Advanced Purification Technology for High-Stability Cefotiam Hydrochloride Manufacturing
The pharmaceutical industry constantly seeks robust manufacturing processes that guarantee the stability and safety of life-saving antibiotics. Patent CN109384800B introduces a significant breakthrough in the preparation of cefotiam hydrochloride, a second-generation semi-synthetic cephalosporin widely used for treating severe bacterial infections. This innovation addresses a critical pain point in cephalosporin manufacturing: the tendency of the drug to degrade via hydrolysis and oxidation when residual moisture and solvents are not strictly controlled. By implementing a specialized purification workflow that integrates antioxidant stabilization with a unique multi-stage nitrogen drying protocol, this method achieves exceptionally low water content and organic residue levels. The result is a high-purity active pharmaceutical ingredient with superior storage stability, minimizing the formation of dangerous polymer impurities that can trigger allergic reactions in patients.
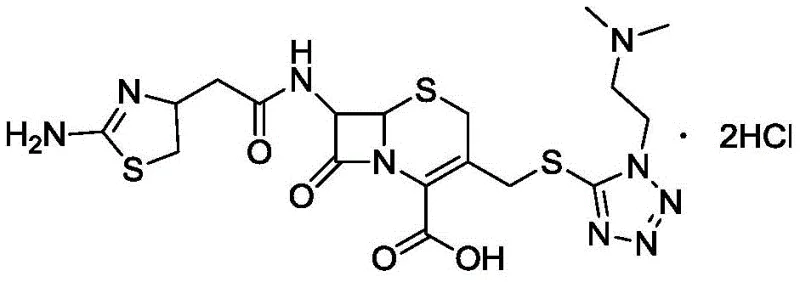
For R&D directors and process chemists, understanding the structural vulnerabilities of cefotiam hydrochloride is paramount. As illustrated in the molecular diagram, the compound features a sensitive beta-lactam ring fused to a dihydrothiazine ring, making it inherently prone to degradation under suboptimal processing conditions. The patented method mitigates these risks through a rigorous control of the crystallization environment and a sophisticated drying regime that preserves the crystal lattice integrity while efficiently stripping away volatile impurities.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional purification strategies for cephalosporins often rely on standard vacuum drying or simple air-drying techniques that fail to address the specific thermodynamic requirements of the cefotiam hydrochloride crystal lattice. In many legacy processes, the removal of residual solvents like ethyl acetate is incomplete, leaving trace amounts that can catalyze degradation reactions over time. Furthermore, conventional drying often lacks precise temperature staging; applying heat too aggressively can cause thermal degradation of the beta-lactam moiety, while insufficient heat leaves bound water within the crystal structure. This residual moisture acts as a plasticizer and a reactant, accelerating hydrolysis and promoting the formation of high-molecular-weight polymers. These polymers are not merely impurities; they are immunogenic triggers responsible for the anaphylactic shocks occasionally associated with cephalosporin therapy. Additionally, older methods frequently neglect the oxidative vulnerability of the thioether and amino-thiazole side chains during the dissolution phase, leading to discoloration and potency loss before the crystallization even begins.
The Novel Approach
The methodology disclosed in patent CN109384800B represents a paradigm shift by integrating chemical stabilization with physical process engineering. The novel approach initiates with the addition of a specific antioxidant system, sodium sulfite-EDTA, directly into the aqueous dissolution phase. This proactive measure neutralizes oxidative threats immediately, protecting the delicate sulfur-containing structures from degradation. Following decolorization and filtration, the process employs a controlled cooling crystallization strategy where ethyl acetate is introduced to create a supersaturated state at precisely 14°C before seeding. This ensures the formation of uniform, high-quality crystals that trap fewer impurities. The true innovation, however, lies in the drying stage. Instead of a single-step drying process, the invention utilizes a dynamic nitrogen blowing technique. The crystals are first pre-dried with nitrogen at mild temperatures (15-25°C) to remove surface solvent without shocking the crystals. Subsequently, the nitrogen temperature is gradually increased to 25-55°C and then maintained at 30-50°C. This gradient approach facilitates the diffusion of internal solvent molecules out of the crystal lattice without causing case hardening or thermal decomposition, resulting in a product with moisture content consistently below 3% and negligible ethyl acetate residue.
