Advanced Catalytic Routes for High-Purity Halogenated Aminomethyl Pyridines and Commercial Scalability
Introduction to Advanced Pyridine Functionalization Technologies
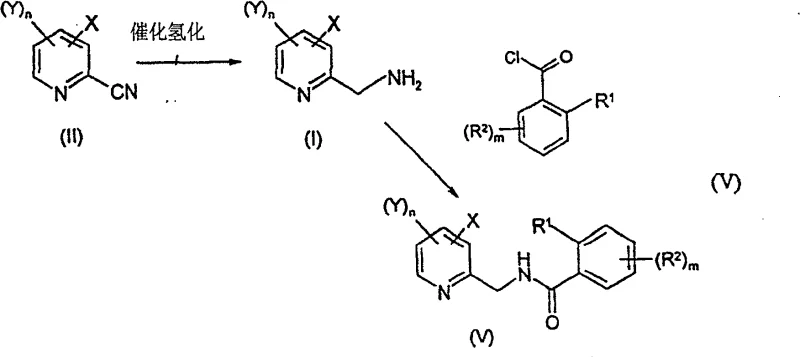
The chemical industry's demand for high-purity heterocyclic intermediates, particularly halogenated aminomethyl pyridines, has surged due to their critical role in the synthesis of next-generation agrochemicals and pharmaceuticals. Patent CN100564360C introduces a transformative methodology for preparing compounds of general formula (I), specifically focusing on 2-aminomethyl-3-chloro-5-trifluoromethylpyridine, a key building block for potent fungicides. This intellectual property delineates a robust two-stage synthetic strategy that begins with an innovative aqueous cyanation to form the nitrile intermediate (Formula II), followed by a highly selective catalytic hydrogenation to yield the target amine. By shifting away from traditional stoichiometric heavy metal reagents and harsh reaction conditions, this technology offers a compelling value proposition for manufacturers seeking to optimize both environmental compliance and production economics. The integration of phase-transfer catalysis in aqueous media coupled with controlled low-pressure hydrogenation represents a significant leap forward in process chemistry, addressing long-standing challenges regarding waste generation and selectivity in heterocyclic synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the introduction of a cyano group onto the pyridine ring, a prerequisite for generating the aminomethyl functionality, has relied heavily on the Rosenmund-von Braun reaction or variations thereof involving transition metal cyanides. These traditional protocols typically necessitate the use of copper(I) cyanide or nickel-based catalysts in high-boiling polar aprotic solvents such as dimethylformamide (DMF) or dimethyl sulfoxide (DMSO) at elevated temperatures exceeding 120°C. Such conditions present severe drawbacks for industrial application, including the generation of toxic heavy metal-containing liquid effluents that require complex and costly wastewater treatment infrastructure. Furthermore, the recovery of high-boiling solvents is energy-intensive, often leading to thermal degradation of sensitive intermediates and reduced overall yields. Additionally, alternative routes involving N-oxide activation or N-alkyl pyridine precursors introduce multiple synthetic steps, increasing material throughput and operational complexity, which ultimately inflates the cost of goods sold and extends the manufacturing lead time for critical intermediates.
The Novel Approach
In stark contrast to these legacy methodologies, the novel approach detailed in the patent data utilizes a direct nucleophilic aromatic substitution facilitated by phase-transfer catalysis in an aqueous or solvent-free environment. This method treats a halopyridine precursor (Formula III), such as 3-chloro-2-fluoro-5-trifluoromethylpyridine, with a simple cyanide source like potassium cyanide in the presence of quaternary ammonium salts. This eliminates the need for toxic heavy metal reagents entirely, replacing them with benign alkali metal salts that generate manageable inorganic waste streams. Following the cyanation, the subsequent conversion to the amine is achieved via catalytic hydrogenation under remarkably mild conditions—typically between 20°C and 30°C and at low hydrogen pressures of 1 to 4 atmospheres. This gentle approach not only preserves sensitive halogen substituents that would otherwise be lost through hydrodehalogenation but also allows for the direct isolation of the product as a stable salt, streamlining the downstream purification process and enhancing the overall atom economy of the synthesis.
