Strategic Process Optimization for 10-Methoxyiminostilbene Manufacturing and Commercial Scale-Up
Introduction to Next-Generation Intermediate Synthesis
The pharmaceutical industry constantly seeks robust, scalable, and environmentally sustainable routes for critical drug intermediates, particularly for antiepileptic medications like Oxcarbazepine. A recent technological breakthrough, documented in patent CN114957122A, introduces a highly optimized preparation method for 10-methoxyiminostilbene, a pivotal precursor in this therapeutic class. This innovation addresses long-standing challenges in waste management and cost efficiency by integrating a comprehensive recycling loop directly into the synthesis workflow. The core of this advancement lies not just in the formation of the target molecule, shown below, but in the sophisticated handling of the reaction byproducts.
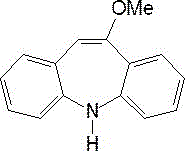
Traditionally, the production of such intermediates involves harsh conditions that generate substantial volumes of saline wastewater, posing significant disposal costs and environmental risks. However, this new methodology transforms the waste stream into a value stream. By implementing a specific recovery step that isolates methanol and converts residual potassium ions into high-purity potassium bromide, the process achieves a dual objective: minimizing ecological footprint and maximizing economic return. For R&D directors and supply chain leaders, this represents a shift from linear consumption to circular chemical manufacturing, ensuring a more reliable pharmaceutical intermediates supplier partnership capable of meeting stringent global compliance standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art, such as the method disclosed in CN106467491A, typically relies on a straightforward dehydrobromination using potassium hydroxide in mixed solvents. While effective for conversion, these legacy processes suffer from a critical flaw in their downstream processing: the generation of large volumes of aqueous waste laden with unreacted potassium ions and organic residues. In standard operations, the mass ratio of KOH to the starting dibromo-imino-dibenzyl is often high (around 3:2), leading to a water layer post-reaction that is saturated with potassium salts. This wastewater cannot be directly discharged due to environmental regulations, nor can it be easily reused because of accumulated impurities. Consequently, manufacturers face escalating costs associated with wastewater treatment facilities and the loss of valuable solvents like methanol, which are often difficult to separate cleanly from the aqueous phase without energy-intensive distillation.
The Novel Approach
The innovative process described in CN114957122A fundamentally re-engineers the workup phase to turn these liabilities into assets. Instead of treating the aqueous layer as waste, the new method subjects it to a multi-stage recovery protocol. This begins with the separation of the organic layer containing the crude 10-methoxyiminostilbene, followed by a rigorous treatment of the water layer. The reaction scheme below illustrates the primary transformation where the dibromo precursor is converted into the target azepine structure.
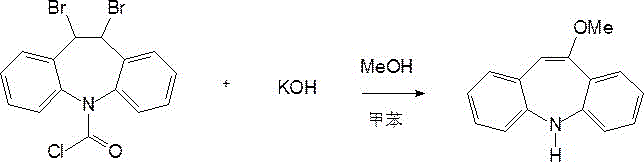
Crucially, the aqueous phase undergoes distillation in a hypergravity bed to recover methanol for immediate reuse in the feeding step, drastically cutting raw material procurement needs. Furthermore, the remaining potassium ion mother liquor is neutralized with hydrobromic acid to precipitate potassium bromide. This byproduct is then purified to over 94% purity, creating a secondary revenue stream that offsets production costs. This approach not only solves the discharge problem but also enhances the overall atom economy of the synthesis, offering a compelling case for cost reduction in pharmaceutical intermediates manufacturing through intelligent process design rather than simple scale expansion.
Mechanistic Insights into Dehydrobromination and Solvent Recovery
The core chemical transformation involves an elimination reaction where potassium hydroxide acts as a strong base to abstract protons and facilitate the removal of bromine atoms from the 10,11-dibromoiminodibenzyl scaffold. This dehydrobromination is conducted in a biphasic system of methanol and an aromatic solvent like toluene. The choice of solvent system is critical; methanol serves as the polar medium to dissolve the inorganic base, while toluene helps solubilize the organic substrate and facilitates the subsequent phase separation. The reaction is driven thermodynamically by the formation of stable potassium bromide salts and the aromatization of the central seven-membered ring. Maintaining the reaction temperature between 80-90°C ensures complete conversion while minimizing side reactions that could lead to polymeric impurities or over-oxidation.
Beyond the primary reaction, the true mechanistic sophistication lies in the separation physics employed during the recovery phase. The use of a hypergravity bed (a baffling type hypergravity revolving bed) for methanol recovery is a standout feature. Unlike traditional static distillation columns, this rotating equipment generates high gravity fields that intensify mass transfer rates. This allows for the rapid and efficient separation of methanol from the aqueous potassium matrix at lower energy inputs. Following methanol recovery, the pH of the mother liquor is carefully adjusted to 6-7 using hydrobromic acid. This neutralization step is precise; it converts soluble potassium hydroxide/carbonate residues into potassium bromide without introducing new cationic impurities. Subsequent decolorization with activated carbon removes trace organic contaminants, ensuring the final potassium bromide crystal lattice is pure enough for commercial sale, thereby closing the loop on material usage.
