Advanced Synthesis of 2-(4-Nitrophenyl)butyric Acid for Scalable Indobufen Production
The pharmaceutical industry's relentless pursuit of efficient, scalable, and environmentally compliant synthetic routes for critical API intermediates has found a significant breakthrough in the technology disclosed in patent CN114085152A. This patent details a novel method for preparing 2-(4-nitrophenyl)butyric acid, a pivotal precursor in the synthesis of Indobufen, a potent antiplatelet agent used globally for treating cardiovascular and cerebrovascular diseases. Unlike legacy processes that rely on harsh nitration conditions prone to generating complex impurity profiles, this innovation leverages a strategic nucleophilic aromatic substitution followed by a selective reduction. For R&D directors and procurement strategists alike, this represents a paradigm shift towards cleaner manufacturing, offering a pathway to high-purity pharmaceutical intermediates that drastically simplifies downstream purification and enhances overall process economics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of 2-(4-nitrophenyl)butyric acid has been plagued by inherent chemical inefficiencies associated with electrophilic aromatic substitution. The traditional route typically involves the nitration of 2-phenylbutyric acid using a mixture of concentrated sulfuric and nitric acids. As illustrated in the reaction scheme below, this non-selective nitration inevitably produces a mixture of regioisomers, primarily the desired para-product alongside the unwanted ortho-isomer, 2-(2-nitrophenyl)butyric acid.
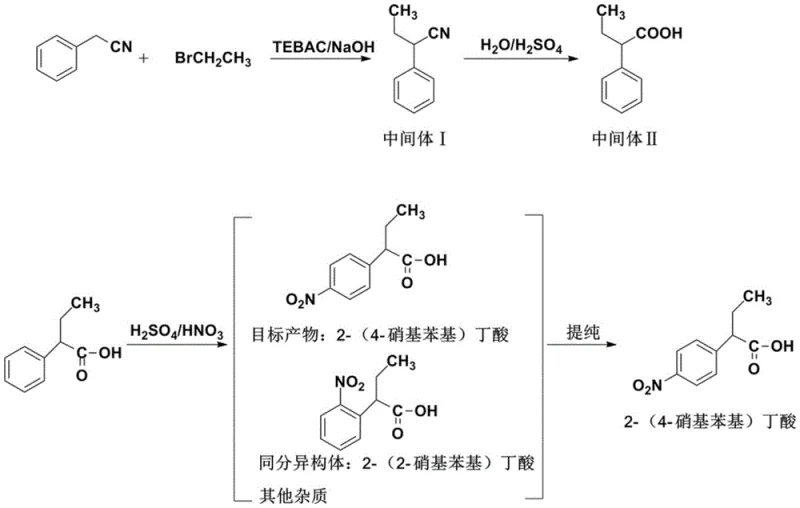
The presence of these structural analogs creates a formidable separation challenge, often requiring multiple recrystallizations or complex chromatographic techniques that erode overall yield and inflate production costs. Furthermore, the reliance on mixed acid generates substantial quantities of hazardous waste acid liquor, imposing severe environmental compliance burdens and escalating waste disposal costs. The low atom economy and the difficulty in achieving high purity standards make this conventional approach increasingly untenable for modern, sustainability-focused pharmaceutical manufacturing facilities seeking to optimize their supply chains.
The Novel Approach
In stark contrast, the methodology outlined in CN114085152A circumvents these pitfalls by reversing the synthetic logic, constructing the carbon skeleton onto a pre-functionalized aromatic ring. By utilizing p-nitrohalobenzene (such as p-nitrofluorobenzene or p-nitrochlorobenzene) as the starting material, the nitro group is already positioned correctly at the para-position, rendering the formation of ortho-isomers chemically impossible. This route proceeds through a nucleophilic substitution with alkyl acetoacetate to form a stable intermediate, which is subsequently reduced to the target acid. This strategic redesign not only guarantees exceptional regioselectivity but also streamlines the workflow into just two high-yielding steps, effectively eliminating the need for difficult isomer separation and significantly reducing the environmental footprint associated with waste acid generation.