Mechanistic Insights into Phase-Transfer Cyanation and Selective Hydrogenation
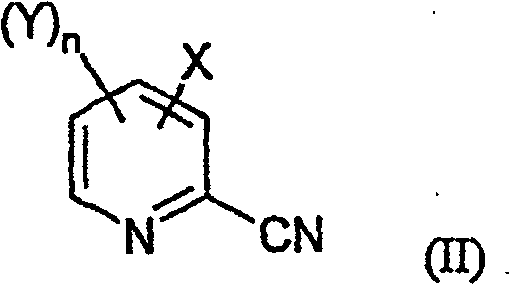
The core innovation of this synthetic route lies in the mechanistic efficiency of the phase-transfer catalyzed cyanation and the chemoselective hydrogenation steps. In the first stage, the phase-transfer catalyst, such as Aliquat 336 or tetrabutylammonium bromide, acts as a molecular shuttle, transporting the cyanide anion from the aqueous phase into the organic phase or to the interface where the halopyridine substrate resides. This dramatically enhances the nucleophilicity of the cyanide ion, allowing the displacement of the fluorine atom on the pyridine ring to proceed rapidly at moderate temperatures (20-40°C) without the need for aggressive activators. The absence of heavy metal coordination complexes means the reaction pathway is cleaner, with fewer side products arising from metal-mediated coupling or decomposition. This mechanistic simplicity translates directly to higher purity profiles for the resulting 2-cyanopyridine intermediate (Formula II), reducing the burden on subsequent purification units.
Regarding the hydrogenation mechanism, the preservation of the chlorine atom during the reduction of the nitrile group is paramount, as dehalogenation is a common competing side reaction with palladium catalysts. The patent specifies the crucial role of adding a strong acid, such as hydrochloric acid, to the alcoholic solvent system. Mechanistically, the acid serves a dual purpose: it protonates the nascent amine product immediately upon formation, converting it into a non-coordinating ammonium salt that does not poison the palladium active sites. Simultaneously, the acidic environment suppresses the oxidative addition of the aryl-chlorine bond to the metal center, effectively shutting down the hydrodehalogenation pathway. This precise control over the catalyst surface chemistry ensures that the reduction is exclusive to the cyano group, yielding the desired 2-aminomethyl-3-chloro-5-trifluoromethylpyridine with exceptional selectivity and minimal formation of dechlorinated impurities, which is critical for meeting the stringent quality specifications required in agrochemical active ingredient synthesis.
How to Synthesize 2-Aminomethyl-3-chloro-5-trifluoromethylpyridine Efficiently
The execution of this synthesis requires careful attention to reagent stoichiometry and phase management to maximize yield and safety. The process begins with the cyanation step where the halopyridine is stirred with an aqueous cyanide solution and a catalytic amount of phase-transfer agent, followed by separation of the organic layer and distillation to isolate the nitrile. The subsequent hydrogenation is performed by suspending the nitrile in methanol with concentrated hydrochloric acid and adding a palladium on carbon catalyst under a hydrogen atmosphere. While the general principles are straightforward, maintaining the correct acid-to-substrate ratio and monitoring hydrogen uptake are critical for preventing over-reduction or catalyst deactivation. For detailed operational parameters, safety protocols, and specific workup procedures required to replicate this high-efficiency route in a GMP-compliant environment, please refer to the standardized synthesis guide below.
- Perform nucleophilic substitution of 3-chloro-2-fluoro-5-trifluoromethylpyridine with potassium cyanide in water using a phase-transfer catalyst like Aliquat 336 at 20-40°C.
- Isolate the resulting 3-chloro-2-cyano-5-trifluoromethylpyridine intermediate via phase separation and distillation.
