Scalable Synthesis of 3-Dehydroshikimic Esters: A Breakthrough in Renewable Fine Chemical Manufacturing
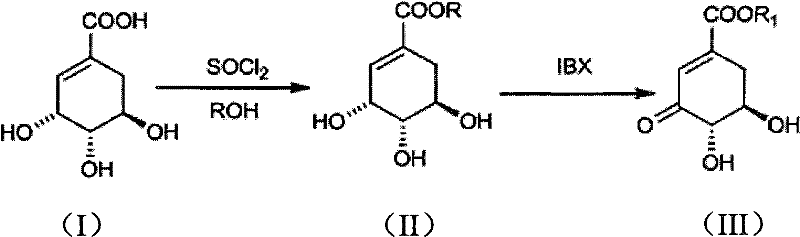
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable pathways to access high-value hydroaromatic intermediates, particularly those derived from renewable biomass sources. Patent CN101973874A introduces a transformative preparation method for 3-dehydroshikimic ester compounds, addressing critical bottlenecks in the synthesis of key antioxidants and pharmaceutical precursors. This technology leverages shikimic acid, a naturally occurring compound found in star anise, as a foundational building block, converting it through a streamlined two-step chemical process involving esterification and selective oxidation. By shifting away from complex biological fermentation or hazardous diazomethane-based protocols, this method offers a compelling value proposition for reliable pharmaceutical intermediate suppliers aiming to optimize their production portfolios. The core innovation lies in the utilization of thionyl chloride for efficient esterification followed by the precise oxidative dehydrogenation using 2-iodoxybenzoic acid (IBX), a reagent known for its chemoselectivity and operational safety. This approach not only enhances the overall yield but also significantly simplifies the purification workflow, making it highly attractive for commercial scale-up of complex intermediates intended for the synthesis of valuable downstream products like protocatechuic acid, vanillin, and sialic acid.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of 3-dehydroshikimic acid and its esters has been plagued by significant technical and economic hurdles that hinder widespread industrial adoption. Traditional biological fermentation processes, while theoretically green, often suffer from low volumetric productivity, complex downstream separation challenges, and strict sterility requirements that drive up capital expenditure. Furthermore, alternative chemical routes relying on diazomethane for esterification present severe safety liabilities due to the explosive and highly toxic nature of diazomethane, necessitating specialized low-temperature equipment and rigorous safety protocols that are difficult to maintain in large-scale facilities. These conventional methods also frequently struggle with inconsistent batch-to-batch reproducibility and limited substrate scope, restricting the ability to produce diverse ester derivatives efficiently. The reliance on specific enzymatic pathways or hazardous reagents creates a fragile supply chain vulnerable to disruptions, ultimately inflating the cost of goods sold and limiting the availability of these critical intermediates for the broader fine chemical manufacturing sector.
The Novel Approach
In stark contrast, the methodology disclosed in CN101973874A represents a paradigm shift towards practical, safe, and economically viable synthesis. By employing thionyl chloride as an acylating agent, the process achieves rapid and high-yielding esterification under mild thermal conditions, effectively bypassing the need for dangerous diazomethane. The subsequent oxidation step utilizes IBX, a hypervalent iodine reagent that operates effectively in common organic solvents like tetrahydrofuran at temperatures ranging from ambient to moderately elevated levels. This novel approach eliminates the need for cryogenic conditions and exotic catalysts, drastically reducing energy consumption and equipment complexity. The result is a streamlined workflow that facilitates cost reduction in fine chemical manufacturing by minimizing unit operations and waste generation. Moreover, the versatility of this method allows for the facile preparation of various alkyl esters (methyl, ethyl, dodecyl), providing manufacturers with the flexibility to tailor intermediates for specific downstream applications without retooling entire production lines.
Mechanistic Insights into IBX-Mediated Oxidative Dehydrogenation
The heart of this synthetic strategy lies in the precise mechanistic execution of the oxidative dehydrogenation step, which converts the saturated shikimate ester into the unsaturated 3-dehydroshikimic ester. The reaction initiates with the activation of the secondary hydroxyl group at the C-3 position by the hypervalent iodine center of the IBX molecule. This interaction forms a transient alkoxy-iodinane intermediate, which subsequently undergoes an elimination reaction to generate the desired alpha,beta-unsaturated carbonyl system. The chemoselectivity of IBX is paramount here; it preferentially oxidizes the allylic or benzylic-like hydroxyl groups while leaving other sensitive functionalities, such as the ester moiety and remaining hydroxyl groups, largely intact under optimized conditions. This specificity is crucial for maintaining the structural integrity of the shikimate scaffold, preventing over-oxidation or degradation that could lead to complex impurity profiles difficult to separate. The use of polar aprotic solvents like THF further stabilizes the transition states involved, ensuring smooth conversion kinetics even at relatively low catalyst loadings.
From an impurity control perspective, this mechanism offers distinct advantages over metal-catalyzed oxidations which often leave behind trace heavy metal residues requiring expensive scavenging steps. The byproducts of the IBX oxidation are primarily reduced iodine species which are easily removed via simple filtration or aqueous workup, contributing to a cleaner final product. The reaction conditions, typically maintained between 0°C and 70°C, allow for fine-tuning of the reaction rate to minimize side reactions such as polymerization or isomerization. Understanding these mechanistic nuances enables process chemists to optimize parameters like stoichiometry and addition rates, ensuring that the high-purity 3-dehydroshikimic ester targets are met consistently. This level of control is essential for meeting the rigorous regulatory standards demanded by the pharmaceutical industry, where impurity thresholds are strictly defined and monitored.
