Scalable Catalytic Synthesis of 4-Demethoxydaunomycinone Intermediates for Oncology Applications
Scalable Catalytic Synthesis of 4-Demethoxydaunomycinone Intermediates for Oncology Applications
The development of efficient synthetic routes for anthracycline antibiotics remains a cornerstone of modern oncology drug manufacturing. Patent CN1045259A discloses a groundbreaking methodology for the preparation of general formula (I) anthracyclinones, specifically targeting 4-demethoxy derivatives which serve as critical precursors for potent antitumor agents like idarubicin. This technology represents a significant paradigm shift from laborious total synthesis towards elegant semi-synthetic strategies that leverage natural chiral pools. The core innovation lies in the transition metal-catalyzed reductive removal of sulfonyl groups or carbonylation at the C-4 position, enabling precise structural modification of the anthraquinone backbone. By utilizing specific palladium or nickel catalysts in conjunction with hydride donors or carbon monoxide, manufacturers can achieve high conversion rates while preserving the sensitive stereochemistry of the tetracyclic ring system. This approach not only streamlines the production workflow but also addresses the growing demand for high-purity pharmaceutical intermediates required for clinical-grade active pharmaceutical ingredients (APIs).
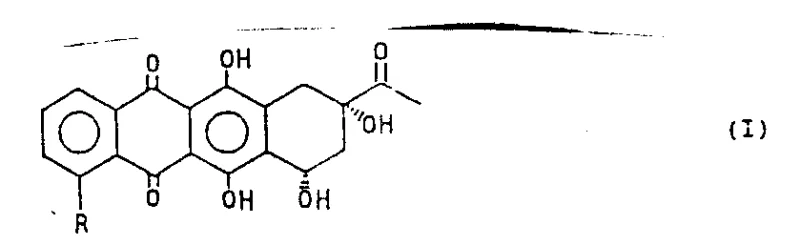
As the global market for anticancer therapeutics expands, the need for reliable pharmaceutical intermediate suppliers who can deliver complex molecules with consistent quality has never been more critical. The methodology outlined in this patent provides a robust framework for producing 4-demethoxydaunomycinone and its esters, which are essential building blocks in the value chain of anthracycline drugs. The versatility of the process allows for the introduction of various functional groups (R = H or COOR1) at the C-4 position, offering flexibility for downstream derivatization. For procurement managers and supply chain directors, understanding the mechanistic underpinnings and operational advantages of this catalytic system is vital for assessing long-term supply security and cost-efficiency in API manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 4-demethoxyanthracyclinones has been plagued by significant challenges related to stereochemical control and process complexity. Traditional total synthesis routes, such as those described in earlier literature like US-A-4046878, often involve lengthy sequences of reactions that require multiple protection and deprotection steps. A major bottleneck in these conventional approaches is the necessity for optical resolution to establish the correct chirality at the C-7 and C-9 positions, which inherently limits the maximum theoretical yield to 50% unless dynamic kinetic resolution is employed. Furthermore, the harsh chemical conditions often required for demethylation or functional group interconversion in the absence of catalytic assistance can lead to degradation of the sensitive anthraquinone chromophore. These factors contribute to elevated production costs, increased waste generation, and prolonged lead times, making it difficult to scale up production to meet commercial demands without compromising on purity specifications. The reliance on stoichiometric reagents rather than catalytic cycles also exacerbates the environmental footprint of the manufacturing process.
The Novel Approach
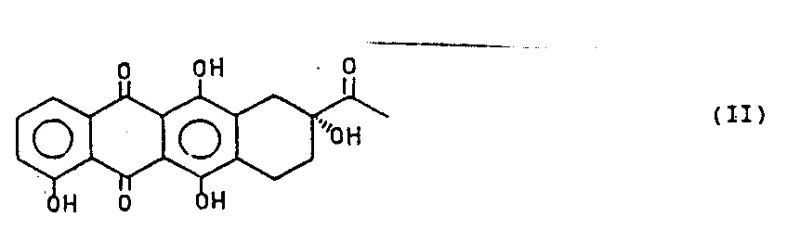
In stark contrast, the novel approach detailed in patent CN1045259A introduces a highly efficient semi-synthetic pathway that circumvents the pitfalls of total synthesis by starting from naturally occurring daunomycinone. This strategy capitalizes on the inherent chirality of the natural product, effectively eliminating the need for resolution steps and ensuring 100% atom economy regarding stereocenters. The key breakthrough involves the conversion of the C-4 hydroxyl group into a superior leaving group, specifically an alkylsulfonyl or arylsulfonyl moiety (Compound V), which is then subjected to transition metal-catalyzed displacement. This transformation is remarkably versatile; under reductive conditions, it yields the 4-demethoxy derivative (R=H), while under carbonylation conditions, it produces the 4-ester derivative (R=COOR1). The use of mild reaction conditions, typically ranging from 60°C to 90°C, preserves the integrity of the molecule while allowing for high throughput. This method significantly reduces the number of synthetic steps and simplifies the purification process, thereby offering a compelling solution for cost reduction in API manufacturing.