- Subject the nitrile intermediate to catalytic hydrogenation using palladium on carbon in methanol with hydrochloric acid at low pressure (1-4 atm) to yield the final amine salt.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented technology offers profound strategic advantages beyond mere technical feasibility. The elimination of copper and nickel reagents removes a significant regulatory and disposal burden, directly correlating to lower operational expenditures related to hazardous waste management. Furthermore, the shift from high-boiling organic solvents to water or solvent-free conditions drastically reduces the energy consumption associated with solvent recovery and distillation, contributing to a smaller carbon footprint and alignment with modern sustainability goals. The mild reaction conditions also imply that standard glass-lined or stainless steel reactors can be utilized without the need for specialized high-pressure or high-temperature equipment, lowering the barrier to entry for contract manufacturing organizations and enhancing the overall resilience of the supply chain against equipment bottlenecks.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the simplification of the raw material portfolio and the reduction of downstream processing costs. By replacing expensive and toxic heavy metal cyanides with commodity alkali metal cyanides, the direct material costs are significantly lowered. Moreover, the ability to isolate the final amine directly as a hydrochloride salt eliminates the need for a separate salt formation step and the associated solvent usage, effectively collapsing two unit operations into one. The avoidance of difficult-to-remove polar solvents like DMSO further reduces utility costs, as the energy required for solvent stripping is minimized, leading to substantial overall cost savings in the manufacturing of complex pyridine intermediates.
- Enhanced Supply Chain Reliability: From a sourcing perspective, the reagents utilized in this protocol—potassium cyanide, quaternary ammonium salts, and palladium on carbon—are widely available commodities with stable global supply chains, unlike specialized organometallic catalysts which may face availability fluctuations. The robustness of the aqueous cyanation step ensures consistent batch-to-batch quality, reducing the risk of production delays caused by out-of-specification intermediates. Additionally, the mild operating conditions reduce the wear and tear on production assets, decreasing maintenance downtime and ensuring a more predictable delivery schedule for downstream customers who rely on just-in-time inventory models for their own active ingredient production.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies safety and environmental risks, but this methodology is inherently designed for technical scale operation. The use of water as a solvent in the cyanation step provides excellent heat dissipation properties, mitigating the risk of thermal runaways that are common in exothermic nucleophilic substitutions. The low-pressure hydrogenation (1-4 atm) can be safely conducted in standard hydrogenation reactors without requiring ultra-high-pressure vessels, simplifying the engineering requirements for scale-up. Furthermore, the aqueous waste streams generated are free of heavy metals and organic solvents, making them far easier to treat in standard effluent treatment plants, thereby ensuring compliance with increasingly stringent environmental regulations across different jurisdictions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. Understanding the nuances of catalyst selection, impurity profiles, and scalability is essential for R&D teams evaluating this technology for potential technology transfer or process optimization projects. The answers provided are derived directly from the experimental data and claims within the patent literature, ensuring accuracy and relevance for technical decision-makers.
Q: How does this method avoid heavy metal contamination compared to traditional Rosenmund-von Braun reactions?
A: Unlike conventional methods utilizing stoichiometric copper(I) cyanide or nickel catalysts which generate toxic heavy metal waste streams, this patented process employs alkali metal cyanides (KCN/NaCN) with phase-transfer catalysts in aqueous media, significantly simplifying effluent treatment and reducing environmental compliance costs.
Q: What ensures the selectivity of the hydrogenation step to prevent dehalogenation?
A: The process utilizes a specific combination of palladium on carbon catalyst in an acidic alcoholic solvent (methanol/HCl). The presence of strong acid protonates the amino product immediately, preventing catalyst poisoning and suppressing competitive hydrodehalogenation side reactions, thereby preserving the critical chlorine substituent.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the method is designed for technical scale application. It operates at moderate temperatures (20-60°C) and low hydrogen pressures (1-4 atm), utilizing water as a primary solvent for the cyanation step, which eliminates the need for expensive, high-boiling polar aprotic solvents and facilitates easier downstream processing and scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Aminomethyl-3-chloro-5-trifluoromethylpyridine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising metrics observed in patent literature are faithfully reproduced in our facilities. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch of 2-aminomethyl-3-chloro-5-trifluoromethylpyridine meets the exacting standards required for agrochemical active ingredient synthesis. Our commitment to quality assurance ensures that our clients receive intermediates that facilitate smooth downstream processing and consistent final product performance.
We invite procurement leaders and R&D directors to engage with us to explore how this advanced synthetic route can be integrated into your supply chain to drive efficiency and reduce costs. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic impact of switching to this greener, more efficient methodology. We encourage you to contact our technical procurement team today to obtain specific COA data for our current stock and to discuss route feasibility assessments tailored to your specific volume requirements and timeline constraints.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
