operations such as filtration and crystallization rather than labor-intensive column separation, making it highly amenable to commercial scale-up.
Q: What oxidants are compatible with this reaction pathway?
A: The patent specifies a wide range of oxidants including hydrogen peroxide, trifluoroperoxyacetic acid, sodium dichromate, and m-chloroperoxybenzoic acid, offering flexibility in reagent sourcing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,7-Dihydroxy-9-fluorenone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of life-saving medicines and advanced materials. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the demands of both clinical trials and full-scale manufacturing. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that utilize state-of-the-art analytical instrumentation. Our capability to implement the efficient synthesis routes described in patents like CN102424651A allows us to offer competitive pricing without compromising on quality or safety standards.
We invite global partners to collaborate with us to optimize their supply chains for fluorenone derivatives. By leveraging our technical expertise, you can achieve significant efficiencies in your own manufacturing processes. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our solutions can drive value for your organization.
