Scalable Copper-Catalyzed Synthesis of 2-Aryl Benzotriazole Intermediates for Global Markets
Introduction to Advanced Benzotriazole Synthesis
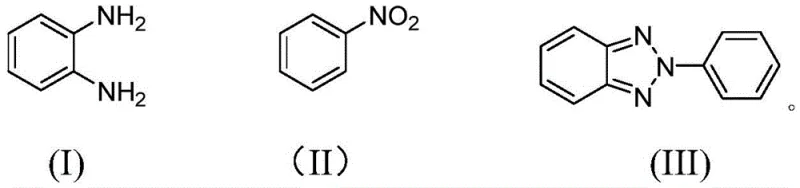
The landscape of heterocyclic chemistry is constantly evolving, driven by the demand for more efficient and sustainable manufacturing processes for key pharmaceutical and material science intermediates. A significant breakthrough in this domain is detailed in patent CN111100082A, which discloses a robust preparation method for 2-aryl benzotriazole compounds. This class of molecules serves as a critical molecular backbone in various applications, ranging from active pharmaceutical ingredients (APIs) to organic electronic materials and ultraviolet stabilizers. The disclosed technology addresses long-standing challenges in the field by utilizing a copper-catalyzed system that couples readily available o-phenylenediamines with aryl nitrogen sources, specifically nitroarenes. This approach not only simplifies the synthetic route but also enhances the overall economic viability of producing these high-value intermediates, making it a compelling option for industrial scale-up.
For R&D directors and process chemists, the significance of this patent lies in its ability to bypass the complex selectivity issues inherent in benzotriazole N-arylation. Traditionally, controlling the formation of the 2-aryl isomer versus the 1-aryl isomer has been a major hurdle, often requiring tedious separation steps or expensive directing groups. The method described in CN111100082A leverages a specific combination of a copper-containing catalyst, a strong alkaline environment, and a polar organic solvent to drive the reaction towards the desired 2-aryl product with high fidelity. This level of control is paramount for ensuring the purity profiles required in regulated industries, thereby positioning this technology as a cornerstone for future manufacturing strategies in the fine chemical sector.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-aryl benzotriazoles has been plagued by several intrinsic drawbacks that hinder large-scale commercial adoption. Conventional routes often rely on the use of pre-functionalized azobenzenes containing nitro, amino, or azide groups, which necessitate multi-step preparation and increase the overall cost of goods. Furthermore, many established protocols utilize sodium azide as a nitrogen source; while effective, sodium azide is highly toxic and poses significant safety risks during handling and storage, complicating regulatory compliance and workplace safety protocols. Another major limitation involves the reliance on noble metal catalysts such as rhodium and silver to achieve cyclization. These metals are not only prohibitively expensive but also subject to volatile market pricing, introducing financial instability into the supply chain. Additionally, the low selectivity of N-arylation in traditional methods often results in mixtures of 1H- and 2H-benzotriazole tautomers, requiring energy-intensive purification processes that further erode profit margins.
The Novel Approach
In stark contrast to these legacy methods, the novel approach outlined in the patent utilizes a direct coupling strategy between o-phenylenediamine compounds and nitroarenes. This methodology eliminates the need for hazardous azide reagents and expensive noble metals, replacing them with inexpensive copper catalysts and commercially abundant nitro compounds. The reaction proceeds through a tandem sequence of intermolecular N-N coupling followed by intramolecular cyclization, all facilitated within a strong alkaline environment. This streamlined process drastically reduces the number of unit operations required, translating to lower operational expenditures and a smaller environmental footprint. The use of simple starting materials like nitrobenzene and o-phenylenediamine ensures a stable and reliable supply chain, as these commodities are produced globally in massive quantities. By shifting the paradigm from complex, hazard-laden syntheses to a straightforward copper-catalyzed coupling, this technology offers a clear pathway for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Copper-Catalyzed N-N Coupling
The success of this synthetic route hinges on the intricate interplay between the copper catalyst, the strong base, and the solvent system. Mechanistically, the reaction is believed to initiate with the activation of the o-phenylenediamine by the copper species, forming a reactive intermediate capable of engaging with the nitro group of the aryl nitrogen source. Under the promotion of a strong base like potassium hydroxide, the system facilitates the initial intermolecular N-N bond formation. This is followed by a crucial intramolecular cyclization step that closes the triazole ring. The choice of tetrahydrofuran (THF) as the solvent is critical, as its polarity and coordinating ability likely stabilize the transition states involved in the copper catalytic cycle. The reaction conditions, typically maintained around 90 °C for 24 hours, provide sufficient thermal energy to overcome activation barriers without promoting decomposition of sensitive functional groups. This balance allows for the efficient conversion of starting materials into the target heterocycle with minimal byproduct formation.