Mechanistic Insights into Controlled Nitrogen Drying and Antioxidant Stabilization
The core mechanism driving the success of this preparation method is the synergistic effect between chemical protection and thermodynamic control. During the dissolution step, the inclusion of sodium sulfite-EDTA serves a dual purpose. Sodium sulfite acts as a reducing agent, scavenging dissolved oxygen and preventing the oxidation of the sulfide bridge and the amino group on the thiazole ring. Simultaneously, EDTA functions as a chelating agent, binding trace metal ions (such as copper or iron) that might be present in the water for injection or the crude product. These metal ions are potent catalysts for oxidative degradation; by sequestering them, the process effectively halts metal-catalyzed radical chain reactions that would otherwise compromise the drug's purity. This chemical stabilization is crucial because once the beta-lactam ring is oxidized or opened, the biological activity is lost, and toxic byproducts are formed. The maintenance of a strong acidic environment (pH 0.1-1.0) further stabilizes the molecule by protonating basic sites, reducing the nucleophilicity that could lead to intramolecular attacks on the beta-lactam carbonyl.
From a physical chemistry perspective, the multi-stage nitrogen drying protocol optimizes the mass transfer of solvent molecules from the solid phase to the gas phase. In the initial pre-drying phase at 18-24°C, the nitrogen stream removes the bulk of the mother liquor adhering to the crystal surfaces. This low-temperature step is vital to prevent the rapid evaporation of solvent from the surface, which could otherwise form a crust that traps solvent inside the crystal (a phenomenon known as case hardening). As the process moves to the intermediate and final drying stages, the nitrogen temperature is carefully ramped up to 30-55°C. This provides the necessary activation energy for the diffusion of solvent molecules trapped within the crystal lattice channels. The continuous flow of dry nitrogen maintains a low partial pressure of water and ethyl acetate in the headspace, driving the equilibrium towards the dry solid state. This precise control ensures that the final product meets stringent pharmacopoeial standards for residual solvents while maintaining the crystalline form required for optimal bioavailability and flow properties during downstream tableting or encapsulation.
How to Synthesize Cefotiam Hydrochloride Efficiently
The synthesis and purification of cefotiam hydrochloride described in this patent offer a scalable route for producing high-quality antibiotic intermediates suitable for sterile formulations. The process is designed to be compatible with standard pharmaceutical manufacturing equipment, including stainless steel refining tanks, jacketed crystallizers, and filter-dryers. By adhering to the specific parameters regarding antioxidant concentration, pH adjustment, and the temperature profile of the nitrogen drying cycle, manufacturers can consistently achieve a product with superior stability profiles compared to prior art. The following guide outlines the critical operational phases derived from the patent examples, emphasizing the control points necessary to replicate the high purity and low moisture content demonstrated in the experimental data.
- Dissolve the crude cefotiam hydrochloride in water for injection with sodium sulfite-EDTA antioxidant, adjust to strong acid pH, and decolorize with activated carbon.
- Transfer filtrate to a crystallizer, induce supersaturation with ethyl acetate, seed at 14°C, and grow crystals at 0-5°C.
- Filter and wash crystals, then perform multi-stage drying using temperature-controlled nitrogen flow (15-55°C) to achieve low moisture content.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this advanced preparation method translates into tangible operational efficiencies and risk mitigation. The primary commercial advantage lies in the significant enhancement of product shelf-life and stability. By drastically reducing moisture and residual solvent levels, the risk of product rejection due to failed stability testing during the expiration period is minimized. This reliability reduces waste and ensures a more consistent supply of qualified material for formulation teams. Furthermore, the process eliminates the need for complex, multi-step recrystallization sequences or extended vacuum drying cycles that often bottleneck production throughput. The streamlined nitrogen drying protocol is faster and more energy-efficient than traditional methods, allowing for quicker batch turnover times. This efficiency gain directly contributes to cost reduction in pharmaceutical intermediate manufacturing by lowering utility consumption and maximizing equipment utilization rates without compromising on quality standards.
