Advanced Recycling Technology for Perindopril Intermediates Enhancing Commercial Scale-Up Capabilities
Advanced Recycling Technology for Perindopril Intermediates Enhancing Commercial Scale-Up Capabilities
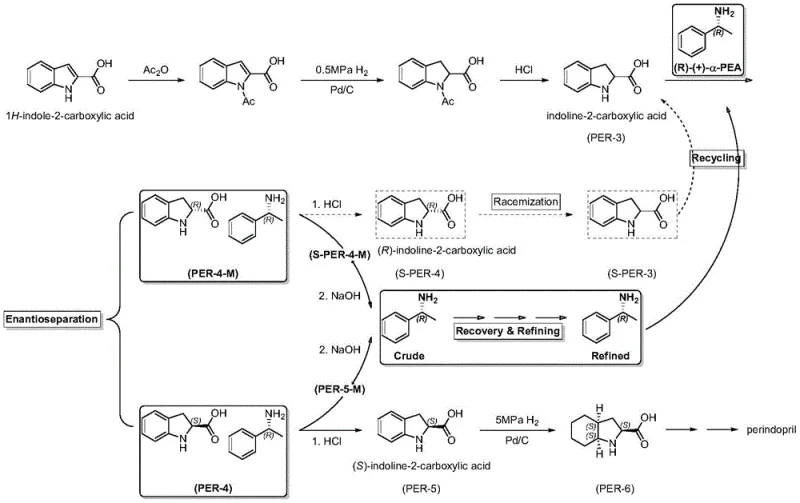
The global demand for antihypertensive medications continues to drive the need for efficient manufacturing processes for key active pharmaceutical ingredients like perindopril. A critical bottleneck in this supply chain has historically been the recovery and reuse of expensive chiral resolving agents, specifically (R)-(+)-alpha-phenylethylamine. Patent CN113087630A discloses a groundbreaking method that transforms the recovery of this vital reagent from a loss-prone operation into a high-efficiency circular process. By integrating a novel acidic crystallization step into the traditional workflow, this technology ensures that the recycled amine meets the stringent purity and optical rotation specifications required for industrial-scale resolution of indoline-2-carboxylic acid. This innovation not only addresses the chemical instability of free amines but also establishes a robust framework for cost reduction in pharmaceutical intermediate manufacturing, making it a pivotal development for producers aiming to optimize their production lines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the recovery of chiral amines from resolution mother liquors has relied on a straightforward sequence of alkalization, solvent extraction, and direct distillation. While conceptually simple, this conventional approach suffers from significant technical deficiencies when applied to sensitive molecules like (R)-(+)-alpha-phenylethylamine. The free amine is highly susceptible to air oxidation, leading to the formation of colored impurities and a marked decrease in optical purity, often resulting in specific rotation values as low as 35 to 37 degrees. Furthermore, direct distillation fails to effectively separate non-volatile organic impurities and degradation products that co-extract with the amine, yielding a product with GC purity typically ranging between 92% and 97%. These quality deficits compromise the efficacy of the resolving agent in subsequent batches, leading to lower yields of the desired (S)-indoline-2-carboxylic acid and necessitating frequent replenishment with fresh, costly reagents.
The Novel Approach
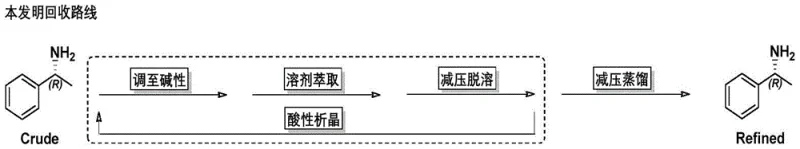
In stark contrast to the legacy methods, the technology described in patent CN113087630A introduces a sophisticated purification loop centered around an acidic crystallization step. Instead of rushing to distillation, the process first converts the extracted free amine into a stable salt form within a specific organic solvent system at low temperatures. This strategic pivot allows for the physical separation of the target amine salt from soluble impurities and oxidized byproducts that do not crystallize under these conditions. The resulting wet cake is then liberated and distilled, but because the feed material is already significantly purified, the final distillation yields a colorless, transparent product with exceptional quality. This method effectively breaks the cycle of quality degradation, ensuring that the recycled resolving agent performs identically to fresh material, thereby stabilizing the entire production workflow for high-purity pharmaceutical intermediates.
Mechanistic Insights into Acidic Crystallization Purification
The core mechanistic advantage of this new process lies in the differential solubility and stability of the amine salt versus its impurities. In the traditional basic extraction environment, oxidized impurities and polymeric byproducts often remain dissolved alongside the target amine, carrying over into the distillation pot where thermal stress can further degrade the product. By introducing an inorganic acid to adjust the pH to a weakly alkaline range (pH 7-9) in a solvent like toluene or ethanol at temperatures below 10°C, the process selectively precipitates the (R)-(+)-alpha-phenylethylamine salt. This crystallization acts as a rigorous filtration barrier; impurities that are either too polar or structurally distinct from the salt lattice remain in the mother liquor and are washed away. This step is crucial for removing the chromophores responsible for the yellowing of recovered amines, ensuring the final product is colorless and transparent, which is a key visual indicator of chemical integrity for reliable pharmaceutical intermediate suppliers.
