Advanced Synthesis of Baloxavir Marboxil Isomer for Pharmaceutical Quality Control
Advanced Synthesis of Baloxavir Marboxil Isomer for Pharmaceutical Quality Control
The pharmaceutical industry continuously demands higher purity standards and more efficient synthetic routes for critical antiviral agents like Baloxavir Marboxil. Patent CN116375734A introduces a groundbreaking methodology for the preparation of a specific Baloxavir dipivoxil isomer, addressing the critical need for high-quality reference standards in drug development. This innovation is particularly significant for R&D teams focused on impurity profiling, pharmacokinetic studies, and toxicological assessments, where the availability of single-chirality isomers is paramount. By leveraging a sophisticated asymmetric transfer hydrogenation strategy, this process bypasses the traditional bottlenecks associated with racemic mixtures, offering a streamlined pathway that enhances both yield and stereochemical integrity. For global supply chain leaders, this represents a shift towards more robust and predictable manufacturing protocols for complex pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral intermediates for Baloxavir analogues has been plagued by the formation of racemic mixtures, necessitating cumbersome and inefficient chiral resolution steps. Conventional reduction methods typically yield a 1:1 mixture of R and S configurations, forcing manufacturers to discard nearly half of their product or engage in expensive chromatographic separations to isolate the desired enantiomer. This not only drastically reduces the overall atom economy but also introduces significant variability in batch-to-batch consistency. Furthermore, the reliance on resolution often requires specialized chiral columns or resolving agents that are costly and difficult to source at scale, creating bottlenecks in the supply chain for high-purity API intermediates. These inefficiencies translate directly into higher production costs and extended lead times, which are unacceptable in the fast-paced environment of antiviral drug development.
The Novel Approach
In stark contrast, the methodology disclosed in CN116375734A employs a direct asymmetric synthesis strategy that establishes the desired stereochemistry at an early stage, effectively rendering chiral resolution obsolete. The process initiates with readily available 3-fluoro-2-methylbenzoic acid, proceeding through a series of well-defined transformations including esterification, bromination, and sulfur substitution to construct the dibenzothiepinone core. The pivotal innovation lies in the asymmetric reduction step, which selectively generates the S-configuration alcohol with high fidelity. 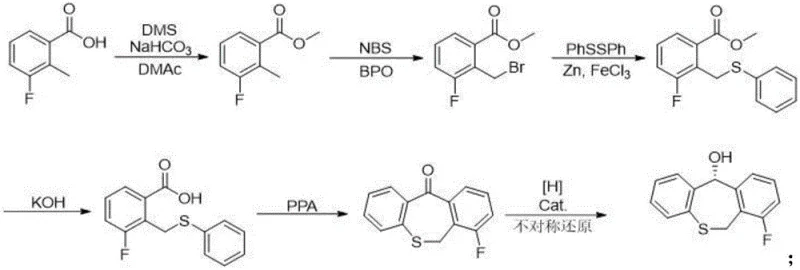 This approach not only simplifies the workflow by removing purification hurdles but also ensures that the subsequent coupling reactions proceed with a homogenous chiral substrate, thereby minimizing the formation of diastereomeric impurities. The result is a cleaner reaction profile and a significantly more cost-effective manufacturing process for this critical pharmaceutical intermediate.
This approach not only simplifies the workflow by removing purification hurdles but also ensures that the subsequent coupling reactions proceed with a homogenous chiral substrate, thereby minimizing the formation of diastereomeric impurities. The result is a cleaner reaction profile and a significantly more cost-effective manufacturing process for this critical pharmaceutical intermediate.
