Advanced Pd-Catalyzed Synthesis of 2,3-Dihydro-1,4-Benzothiazine Intermediates for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to construct privileged scaffolds efficiently. Patent CN103102329A introduces a groundbreaking synthetic route for 2,3-dihydro-[1,4]-benzothiazine compounds, a core structure prevalent in numerous bioactive molecules. This innovation addresses critical bottlenecks in traditional synthesis by employing a palladium-catalyzed cyclization strategy using sodium thiosulfate as a sulfur source. The significance of this scaffold cannot be overstated, as it serves as the fundamental backbone for potent drugs such as Rufloxacin, DHBT-NE-1, and Heronamycin A, which exhibit diverse biological activities ranging from antibacterial to antidiabetic properties. 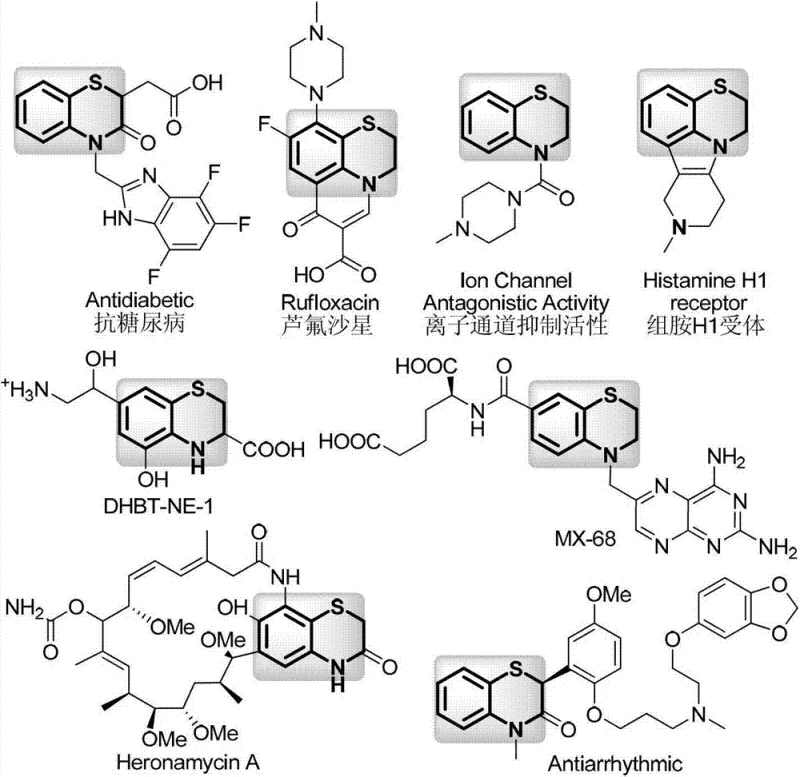 By leveraging this patented technology, manufacturers can access a reliable pharmaceutical intermediate supplier pathway that ensures high purity and structural integrity, essential for downstream drug development.
By leveraging this patented technology, manufacturers can access a reliable pharmaceutical intermediate supplier pathway that ensures high purity and structural integrity, essential for downstream drug development.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the 2,3-dihydro-[1,4]-benzothiazine ring system has relied heavily on the condensation of substituted o-aminothiophenols with appropriate electrophiles. This conventional approach suffers from severe practical drawbacks that hinder its applicability in modern green chemistry and large-scale manufacturing. Substituted o-aminothiophenols are notoriously unstable, prone to rapid oxidation upon exposure to air, which complicates storage and handling protocols significantly. Furthermore, these precursors possess an extremely offensive and persistent odor, creating hazardous working environments and necessitating expensive containment infrastructure. The synthesis of these thiol precursors themselves is often multi-step and low-yielding, adding unnecessary complexity and cost to the overall supply chain. Consequently, the reliance on such problematic starting materials restricts the substrate scope and limits the economic feasibility of producing complex benzothiazine derivatives for commercial applications.
The Novel Approach
In stark contrast, the methodology disclosed in CN103102329A revolutionizes this landscape by utilizing 2-iodo-N-(2-iodoethyl)aniline derivatives as stable, odorless starting materials. This novel route employs sodium thiosulfate (Na2S2O3) as an inexpensive and benign sulfurization reagent, effectively bypassing the need for volatile thiols. The reaction proceeds under the influence of a palladium catalyst, facilitating an intramolecular C-S bond formation that constructs the heterocyclic ring with high efficiency. 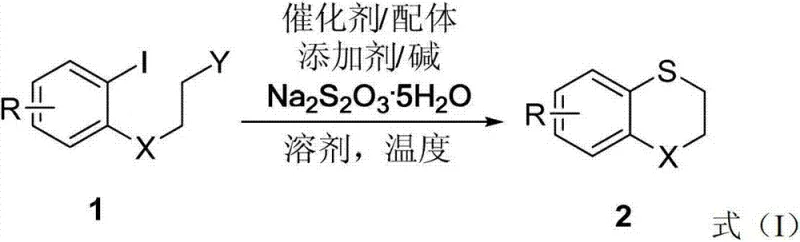 This shift in strategy not only eliminates the olfactory hazards associated with traditional methods but also significantly broadens the functional group tolerance, allowing for the introduction of diverse substituents such as halogens, alkyl groups, and electron-withdrawing moieties. The result is a streamlined process that offers superior operational simplicity and environmental compatibility, positioning it as a preferred choice for cost reduction in pharmaceutical intermediates manufacturing.
This shift in strategy not only eliminates the olfactory hazards associated with traditional methods but also significantly broadens the functional group tolerance, allowing for the introduction of diverse substituents such as halogens, alkyl groups, and electron-withdrawing moieties. The result is a streamlined process that offers superior operational simplicity and environmental compatibility, positioning it as a preferred choice for cost reduction in pharmaceutical intermediates manufacturing.