Mechanistic Insights into Pd-Catalyzed Reductive Desulfonylation
The heart of this innovative process is the transition metal-catalyzed transformation of the sulfonate intermediate (Compound V) into the target anthracyclinone (Compound VII). This reaction proceeds through a sophisticated catalytic cycle involving oxidative addition, transmetallation (or hydride insertion), and reductive elimination. When operating under reductive conditions, the system utilizes a hydride donor, such as a mixture of formic acid and trialkylamine, which generates a reactive metal-hydride species in situ. The palladium or nickel catalyst, ligated with electron-rich phosphines like triphenylphosphine or chelating diphosphines such as 1,3-bis(diphenylphosphino)propane, facilitates the cleavage of the carbon-sulfur bond. The choice of ligand is critical; bidentate ligands often enhance the stability of the catalytic species and improve regioselectivity, preventing unwanted side reactions at other electrophilic sites on the anthraquinone ring. The reaction temperature is carefully controlled, generally around 60°C, to balance reaction kinetics with the thermal stability of the substrate. This precise control over the catalytic environment ensures that the reduction occurs exclusively at the C-4 position, leaving the C-13 ketone (protected as a dioxolane) and the C-7/C-9 stereocenters untouched.
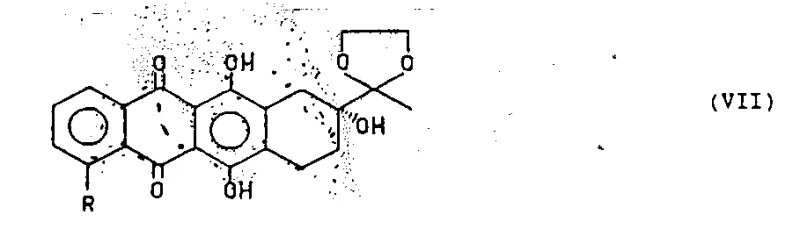
Alternatively, the mechanism can be diverted towards carbonylation by introducing carbon monoxide into the reaction vessel. In this variant, the nucleophilic reagent R1OH attacks the acyl-metal intermediate formed after CO insertion, leading to the formation of ester functionalities at the C-4 position. This dual functionality of the catalytic system—capable of both reduction and carbonylation depending on the atmosphere—highlights its robustness and adaptability for synthesizing a library of analogues. The patent specifies that the molar ratio of catalyst to substrate can range widely from 1:1 to 1:10000, with preferred embodiments utilizing ratios between 1:20 and 1:1000, indicating a highly active catalytic system that minimizes the load of expensive precious metals. Furthermore, the tolerance of the catalyst system to various sulfonyl leaving groups, including trifluoromethanesulfonyl (triflate) and tosyl groups, provides process chemists with flexibility in selecting the most cost-effective sulfonating agent for large-scale operations. The subsequent acid hydrolysis step to remove the 13-dioxolane protecting group and introduce the 7-alpha-hydroxyl group is performed under mild acidic conditions, ensuring that the final product retains its biological potency and structural fidelity.
How to Synthesize 4-Demethoxydaunomycinone Efficiently
The synthesis of 4-demethoxydaunomycinone via this catalytic route involves a sequence of well-defined unit operations that are amenable to standard pharmaceutical manufacturing equipment. The process begins with the protection of the C-13 ketone of 4-demethylation-7-deoxygenation daunomycinone using ethylene glycol to form the dioxolane derivative (Compound IV). This is followed by selective sulfonation at the C-4 position using sulfonyl chlorides or anhydrides in the presence of a base. The resulting sulfonate (Compound V) is then subjected to the key catalytic step in a suitable solvent such as dimethylformamide or dioxane. Detailed standardized synthesis steps see the guide below.
- Preparation of the sulfonate precursor (Compound V) via selective sulfonation of the C-4 hydroxyl group on the protected daunomycinone scaffold.
- Execution of the key catalytic step using a Palladium or Nickel catalyst with phosphine ligands under reductive conditions (formic acid/amine) or carbonylation atmosphere.