How to Synthesize 10-Methoxyiminostilbene Efficiently
Executing this synthesis requires precise control over feeding rates and temperature gradients to manage the exothermic nature of the base addition and the subsequent elimination. The process begins with the dissolution of potassium hydroxide in methanol at low temperatures (around 10°C) to prevent premature degradation or side reactions before the substrate is introduced. Once the substrate is added and the reaction reaches completion, the workup becomes the defining factor for yield and purity. The organic layer is washed with hot water to extract residual salts, while the aqueous layer is immediately routed to the recovery train. Detailed operational parameters, including specific stirring speeds, vacuum levels for distillation (less than -0.08 MPa), and crystallization cooling profiles, are essential for replicating the high yields (>91%) reported in the patent data.
- Feed 10,11-dibromoiminodibenzyl into a mixed solvent of methanol and toluene with potassium hydroxide, maintaining strict temperature control during the addition phase.
- Execute the reaction at elevated temperatures (80-90°C) followed by a specialized water washing step to separate the organic product layer from the aqueous waste layer containing potassium ions.
- Process the aqueous layer through a hypergravity bed for methanol recovery, neutralize with hydrobromic acid, and purify the resulting potassium bromide via crystallization and vacuum drying.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented process translates into tangible strategic advantages beyond mere chemical yield. The most significant impact is on the total cost of ownership (TCO) for the intermediate. By recovering and recycling methanol and toluene, the facility drastically reduces its dependency on volatile solvent markets. Moreover, the ability to sell purified potassium bromide transforms a waste disposal cost center into a profit center. This internal subsidization mechanism provides a buffer against raw material price fluctuations, ensuring more stable pricing for the final API intermediate. The reduction in wastewater volume also lowers the burden on effluent treatment plants (ETP), reducing both capital expenditure on infrastructure and ongoing operational fees associated with environmental compliance.
- Cost Reduction in Manufacturing: The integration of a closed-loop solvent recovery system eliminates the need for purchasing fresh methanol for every batch, leading to substantial savings in raw material expenditures. Additionally, the valorization of the potassium waste stream into a marketable commodity (potassium bromide) creates a direct revenue offset. This dual mechanism of cost avoidance and revenue generation significantly improves the gross margin profile of the manufacturing process without compromising on the quality of the active pharmaceutical ingredient.
- Enhanced Supply Chain Reliability: Traditional processes that generate difficult-to-treat hazardous waste are prone to regulatory bottlenecks and shutdowns. By converting hazardous saline waste into a benign, saleable solid, this process de-risks the supply chain. It ensures continuous operation even during periods of strict environmental enforcement. Furthermore, the simplified equipment requirements—relying on standard reactors, centrifuges, and hypergravity beds rather than exotic catalysts—mean that the technology is easily transferable and scalable across different manufacturing sites, guaranteeing consistent supply continuity.
- Scalability and Environmental Compliance: The process is designed with industrial scalability in mind. The use of centrifugal separation for the potassium bromide crystals offers higher throughput and easier handling compared to traditional filtration, which can be prone to clogging and inefficiency at large scales. The reduced discharge of liquid waste aligns perfectly with modern green chemistry principles and increasingly stringent global environmental regulations. This proactive compliance posture future-proofs the supply chain against tightening legislation, making it a preferred choice for multinational corporations with strict ESG (Environmental, Social, and Governance) mandates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a clear picture of what partners can expect regarding yield, purity, and operational complexity. Understanding these nuances is vital for technical teams evaluating the feasibility of technology transfer.
Q: How does this process improve upon conventional methods for 10-methoxyiminostilbene?
A: Unlike conventional methods that generate difficult-to-treat potassium-rich wastewater, this patented process converts waste potassium ions into sellable potassium bromide (purity >94%) and recycles methanol and water, significantly reducing environmental burden and raw material costs.
Q: What is the role of the hypergravity bed in this synthesis?
A: The hypergravity bed is utilized during the methanol recovery step to efficiently separate methanol from the aqueous potassium ion mother liquor. This novel rectification equipment offers high efficiency and low energy consumption compared to traditional distillation, ensuring high-quality solvent recovery for reuse.
Q: Can the potassium bromide byproduct be commercially utilized?
A: Yes, the process includes a dedicated purification step involving neutralization, decolorization with activated carbon, and crystallization, yielding potassium bromide with a purity exceeding 94wt%, making it suitable for sale as a chemical raw material.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 10-Methoxyiminostilbene Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory patent to commercial reality requires more than just chemical knowledge; it demands engineering excellence. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facilities are equipped to handle the specific unit operations required by this advanced process, including hypergravity distillation and precision crystallization. We adhere to stringent purity specifications and operate rigorous QC labs to ensure that every batch of 10-methoxyiminostilbene meets the exacting standards required for downstream Oxcarbazepine synthesis, guaranteeing consistency and reliability for your drug development pipeline.
We invite you to collaborate with us to leverage this innovative manufacturing route for your projects. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this waste-valorization strategy can improve your bottom line. Please contact our technical procurement team today to request specific COA data, route feasibility assessments, and to discuss how we can secure your supply chain with high-purity, sustainably produced intermediates.