Mechanistic Insights into Pd-Catalyzed Cyclization
The success of this transformation hinges on a sophisticated palladium-catalyzed cycle that orchestrates the cleavage of carbon-halogen bonds and the subsequent formation of carbon-sulfur linkages. The mechanism likely initiates with the oxidative addition of the aryl iodide moiety of the substrate to the active Pd(0) species generated in situ from the precatalyst. This step creates an aryl-palladium(II) intermediate, which is primed for nucleophilic attack. Simultaneously, the thiosulfate anion acts as a sulfur donor, potentially undergoing decomposition or ligand exchange to generate a reactive sulfur species capable of coordinating with the metal center. The presence of the phase transfer catalyst TBAB enhances the solubility and reactivity of the inorganic thiosulfate salt in the organic solvent medium, ensuring efficient interaction with the catalytic complex.
Following the coordination of the sulfur species, an intramolecular nucleophilic substitution or transmetallation event occurs at the alkyl halide side chain, or alternatively, a sequential C-S and C-N bond formation pathway may be operative depending on the specific substrate configuration. The final step involves reductive elimination, which releases the cyclized 2,3-dihydro-[1,4]-benzothiazine product and regenerates the active Pd(0) catalyst to continue the cycle. Crucially, the use of Cs2CO3 as a base facilitates the deprotonation steps required to drive the equilibrium forward and neutralize acidic byproducts. This mechanistic elegance ensures high atom economy and minimizes the formation of undesired side products, thereby simplifying the impurity profile and reducing the burden on downstream purification processes.
How to Synthesize 2,3-Dihydro-1,4-Benzothiazine Efficiently
To implement this synthesis effectively, precise control over reaction parameters is essential to maximize yield and purity. The protocol typically involves charging a reaction vessel with the di-iodo aniline substrate, the palladium catalyst system, and the sulfur source under an inert atmosphere to prevent catalyst deactivation. The choice of solvent system, specifically a mixture of acetonitrile and water, plays a pivotal role in dissolving both the organic substrate and the inorganic reagents. Detailed standardized synthesis steps see the guide below.
- Prepare the reaction vessel under inert atmosphere and charge with 2-iodo-N-(2-iodoethyl)aniline derivative, PdCl2(dppf) catalyst, Cs2CO3 base, and Na2S2O3 sulfur source.
- Add the solvent system consisting of acetonitrile and water along with TBAB additive, then heat the mixture to 150°C for 8 hours.
- Upon completion, cool the reaction, adsorb onto silica gel, and purify via column chromatography to isolate the target benzothiazine product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis route offers tangible strategic benefits that extend beyond mere chemical efficiency. The transition from malodorous thiols to stable iodo-anilines fundamentally alters the risk profile of the manufacturing process, reducing safety incidents and regulatory compliance costs associated with hazardous material handling. This stability translates directly into enhanced supply chain reliability, as raw materials can be sourced globally without the degradation issues common to thiol-based precursors. Furthermore, the use of commodity chemicals like sodium thiosulfate and cesium carbonate ensures that input costs remain predictable and low, shielding the production budget from the volatility often seen with specialized fine chemical reagents.
- Cost Reduction in Manufacturing: The elimination of expensive and difficult-to-handle thiol starting materials results in substantial cost savings throughout the production lifecycle. By utilizing sodium thiosulfate, a widely available industrial chemical, the direct material costs are drastically simplified compared to traditional routes. Additionally, the high yields reported in the patent minimize waste generation and reduce the consumption of solvents and energy per unit of product, contributing to a leaner and more economical manufacturing process that enhances overall profit margins.
- Enhanced Supply Chain Reliability: The robustness of the starting materials ensures a consistent and uninterrupted supply of key intermediates. Unlike o-aminothiophenols which require special storage conditions due to oxidation sensitivity, the iodo-aniline derivatives are shelf-stable and easy to transport. This stability reduces lead time for high-purity pharmaceutical intermediates by eliminating the need for expedited shipping or specialized cold-chain logistics, allowing for more flexible inventory management and just-in-time production strategies.
- Scalability and Environmental Compliance: The process is inherently designed for commercial scale-up of complex pharmaceutical intermediates, utilizing standard reactor equipment and straightforward workup procedures like silica gel chromatography. The absence of noxious sulfur odors significantly improves the working environment and simplifies waste gas treatment systems, ensuring strict adherence to environmental regulations. This eco-friendly profile facilitates smoother regulatory approvals and supports sustainable manufacturing goals, making it an attractive option for long-term production partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthesis method. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on operational specifics and performance metrics.
Q: What are the advantages of using Na2S2O3 over o-aminothiophenols?
A: Sodium thiosulfate is odorless, stable, and inexpensive, whereas o-aminothiophenols are malodorous, prone to oxidation, and difficult to handle, making the new method safer and more environmentally friendly.
Q: What is the optimal catalyst system for this cyclization?
A: The patent identifies PdCl2(dppf) as the preferred catalyst, often used in conjunction with dppf ligand and TBAB additive to achieve yields up to 99%.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the method utilizes commercially available raw materials, mild reaction conditions, and simple workup procedures, making it highly adaptable for industrial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,3-Dihydro-1,4-Benzothiazine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of accessing high-quality intermediates for your drug development pipelines. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and consistency. We are committed to maintaining stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of 2,3-dihydro-1,4-benzothiazine derivative meets the highest industry standards for safety and efficacy.
We invite you to collaborate with us to leverage this advanced synthetic technology for your specific applications. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our capabilities can accelerate your time to market while optimizing your production costs.