Mechanistic Insights into Rh-Catalyzed Asymmetric Transfer Hydrogenation
The heart of this synthetic breakthrough is the utilization of a specialized Rhodium-based catalyst for asymmetric transfer hydrogenation, which dictates the stereochemical outcome of the ketone reduction. Unlike standard hydride reductions that attack the carbonyl plane indiscriminately, this catalytic system creates a chiral environment that favors the delivery of hydride to one specific face of the ketone substrate. The catalyst, characterized by its complex ligand architecture involving chiral diamines and arene groups, coordinates with the substrate to lower the activation energy for the formation of the S-enantiomer while sterically hindering the formation of the R-isomer. 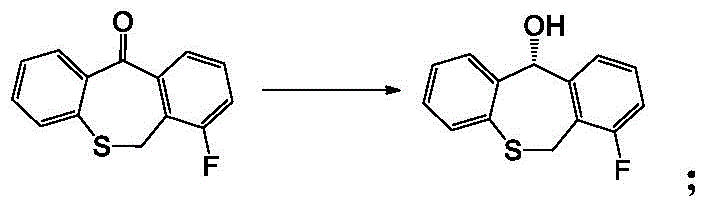 This mechanistic precision is crucial for maintaining the optical purity required for regulatory compliance in pharmaceutical applications. By controlling the transition state geometry, the process achieves high enantiomeric excess without the need for external chiral auxiliaries that would later need to be removed.
This mechanistic precision is crucial for maintaining the optical purity required for regulatory compliance in pharmaceutical applications. By controlling the transition state geometry, the process achieves high enantiomeric excess without the need for external chiral auxiliaries that would later need to be removed.
Furthermore, the integration of this asymmetric step into the broader synthetic sequence ensures that impurity profiles remain manageable throughout the production lifecycle. The subsequent Mitsunobu coupling reaction, which links the chiral alcohol intermediate with the triazine key component, proceeds with inversion of configuration, ultimately delivering the target isomer with the correct absolute stereochemistry. 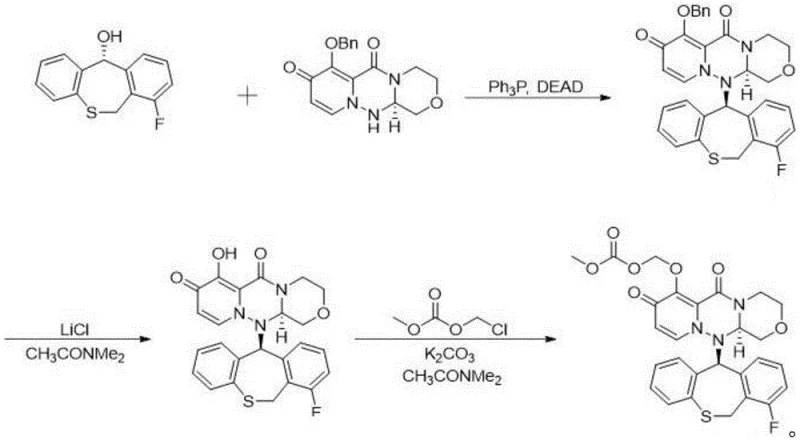 This level of control over the reaction mechanism allows for the systematic exclusion of unwanted stereoisomers, thereby simplifying the downstream purification requirements. For quality control laboratories, this means a more reliable source of reference standards that accurately mimic the degradation pathways of the active drug, facilitating better stability testing and shelf-life determination.
This level of control over the reaction mechanism allows for the systematic exclusion of unwanted stereoisomers, thereby simplifying the downstream purification requirements. For quality control laboratories, this means a more reliable source of reference standards that accurately mimic the degradation pathways of the active drug, facilitating better stability testing and shelf-life determination.
How to Synthesize Baloxavir Marboxil Isomer Efficiently
The execution of this synthesis requires precise control over reaction parameters, particularly during the cyclization and reduction phases, to maximize yield and purity. The process begins with the functionalization of the aromatic ring, followed by the construction of the seven-membered sulfur-containing ring system using polyphosphoric acid catalysis. Once the ketone intermediate is secured, the asymmetric reduction is performed under mild conditions using formic acid as the hydrogen source, ensuring safety and scalability. The final stages involve the strategic coupling of the chiral core with the heterocyclic side chain, followed by deprotection and carbonate formation. For a detailed breakdown of the specific reagents, stoichiometry, and workup procedures required to replicate this high-efficiency route, please refer to the standardized protocol below.
- Esterification and Bromination: Convert 3-fluoro-2-methylbenzoic acid to methyl ester, followed by radical bromination to introduce the reactive benzylic bromide.