Mechanistic Insights into Nucleophilic Substitution and Huang Minlon Reduction
The core of this innovative synthesis lies in its two distinct mechanistic phases, both of which are optimized for high conversion and selectivity. The first step involves a nucleophilic aromatic substitution where the enolate of an alkyl acetoacetate, generated in situ using a strong base like sodium methoxide or sodium hydride, attacks the electron-deficient aromatic ring of the p-nitrohalobenzene. The strong electron-withdrawing nature of the nitro group activates the ring towards nucleophilic attack, facilitating the displacement of the halide leaving group. This reaction is highly specific; because the starting material is exclusively the para-isomer, the resulting intermediate, 2-(4-nitrophenyl)alkyl acetoacetate, possesses the exact substitution pattern required for the final product, ensuring that no positional isomers are introduced at this stage.
The second phase employs a classic Huang Minlon modification of the Wolff-Kishner reduction to convert the beta-keto ester functionality into the corresponding alkyl chain. Under strongly alkaline conditions and elevated temperatures (typically 190-200°C) in a high-boiling solvent like polyethylene glycol, hydrazine hydrate reacts with the carbonyl group to form a hydrazone intermediate. Subsequent decomposition of this hydrazone releases nitrogen gas and yields the saturated alkyl chain, effectively reducing the ketone to a methylene group while preserving the sensitive nitro functionality on the aromatic ring. This chemoselectivity is crucial, as it allows for the transformation of the side chain without affecting the nitro group, which would otherwise require protection-deprotection strategies in less robust synthetic routes.
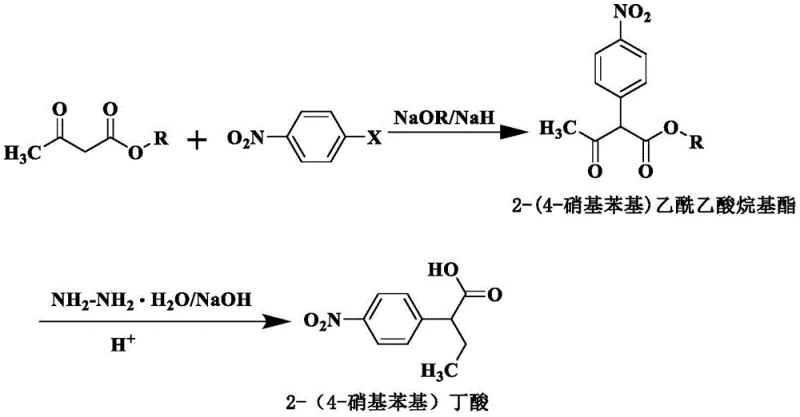
Following the reduction, the reaction mixture is carefully acidified to a pH of 4-6, inducing the crystallization of the free acid. This pH-controlled workup is a critical purification step that leverages the solubility differences between the product and inorganic salts or unreacted hydrazine, allowing for the isolation of high-purity 2-(4-nitrophenyl)butyric acid directly from the reaction matrix. The combination of these mechanisms results in a process that is not only chemically elegant but also operationally simple, avoiding the use of transition metal catalysts or hazardous reagents that often complicate scale-up and regulatory approval.
How to Synthesize 2-(4-Nitrophenyl)butyric Acid Efficiently
The practical implementation of this synthesis requires precise control over reaction parameters to maximize yield and purity, as detailed in the patent examples. The process begins with the careful generation of the nucleophile in an anhydrous environment, followed by the controlled addition of the nitro-halide substrate. The subsequent reduction step demands rigorous temperature management to ensure complete conversion of the hydrazone intermediate while preventing thermal degradation of the product. By adhering to these standardized protocols, manufacturers can reliably produce this critical intermediate with consistent quality suitable for GMP environments.
- React p-nitrohalobenzene (such as p-nitrofluorobenzene) with alkyl acetoacetate in the presence of a base like sodium methoxide or sodium hydride to form 2-(4-nitrophenyl)alkyl acetoacetate.
- Subject the intermediate to Huang Minlon reduction using hydrazine hydrate under alkaline conditions at elevated temperatures (190-200°C).
- Adjust the pH to 4-6 using acid, followed by crystallization to isolate high-purity 2-(4-nitrophenyl)butyric acid without isomer impurities.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology offers profound strategic advantages that extend far beyond simple chemical yield improvements. The elimination of the mixed acid nitration step fundamentally alters the cost structure of production by removing the need for expensive corrosion-resistant equipment and complex waste neutralization infrastructure. This shift translates directly into lower capital expenditure requirements for new production lines and reduced operational expenditures for existing facilities, making the manufacturing of 2-(4-nitrophenyl)butyric acid significantly more economically viable in a competitive global market.
