Optimizing Veterinary Antibiotic Quality: A Deep Dive into the Novel Ceftiofur Hydrochloride Refining Technology
The global veterinary pharmaceutical landscape is increasingly demanding higher standards for antibiotic purity and stability, particularly for critical third-generation cephalosporins like ceftiofur hydrochloride. Patent CN116003439A introduces a groundbreaking refining method that addresses the persistent challenges of product degradation and impurity accumulation during storage and synthesis. This technology offers a robust solution for transforming unqualified or degraded crude products into high-purity active pharmaceutical ingredients (APIs) suitable for rigorous animal health applications. By leveraging a precise pH-controlled crystallization strategy, the process ensures the removal of color bodies and related substances that compromise drug efficacy. For procurement leaders and R&D directors, this patent represents a significant opportunity to enhance supply chain resilience by recovering value from off-spec materials while maintaining stringent quality benchmarks.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the purification of ceftiofur hydrochloride has been plagued by complex operational procedures that result in substantial material loss and inconsistent quality outcomes. Prior art, such as the method disclosed in patent CN106478669A, relies on a multi-step sequence involving dissolution, ion exchange or alkali treatment to remove hydrogen chloride, followed by a secondary conversion back to the hydrochloride salt. This convoluted approach necessitates two distinct crystallization steps, which inherently leads to a cumulative yield loss exceeding 10 percent, often resulting in final yields between 81.9 percent and 87.1 percent. Furthermore, these traditional methods frequently fail to adequately address specific impurities mandated by regulatory standards, such as the 2020 Veterinary Drug Dictionary, leaving residual contaminants like 4-ketoceftiofur or unknown molecular weight species that can affect the stability and safety of the final drug product. The extended production time and operational complexity also introduce greater risks of batch-to-batch variability, making it difficult for manufacturers to guarantee consistent supply for large-scale veterinary formulations.
The Novel Approach
In stark contrast, the refining method detailed in CN116003439A streamlines the purification workflow into a highly efficient four-step process that maximizes recovery while ensuring exceptional purity levels exceeding 99.7 percent. The innovation lies in the strategic use of a mixed solvent system comprising water and organic solvents like acetone or ethyl acetate, combined with a precise pH swing from weakly alkaline conditions to strong acidity. By initially adjusting the pH to a range of 5.5 to 6.5, the process facilitates effective decolorization using activated carbon without inducing significant degradation of the sensitive beta-lactam ring structure. Subsequent acidification to a pH of 0.6 to 0.8 triggers the selective precipitation of the pure hydrochloride salt, effectively excluding a wide spectrum of related substances including ceftiofur delta-3 isomers and N-deacetyl derivatives. This simplified approach not only drastically reduces processing time but also enhances the overall economic viability of the manufacturing process by minimizing solvent usage and maximizing the yield of saleable product from crude starting materials.
Mechanistic Insights into pH-Controlled Crystallization and Solvent Engineering
The core scientific advancement of this refining technique rests on the delicate manipulation of solubility differentials and ionization states of the ceftiofur molecule within a tailored solvent matrix. Ceftiofur hydrochloride possesses both acidic and basic functional groups, making its solubility highly dependent on the pH of the surrounding medium. The process begins by dissolving the crude material in a water-organic solvent mixture, where the addition of a mild alkali such as triethylamine or ammonia water adjusts the environment to a near-neutral pH of 5.5 to 6.5. At this specific pH range, the molecule exists in a state that allows for optimal interaction with activated carbon, facilitating the adsorption of colored impurities and high-molecular-weight byproducts without precipitating the active ingredient prematurely. This step is critical for ensuring the visual quality and stability of the final API, as color deepening is often an indicator of oxidative degradation or impurity presence in cephalosporins.
Following decolorization, the introduction of hydrochloric acid serves a dual purpose: it reprotonates the molecule to form the stable hydrochloride salt and drastically alters the solubility profile to induce crystallization. By controlling the temperature at 15-20°C and carefully titrating the pH down to 0.6-0.8, the process creates a supersaturated environment where only the purest form of ceftiofur hydrochloride nucleates and grows. This aggressive acidification ensures that impurities with different pKa values or solubility characteristics remain in the mother liquor, effectively purifying the solid phase. The choice of organic co-solvents, specifically the 3:1 volume ratio of acetone to ethyl acetate or methanol, further fine-tunes the dielectric constant of the solution, optimizing the crystal lattice formation for maximum purity and filterability. 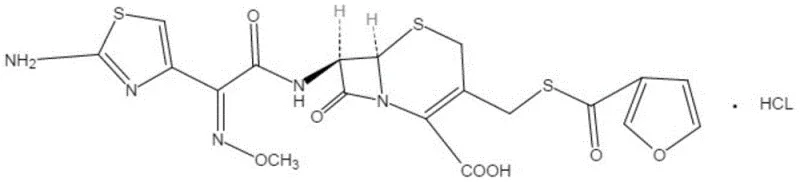
How to Synthesize Ceftiofur Hydrochloride Efficiently
The implementation of this refining protocol requires strict adherence to temperature and pH parameters to replicate the high yields and purity demonstrated in the patent examples. Operators must ensure that the dissolution and decolorization steps are maintained within the narrow 15-20°C window to prevent thermal degradation of the thermolabile beta-lactam core. The subsequent acidification must be performed dropwise to control the rate of nucleation, preventing the occlusion of impurities within the growing crystals. Detailed standardized operating procedures regarding solvent ratios, agitation speeds, and filtration techniques are essential for translating this laboratory-scale success into commercial production. The following guide outlines the critical operational phases required to achieve the specified quality attributes.