From an impurity control perspective, the high selectivity of this method is particularly noteworthy. The specific catalytic system appears to favor the formation of the 2-aryl isomer exclusively, avoiding the generation of the 1-aryl regioisomer which is a common contaminant in other methods. This regioselectivity is vital for downstream applications where isomeric purity can dictate biological activity or material performance. Furthermore, the protocol demonstrates remarkable tolerance towards various substituents on the aromatic rings. As illustrated in the substrate scope studies, electron-withdrawing groups such as bromo and chloro, as well as bulky groups like tert-butyl, are well-tolerated. This suggests that the catalytic cycle is robust enough to handle steric and electronic variations without compromising yield, making it a versatile platform for synthesizing a diverse library of benzotriazole derivatives for drug discovery and material optimization.
How to Synthesize 2-Aryl Benzotriazole Efficiently
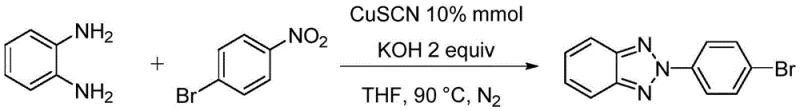
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to the reaction parameters defined in the patent to ensure optimal outcomes. The process is designed to be operationally simple, involving the mixing of solid and liquid reagents in a standard reaction vessel followed by heating under an inert atmosphere. The key to success lies in maintaining the correct stoichiometric ratios, particularly the excess of the aryl nitrogen source and the precise loading of the copper catalyst. While the general procedure is straightforward, scaling up the reaction necessitates rigorous control over temperature and mixing to manage the exothermic nature of the coupling steps. For those looking to adopt this technology, the following guide outlines the standardized steps derived from the exemplary embodiments, providing a clear roadmap for execution.
- Combine o-phenylenediamine, aryl nitrogen source (nitro compound), CuSCN catalyst, and KOH base in a reaction vessel under nitrogen atmosphere.
- Add tetrahydrofuran (THF) as the solvent and stir the mixture at 90 °C for 24 hours to facilitate coupling and cyclization.
- Cool the reaction mixture, filter, concentrate under reduced pressure, and purify the crude product via column chromatography to obtain the target compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this copper-catalyzed methodology represents a strategic opportunity to optimize both cost structures and supply reliability. The primary driver for cost reduction is the substitution of expensive noble metal catalysts with commodity-grade copper salts. This switch not only lowers the direct material cost but also simplifies the removal of metal residues from the final product, a critical step in API manufacturing that often requires specialized scavenging resins. Additionally, the use of non-hazardous nitroarenes instead of sodium azide reduces the costs associated with safety infrastructure, waste disposal, and regulatory compliance. The simplicity of the workup procedure, which involves basic filtration and concentration, further contributes to operational efficiency by reducing solvent consumption and processing time. These factors collectively enhance the gross margin potential for products manufactured using this route.
Enhanced supply chain reliability is another significant benefit derived from the choice of raw materials. Both o-phenylenediamines and nitroarenes are bulk chemicals produced by numerous suppliers worldwide, mitigating the risk of single-source dependency. This abundance ensures that production schedules are less likely to be disrupted by raw material shortages, a common issue with specialized reagents. Moreover, the robustness of the reaction conditions allows for flexible manufacturing; the process can be adapted to different batch sizes without significant re-optimization, facilitating a smoother transition from kilogram-scale development to multi-ton commercial production. The environmental compliance aspect is also improved, as the process avoids the generation of heavy metal waste streams associated with rhodium or silver catalysts, aligning with increasingly stringent global sustainability mandates and reducing the burden on wastewater treatment facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and technical disclosures within the patent documentation, aiming to clarify the practical implications for potential adopters. Understanding these nuances is essential for evaluating the feasibility of integrating this method into existing production lines or new product development pipelines. The answers reflect the specific advantages observed in the reported examples, providing a realistic expectation of performance and scalability.
Q: What are the advantages of this copper-catalyzed method over traditional rhodium or silver catalysis?
A: This method utilizes inexpensive copper catalysts (e.g., CuSCN) instead of costly noble metals like rhodium or silver, significantly reducing raw material costs while maintaining high selectivity for the 2-aryl isomer.
Q: Does this synthesis route tolerate sensitive functional groups like halogens?
A: Yes, the protocol demonstrates excellent functional group tolerance, successfully accommodating substrates with bromo, chloro, and alkynyl groups without dehalogenation or side reactions.
Q: Is the use of hazardous azide reagents required for this transformation?
A: No, unlike traditional methods that often rely on hazardous sodium azide, this process uses commercially available and safer nitroarenes as the nitrogen source, enhancing operational safety.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Aryl Benzotriazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced synthetic methodologies like the copper-catalyzed route for 2-aryl benzotriazoles. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive consistent quality regardless of volume. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of intermediate adheres to the highest industry standards. We understand that the transition from lab-scale innovation to commercial reality requires more than just a recipe; it demands a partner with deep technical expertise and a commitment to excellence in process safety and efficiency.
We invite you to collaborate with us to leverage this cutting-edge technology for your specific application needs. Whether you require custom synthesis for drug development or large-scale manufacturing for material production, our team is ready to provide a Customized Cost-Saving Analysis tailored to your project. We encourage you to reach out to our technical procurement team to request specific COA data and route feasibility assessments. By partnering with us, you gain access to a reliable supply chain and a wealth of chemical knowledge, empowering you to bring your innovative products to market faster and more economically.