- Final deprotection and functionalization involving acid hydrolysis to remove the 13-ketone protecting group and introduce the 7-alpha-hydroxyl moiety.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this catalytic technology offers tangible benefits that extend beyond mere chemical elegance. The primary value proposition lies in the drastic simplification of the supply chain for chiral starting materials. By leveraging naturally occurring daunomycinone, manufacturers eliminate the need for complex asymmetric synthesis or resolution processes, which are often the most expensive and time-consuming parts of API production. This "chiral pool" strategy ensures a consistent supply of enantiomerically pure intermediates, reducing the risk of batch failures due to stereochemical impurities. Moreover, the catalytic nature of the key transformation step means that expensive reagents are used in sub-stoichiometric amounts, directly lowering the bill of materials. The ability to operate at moderate temperatures and pressures further reduces energy consumption and allows for the use of standard glass-lined or stainless steel reactors, avoiding the need for specialized high-pressure equipment required in some alternative hydrogenation processes.
- Cost Reduction in Manufacturing: The elimination of optical resolution steps represents a massive saving in potential yield loss, effectively doubling the theoretical output from the starting material compared to racemic synthesis routes. Additionally, the use of catalytic amounts of palladium or nickel, combined with inexpensive hydride donors like formic acid, significantly lowers reagent costs compared to stoichiometric reducing agents. The high selectivity of the reaction minimizes the formation of by-products, which reduces the burden on downstream purification units such as chromatography columns or crystallization tanks, leading to lower solvent consumption and waste disposal costs. Overall, the streamlined process flow translates to a substantially reduced cost of goods sold (COGS) for the final anthracyclinone intermediate.
- Enhanced Supply Chain Reliability: Relying on natural product derivatives as starting materials provides a stable and predictable supply base, as these materials are produced via established fermentation processes globally. The robustness of the catalytic system, which tolerates a variety of sulfonyl leaving groups and ligands, ensures that production is not bottlenecked by the availability of a single specialized reagent. This flexibility allows procurement teams to source materials from multiple vendors, mitigating the risk of supply disruptions. Furthermore, the mild reaction conditions reduce the likelihood of equipment corrosion or safety incidents, ensuring continuous operation and reliable delivery schedules for downstream API manufacturers who depend on timely intermediate supplies.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing solvents and conditions that are compatible with large-scale batch processing. The high atom economy of the catalytic step and the avoidance of stoichiometric heavy metal reagents align with green chemistry principles, reducing the environmental footprint of the manufacturing facility. The simplified workup procedures, often involving straightforward extraction and crystallization rather than complex separations, facilitate easier scale-up from pilot plant to commercial production volumes. This compliance with environmental standards not only avoids regulatory hurdles but also enhances the corporate sustainability profile of the manufacturing organization, a key metric for modern pharmaceutical partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this anthracyclinone synthesis technology. These answers are derived directly from the experimental data and process descriptions provided in the patent literature, offering clarity on the feasibility and advantages of the method for industrial application.
Q: What is the primary advantage of using the sulfonate displacement method for anthracyclinone synthesis?
A: The primary advantage is the ability to utilize naturally occurring chiral pools, such as daunomycinone, thereby bypassing complex and costly optical resolution steps required in total synthesis while maintaining high stereochemical integrity at the C-7 and C-9 positions.
Q: Which catalyst systems are most effective for the reductive removal of the sulfonyl group?
A: Palladium and Nickel complexes coordinated with phosphine ligands, such as triphenylphosphine, 1,3-bis(diphenylphosphino)propane (DPPP), or ferrocenyl diphosphines, demonstrate superior activity. These systems facilitate efficient turnover under mild thermal conditions (60°C to 90°C).
Q: How does this process impact the impurity profile compared to traditional methods?
A: By employing selective sulfonation followed by clean catalytic displacement, the process minimizes side reactions associated with harsh acidic or basic conditions found in older methodologies. This results in a cleaner crude profile, simplifying downstream purification and enhancing overall yield.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Demethoxydaunomycinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of life-saving oncology therapies. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to market supply is seamless and efficient. We are committed to delivering 4-demethoxydaunomycinone and related anthracyclinone intermediates with stringent purity specifications, supported by our rigorous QC labs that employ advanced analytical techniques to verify identity and potency. Our state-of-the-art manufacturing facilities are equipped to handle the specific requirements of transition metal catalysis, including inert atmosphere handling and precise temperature control, guaranteeing batch-to-batch consistency.
We invite potential partners to engage with our technical procurement team to discuss how this advanced catalytic route can optimize your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this semi-synthetic methodology. We encourage you to contact us to obtain specific COA data and route feasibility assessments tailored to your project needs, ensuring that your development timelines are met with the highest standards of quality and reliability.