- Sulfur Substitution and Cyclization: Substitute bromide with phenylthio group, hydrolyze ester, and perform PPA-catalyzed ring closure to form the dibenzothiepinone core.
- Asymmetric Reduction and Coupling: Utilize Rh-catalyzed asymmetric transfer hydrogenation to establish chirality, followed by Mitsunobu coupling with the triazine key intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route offers substantial strategic benefits beyond mere technical elegance. By eliminating the chiral resolution step, the process inherently reduces the consumption of raw materials and solvents, leading to a leaner and more sustainable manufacturing operation. The reliance on commodity chemicals such as 3-fluoro-2-methylbenzoic acid and standard reagents like N-bromosuccinimide ensures that the supply chain is not vulnerable to the volatility often associated with exotic chiral catalysts or resolving agents. This stability in raw material sourcing translates directly into more predictable pricing models and reduced risk of production delays due to supply shortages. Additionally, the simplified workflow reduces the operational complexity within the plant, allowing for faster batch turnover and improved capacity utilization.
- Cost Reduction in Manufacturing: The most significant economic advantage stems from the drastic simplification of the purification train. By avoiding the loss of material inherent in chiral resolution, the effective yield of the process is maximized, meaning less starting material is required to produce the same amount of final product. This improvement in atom economy directly lowers the cost of goods sold (COGS), making the production of high-purity API intermediates more financially viable. Furthermore, the reduction in processing steps decreases energy consumption and waste generation, contributing to lower operational expenditures and a smaller environmental footprint.
- Enhanced Supply Chain Reliability: The use of robust, non-proprietary reagents and mild reaction conditions enhances the resilience of the supply chain. Manufacturers are not dependent on single-source suppliers for critical chiral inputs, reducing the risk of bottlenecks. The scalability of the route, demonstrated by its successful execution in multi-gram to kilogram scales in the patent examples, assures buyers that the technology can be seamlessly transferred to large-scale commercial production facilities without encountering unforeseen engineering challenges.
- Scalability and Environmental Compliance: The process is designed with green chemistry principles in mind, utilizing safer solvents and minimizing hazardous waste streams. The absence of heavy metal catalysts in the final steps and the ability to recycle certain reagents further align the process with stringent environmental regulations. This compliance is essential for maintaining uninterrupted operations in regulated markets and avoids the costly remediation efforts associated with traditional, less efficient synthetic methods.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is vital for stakeholders evaluating its potential impact on their supply chains. The following questions address common inquiries regarding the scalability, purity, and regulatory implications of this new method. These insights are derived directly from the experimental data and technical disclosures within the patent documentation, providing a factual basis for decision-making.
Q: How does this novel synthesis method improve upon traditional racemic production?
A: Traditional methods often produce a racemic mixture requiring complex and yield-lossing chiral resolution. This patent utilizes asymmetric transfer hydrogenation to directly synthesize the single S-configuration intermediate, eliminating the resolution step entirely and significantly improving overall process efficiency.
Q: What are the critical quality attributes of the produced isomer?
A: The process yields an isomer with a single chiral structure, crucial for serving as a reference standard in impurity profiling. The method ensures high stereochemical purity without the need for post-synthesis separation, guaranteeing consistent quality for analytical and clinical research applications.
Q: Is this synthetic route scalable for industrial production?
A: Yes, the route employs readily available raw materials like 3-fluoro-2-methylbenzoic acid and uses mild, controllable reaction conditions. The elimination of difficult purification steps and the use of standard reagents make it highly suitable for commercial scale-up and industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Baloxavir Marboxil Isomer Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of life-saving antiviral therapies. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the innovative processes described in CN116375734A can be effectively translated into industrial reality. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. Whether you require custom synthesis of complex intermediates or reliable supply of established compounds, our infrastructure is designed to support your most demanding projects with speed and precision.
We invite you to collaborate with us to optimize your supply chain for Baloxavir-related projects. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to help you accelerate your development timelines and secure a competitive advantage in the global pharmaceutical market.