Furthermore, the control of pH and temperature during the crystallization phase plays a vital role in maintaining stereochemical integrity. While racemization is less of a concern for alpha-phenylethylamine compared to amino acids, the gentle conditions prevent any potential thermal degradation that could occur during aggressive distillation of crude mixtures. The subsequent liberation of the amine from the purified salt using a strong inorganic base ensures that the free amine entering the final distillation column is of near-perfect quality. This minimizes the thermal load on the distillation equipment and reduces the formation of new degradation products during the final concentration step. The result is a closed-loop system where the chemical potential of the resolving agent is preserved, allowing for multiple recycling cycles without the accumulation of deleterious side products that typically plague continuous manufacturing processes.
How to Synthesize (R)-(+)-alpha-phenylethylamine Efficiently
The implementation of this recycling protocol requires precise control over reaction parameters to maximize yield and purity. The process begins with the treatment of the resolution mother liquor, where careful pH adjustment and solvent selection are paramount to ensure complete extraction of the free amine. Following the initial concentration, the critical crystallization step must be managed with strict temperature control to optimize crystal growth and impurity rejection. Operators must adhere to the specified solvent ratios and cooling rates to achieve the desired crystal morphology, which facilitates efficient filtration and washing.
- Adjust the free mother liquor to alkaline pH using inorganic base, extract with organic solvent A, and desolventize under controlled temperature.
- Dissolve the residue in organic solvent B, cool to below 10°C, and add inorganic acid to induce crystallization of the amine salt for purification.
- Dissolve the wet salt product in water, basify again, extract with organic solvent A, and perform reduced pressure distillation to collect the refined amine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this advanced recycling technology translates directly into enhanced operational resilience and significant cost optimization. By transforming a waste stream into a high-value resource, manufacturers can drastically reduce their dependency on external suppliers for fresh chiral amines, which are subject to market volatility and price fluctuations. The ability to internally regenerate a critical reagent with purity levels matching fresh material means that production schedules are no longer held hostage by raw material lead times. This self-sufficiency creates a buffer against supply chain disruptions, ensuring consistent output of perindopril intermediates even during periods of global chemical shortage. Moreover, the reduction in hazardous waste discharge aligns with increasingly stringent environmental regulations, mitigating the risk of regulatory fines and shutdowns.
- Cost Reduction in Manufacturing: The primary economic driver of this technology is the substantial decrease in raw material consumption. Since the resolving agent constitutes a significant portion of the variable cost in chiral resolution, recovering it with high efficiency directly improves the gross margin of the final API. The elimination of the need for frequent purchasing of fresh (R)-(+)-alpha-phenylethylamine allows capital to be redirected towards other critical areas of R&D or capacity expansion. Additionally, the recycling of organic solvents used in the process further compounds these savings, creating a leaner and more cost-effective manufacturing model that enhances competitiveness in the global market.
- Enhanced Supply Chain Reliability: Dependence on external vendors for specialty chiral chemicals introduces inherent risks related to logistics, quality consistency, and geopolitical factors. By internalizing the recovery process, a manufacturer secures a steady, on-demand supply of this critical reagent. This vertical integration reduces the lead time associated with sourcing and qualifying new batches of resolving agent, allowing for more agile response to market demand spikes. The consistent quality of the recycled amine also reduces the variability in the downstream synthesis of perindopril, leading to fewer batch failures and a more predictable supply of the final therapeutic ingredient to customers.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing standard unit operations such as extraction, crystallization, and distillation that are easily transferable from pilot to commercial scale. The reduction in ammonia nitrogen wastewater discharge and the efficient recycling of solvents demonstrate a commitment to green chemistry principles. This not only lowers the cost of waste treatment but also strengthens the company's ESG (Environmental, Social, and Governance) profile. For large-scale producers, the ability to operate a sustainable, low-waste process is increasingly becoming a prerequisite for securing contracts with major multinational pharmaceutical companies who prioritize environmentally responsible supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this recycling technology. Understanding these details is essential for R&D teams evaluating the feasibility of integrating this process into existing production lines. The answers are derived directly from the experimental data and process descriptions provided in the patent documentation, ensuring accuracy and relevance for technical decision-makers.
Q: Why is the traditional distillation method insufficient for recovering (R)-(+)-alpha-phenylethylamine?
A: Traditional methods relying solely on alkaline extraction and distillation often result in recovered amine with low GC purity (92-97%) and poor specific rotation due to oxidative degradation and impurity carryover, which negatively impacts subsequent resolution efficiency.
Q: How does the acidic crystallization step improve the quality of the recycled resolving agent?
A: The introduction of an acidic crystallization step allows for the selective precipitation of the amine salt, effectively separating it from non-basic organic impurities and oxidized byproducts that remain in the mother liquor, thereby achieving purity levels exceeding 99.5%.
Q: Can the solvents used in this recycling process be reused?
A: Yes, the process is designed for sustainability, allowing for the recycling of both Organic Solvent A and Organic Solvent B with recovery rates exceeding 75%, which significantly reduces waste discharge and operational costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (R)-(+)-alpha-phenylethylamine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the efficiency of your perindopril production line hinges on the quality and availability of your chiral building blocks. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with unwavering consistency. We understand the critical nature of stringent purity specifications and maintain rigorous QC labs to verify that every batch of (R)-(+)-alpha-phenylethylamine and its derivatives meets the highest industry standards. Our commitment to technical excellence allows us to support complex synthetic routes, providing you with a partner who understands the nuances of chiral resolution and intermediate synthesis.
We invite you to collaborate with us to optimize your supply chain and reduce your manufacturing costs. By leveraging our expertise in process optimization and recycling technologies, we can help you achieve greater efficiency in your production of cardiovascular intermediates. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific production volumes. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our solutions can enhance your operational performance and secure your position in the competitive pharmaceutical market.