How to Synthesize 3-Dehydroshikimic Ester Efficiently
Implementing this synthesis route in a production environment requires careful attention to reaction parameters to maximize efficiency and safety. The process begins with the esterification of shikimic acid, where controlling the exotherm during thionyl chloride addition is critical to prevent degradation. Following the isolation of the shikimate ester, the oxidation step demands precise temperature management to balance reaction speed with selectivity. The detailed standardized synthesis steps below outline the optimal conditions derived from the patent data to ensure reproducible results.
- Perform esterification of shikimic acid with alcohol (e.g., methanol or ethanol) using thionyl chloride (SOCl2) as the acylating reagent at controlled temperatures between 0°C and 65°C.
- Dissolve the resulting shikimate ester in an organic solvent such as tetrahydrofuran (THF) and react with 2-iodoxybenzoic acid (IBX) at temperatures ranging from 0°C to 70°C.
- Filter the reaction mixture to remove solids, concentrate the filtrate, and purify the crude product via recrystallization using ethyl acetate and petroleum ether to obtain high-purity crystals.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthesis route translates into tangible strategic benefits that extend beyond mere technical feasibility. The shift to a fully chemical synthesis driven by renewable biomass feedstocks decouples production from the volatility of fermentation yields and the logistical nightmares associated with hazardous reagents like diazomethane. This stability is a cornerstone for building a resilient supply chain capable of meeting the demanding timelines of global pharmaceutical clients. By simplifying the operational footprint, manufacturers can achieve faster turnaround times and more predictable delivery schedules, directly addressing the pain point of reducing lead time for high-purity intermediates. Furthermore, the elimination of heavy metal catalysts and toxic gases aligns with increasingly stringent environmental regulations, reducing the burden of waste disposal and compliance costs.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the substitution of expensive and hazardous reagents with cost-effective alternatives like thionyl chloride and IBX. The mild reaction conditions eliminate the need for energy-intensive cryogenic cooling systems, leading to substantial savings in utility costs. Additionally, the high selectivity of the oxidation step reduces the formation of byproducts, thereby increasing the overall mass balance and reducing the volume of raw materials required per kilogram of finished product. The simplified purification protocol, relying on standard recrystallization rather than complex chromatography, further drives down processing costs, making the final pharmaceutical intermediate more price-competitive in the global market.
- Enhanced Supply Chain Reliability: Sourcing shikimic acid from renewable plant sources like star anise provides a sustainable and scalable raw material base that is less susceptible to the geopolitical risks associated with petrochemical feedstocks. The robustness of the chemical synthesis ensures consistent output regardless of biological variables that often plague fermentation-based supply chains. This reliability allows procurement teams to negotiate long-term contracts with greater confidence, knowing that production capacity can be ramped up quickly to meet surges in demand without compromising quality. The use of common solvents and reagents also mitigates the risk of supply disruptions for specialized chemicals, ensuring continuous operation.
- Scalability and Environmental Compliance: The process is inherently designed for scale, utilizing unit operations such as filtration, concentration, and crystallization that are standard in multi-ton manufacturing facilities. The absence of toxic diazomethane removes a major safety barrier to scaling, allowing for larger batch sizes without exponential increases in safety infrastructure costs. From an environmental standpoint, the generation of benign byproducts and the potential for solvent recovery contribute to a greener manufacturing profile. This alignment with green chemistry principles not only satisfies corporate sustainability goals but also future-proofs the supply chain against tightening environmental legislation, ensuring long-term operational viability.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and advantageous effects described in the patent literature, providing clarity on the practical aspects of producing 3-dehydroshikimic esters. Understanding these details is crucial for stakeholders evaluating the feasibility of integrating this route into their existing manufacturing frameworks.
Q: What are the primary advantages of using IBX for oxidizing shikimate esters compared to traditional methods?
A: The use of IBX (2-iodoxybenzoic acid) offers distinct advantages over traditional biological fermentation or hazardous diazomethane methods. It operates under mild reaction conditions (0-70°C), eliminates the need for toxic diazomethane, and provides high selectivity for the 3-position hydroxyl group without requiring extreme low-temperature equipment, thereby simplifying the operational complexity and enhancing safety profiles for large-scale manufacturing.
Q: Is the raw material shikimic acid sustainable for long-term commercial production?
A: Yes, shikimic acid is derived from renewable non-crop biomass resources, specifically star anise, which ensures a steady and sustainable supply chain. Unlike petrochemical-derived intermediates, this biomass origin supports green chemistry initiatives and reduces dependency on fluctuating fossil fuel markets, making it an ideal candidate for long-term strategic sourcing in the fine chemical industry.
Q: How does this synthesis route impact the purity profile of the final antioxidant intermediates?
A: This synthetic route allows for rigorous impurity control through straightforward recrystallization steps using common solvents like ethyl acetate. The chemical specificity of the IBX oxidation minimizes side reactions common in enzymatic processes, resulting in a cleaner impurity profile that meets the stringent quality specifications required for downstream pharmaceutical applications such as the synthesis of sialic acid or gallic acid derivatives.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Dehydroshikimic Ester Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of next-generation therapeutics and fine chemicals. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which employ advanced analytical techniques to verify identity and assay. Our expertise in handling sensitive oxidation chemistries allows us to replicate the high yields and selectivity described in CN101973874A, providing our partners with a dependable source of this valuable intermediate.
We invite you to collaborate with us to leverage this advanced synthesis technology for your specific application needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this route can optimize your bill of materials. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to innovation and quality can support your supply chain objectives.