- Dissolve crude ceftiofur hydrochloride in a mixed solution of water and organic solvents such as acetone or ethyl acetate.
- Adjust the pH to 5.5-6.5 using alkali at 15-20°C and perform decolorization with activated carbon to remove impurities.
- Precipitate the solid by dropwise addition of hydrochloric acid, followed by further pH adjustment to 0.6-0.8 for crystal growth.
- Filter the crystals and dry under vacuum at 60°C to obtain the final purified ceftiofur hydrochloride product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this refining technology translates directly into enhanced operational efficiency and significant cost optimization opportunities across the manufacturing value chain. The ability to recover and upgrade degraded or off-spec crude materials reduces the dependency on purchasing expensive high-grade starting materials, thereby insulating the production budget from raw material price volatility. Moreover, the simplified workflow eliminates the need for specialized ion exchange resins and complex multi-stage crystallization setups, which reduces capital expenditure on equipment and lowers the overall maintenance burden of the production facility. The use of common, recyclable solvents further aligns with green chemistry initiatives, potentially reducing waste disposal costs and environmental compliance liabilities.
- Cost Reduction in Manufacturing: The process achieves a substantial reduction in manufacturing costs primarily through the elimination of yield-losing steps and the implementation of solvent recovery systems. Unlike previous methods that suffered from over 10 percent material loss, this technique consistently delivers yields above 90 percent, meaning more saleable product is generated from the same amount of input material. Additionally, the solvents used, such as acetone and ethyl acetate, can be easily separated from the aqueous phase and distilled for reuse, creating a closed-loop system that minimizes fresh solvent purchases. This efficiency gain allows manufacturers to offer more competitive pricing for the final veterinary API while maintaining healthy profit margins.
- Enhanced Supply Chain Reliability: By simplifying the production process and utilizing widely available chemical reagents, the risk of supply chain disruptions is significantly mitigated. The reliance on commodity solvents rather than specialized proprietary reagents ensures that production can continue even if specific niche suppliers face shortages. Furthermore, the robustness of the method in handling crude materials of varying initial quality provides a buffer against inconsistencies in upstream synthesis, ensuring a steady flow of high-purity finished goods to downstream formulation partners. This reliability is crucial for meeting the continuous demand for animal health products in the global market.
- Scalability and Environmental Compliance: The refining method is inherently scalable, utilizing standard unit operations such as mixing, filtration, and vacuum drying that are easily replicated from pilot plant to full commercial scale. The controlled temperature conditions and absence of hazardous catalysts make the process safer to operate and easier to validate under Good Manufacturing Practice (GMP) guidelines. From an environmental perspective, the reduced solvent consumption and the ability to recycle process liquids contribute to a lower carbon footprint and reduced hazardous waste generation, supporting corporate sustainability goals and regulatory compliance in increasingly strict environmental jurisdictions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this novel refining technology. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation to provide clarity for potential partners and stakeholders. Understanding these details is essential for evaluating the feasibility of integrating this process into existing manufacturing lines.
Q: How does the new refining method improve upon previous techniques like CN106478669A?
A: The new method simplifies the process by eliminating complex ion exchange resin steps and double crystallization, resulting in significantly higher yields (over 90% vs 81-87%) and better removal of specific impurities mandated by veterinary standards.
Q: What specific impurities does this purification process target?
A: The process effectively removes ten critical impurities including ceftiofur oxime ethyl ester, 7-aminocephalosporanic acid (7-ACA), 2-furancarboxylic acid, and various isomers, ensuring compliance with the 2020 Veterinary Drug Dictionary standards.
Q: Is the solvent system used in this method cost-effective for large-scale production?
A: Yes, the method utilizes common industrial solvents like acetone, ethanol, and ethyl acetate, which can be easily recovered and recycled through simple distillation, thereby substantially lowering raw material costs and environmental impact.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ceftiofur Hydrochloride Supplier
As the demand for high-quality veterinary antibiotics continues to rise, partnering with a technically proficient manufacturer is essential for ensuring product efficacy and regulatory compliance. NINGBO INNO PHARMCHEM stands at the forefront of this industry, leveraging advanced purification technologies like the one described in CN116003439A to deliver ceftiofur hydrochloride of exceptional purity and stability. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements without compromising on our stringent purity specifications. With our rigorous QC labs and commitment to process optimization, we guarantee a supply of veterinary APIs that meet the highest international standards for animal health applications.
We invite you to collaborate with us to explore how this advanced refining method can optimize your supply chain and reduce your overall manufacturing costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific production needs. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our capabilities can support your long-term business goals in the competitive veterinary pharmaceutical market.