- Cost Reduction in Manufacturing: The most immediate financial benefit arises from the drastic simplification of the purification process. Since the new route generates zero ortho-isomer impurities, the costly and yield-eroding steps associated with separating structural analogs are completely eliminated. This inherent purity means that fewer processing cycles are required to meet stringent pharmaceutical specifications, leading to substantial savings in solvent consumption, energy usage, and labor hours. Furthermore, the raw materials employed, such as p-nitrofluorobenzene and alkyl acetoacetates, are commodity chemicals with stable supply chains, insulating the production cost from the volatility often seen with specialized nitrating agents.
- Enhanced Supply Chain Reliability: By relying on widely available bulk chemicals rather than hazardous mixed acids, the supply chain becomes more resilient and easier to manage. The avoidance of highly corrosive and regulated substances simplifies logistics, storage, and transportation, reducing the administrative burden and risk associated with handling dangerous goods. This reliability ensures consistent production schedules and minimizes the risk of shutdowns due to regulatory inspections or supply disruptions of critical reagents, thereby securing a steady flow of high-purity pharmaceutical intermediates to downstream API manufacturers.
- Scalability and Environmental Compliance: From a sustainability perspective, this process is a model for green chemistry in fine chemical manufacturing. The absence of waste acid liquor removes a major environmental liability, significantly lowering the cost and complexity of wastewater treatment. This alignment with environmental, social, and governance (ESG) goals makes the facility more attractive to international partners who prioritize sustainable sourcing. Additionally, the simplicity of the two-step sequence facilitates straightforward scale-up from pilot plant to commercial tonnage, allowing for rapid response to market demand fluctuations without the technical bottlenecks typical of multi-step nitration processes.
Frequently Asked Questions (FAQ)
To further clarify the technical and commercial implications of this synthesis route, we have compiled answers to common inquiries regarding its implementation and benefits. These insights are derived directly from the experimental data and technical disclosures within the patent documentation, providing a transparent view of the process capabilities. Understanding these nuances is essential for stakeholders evaluating the feasibility of integrating this technology into their existing manufacturing portfolios.
Q: Why is this new method superior to traditional nitration routes for 2-(4-nitrophenyl)butyric acid?
A: Traditional methods involve nitrating 2-phenylbutyric acid with mixed acid, which inevitably generates difficult-to-separate ortho-isomer impurities (2-(2-nitrophenyl)butyric acid). The patented method uses p-nitrohalobenzene as a starting material, ensuring 100% para-selectivity and eliminating isomer contamination entirely.
Q: What are the key raw materials required for this synthesis?
A: The process utilizes commercially available and cost-effective raw materials, specifically p-nitrofluorobenzene or p-nitrochlorobenzene, combined with alkyl acetoacetates (methyl or ethyl esters), making the supply chain robust and less susceptible to volatility compared to specialized nitrating agents.
Q: How does this process impact environmental compliance and waste management?
A: By completely avoiding the use of sulfuric and nitric acid mixtures (mixed acid), the process eliminates the generation of large volumes of hazardous waste acid liquor, significantly reducing the burden on wastewater treatment facilities and aligning with stricter global environmental regulations.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-(4-Nitrophenyl)butyric Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced synthetic routes like the one described in CN114085152A requires a partner with deep technical expertise and robust manufacturing capabilities. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this green chemistry approach are fully realized in a commercial setting. Our state-of-the-art facilities are equipped to handle the specific thermal and safety requirements of the Huang Minlon reduction, while our rigorous QC labs enforce stringent purity specifications to guarantee that every batch of 2-(4-nitrophenyl)butyric acid meets the exacting standards required for Indobufen synthesis.
We invite forward-thinking pharmaceutical companies to collaborate with us to leverage this superior technology for their supply chains. By partnering with our technical procurement team, you can obtain a Customized Cost-Saving Analysis that quantifies the potential efficiencies of switching to this isomer-free route. We encourage you to reach out today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our commitment to innovation can drive value and reliability in your critical API manufacturing operations.