- Cost Reduction in Manufacturing: The implementation of this process offers substantial cost savings by optimizing the drying phase, which is typically the most time-consuming and energy-intensive step in antibiotic production. By utilizing a controlled nitrogen flow rather than prolonged high-vacuum drying, the method reduces energy consumption and equipment wear. Additionally, the high yield of high-purity product reduces the need for re-processing off-spec batches, leading to better overall material efficiency. The use of common, inexpensive antioxidants like sodium sulfite and EDTA avoids the need for costly proprietary additives, keeping raw material costs low while delivering premium quality.
- Enhanced Supply Chain Reliability: The robustness of this purification method ensures a stable supply of cefotiam hydrochloride that meets rigorous international quality standards. The ability to consistently produce material with low polymer content and high assay values reduces the variability in the supply chain, allowing downstream manufacturers to plan their production schedules with greater confidence. The process is less sensitive to minor fluctuations in ambient conditions due to its closed-system nitrogen drying approach, ensuring consistent output regardless of seasonal humidity changes. This reliability is critical for maintaining uninterrupted production of finished dosage forms in a regulated environment.
- Scalability and Environmental Compliance: This preparation method is inherently scalable, having been demonstrated effectively from laboratory to pilot scales using standard unit operations. The process generates minimal hazardous waste, as the primary solvents (water and ethyl acetate) are easily recoverable and recyclable. The elimination of harsh organic solvents in favor of ethyl acetate and water aligns with green chemistry principles, simplifying environmental compliance and waste disposal logistics. The efficient recovery of ethyl acetate from the drying exhaust further enhances the sustainability profile of the operation, making it an attractive option for environmentally conscious manufacturing partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and quality attributes of cefotiam hydrochloride prepared via this advanced method. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing clarity on how this process differentiates itself from conventional manufacturing routes. Understanding these details is essential for technical teams evaluating the feasibility of integrating this high-stability intermediate into their existing supply chains.
Q: Why is moisture control critical in Cefotiam Hydrochloride production?
A: Excessive moisture promotes hydrolysis and oxidation of the beta-lactam ring, leading to increased polymer impurities which can cause severe anaphylactic reactions in patients. The patented method ensures moisture is kept below 3% to maintain stability.
Q: What is the role of Sodium Sulfite-EDTA in this process?
A: Sodium Sulfite-EDTA acts as a potent antioxidant system during the dissolution phase. It scavenges free radicals and chelates metal ions that could catalyze oxidative degradation, thereby preserving the integrity of the active pharmaceutical ingredient before crystallization.
Q: How does the nitrogen drying method improve product quality compared to vacuum drying?
A: The specific multi-stage nitrogen blowing technique allows for precise temperature control that removes residual ethyl acetate and surface water without subjecting the heat-sensitive cephalosporin structure to the potential thermal stress or uneven drying often associated with traditional vacuum methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cefotiam Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize that the quality of the starting material defines the success of the final therapeutic product. Our technical team has extensively analyzed the preparation method of cefotiam hydrochloride described in patent CN109384800B and is fully equipped to implement this advanced purification technology at a commercial scale. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the precise temperature controls and antioxidant protocols required for this process are meticulously maintained. Our facility is outfitted with state-of-the-art reactor systems and rigorous QC labs capable of detecting trace impurities and polymer content, guaranteeing that every batch meets stringent purity specifications and stability requirements essential for parenteral applications.
We invite global pharmaceutical partners to collaborate with us to leverage this superior manufacturing technology for your antibiotic portfolios. By choosing NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team today to request specific COA data from our recent pilot runs and to discuss route feasibility assessments for your upcoming projects. Let us help you secure a supply of high-stability cefotiam hydrochloride that enhances patient safety and optimizes your production economics.
