Revolutionizing Olopatadine Intermediate Production: A Safer, High-Yield One-Pot Synthesis Strategy
The pharmaceutical industry continuously seeks robust manufacturing pathways for critical antihistamine agents, particularly olopatadine hydrochloride, a potent H1 receptor antagonist widely used in treating allergic conjunctivitis and rhinitis. A pivotal breakthrough in this domain is detailed in patent CN111548369B, which discloses a highly efficient, one-pot synthesis method for preparing [3-(dimethylamine)propyl]triphenylphosphonium bromide hydrobromide (Compound 123B2-10). This compound serves as an essential Wittig reagent precursor in the commercial production of olopatadine. Traditional manufacturing routes have long struggled with safety hazards associated with genotoxic intermediates and complex solvent recovery systems. The disclosed innovation addresses these pain points by utilizing n-heptane as a safer reaction medium and eliminating the isolation of hazardous intermediates, thereby offering a compelling value proposition for a reliable olopatadine intermediate supplier aiming to optimize their supply chain.
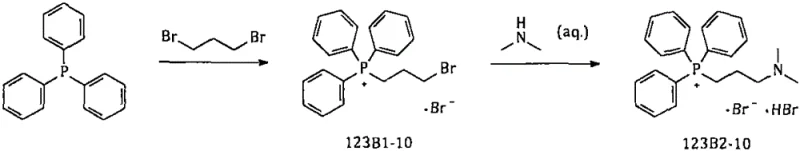
The strategic importance of this intermediate cannot be overstated, as the subsequent Wittig reaction requires a significant molar excess of the phosphonium salt—approximately five times that of the isoxolic acid partner—to drive conversion to completion. Consequently, the demand for high-purity 123B2-10 is substantial, with roughly 15-18 kg of intermediate required to produce just 1 kg of the final active pharmaceutical ingredient (API). The patent outlines a transformative approach that shifts away from the conventional use of toluene and isolated intermediate steps, moving instead toward a streamlined, continuous process that enhances both yield and operational safety.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
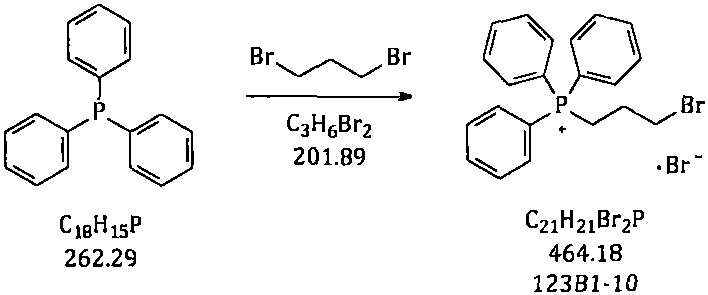
Historically, the synthesis of this key phosphonium salt has relied on a two-step process involving toluene as the primary solvent. In this legacy workflow, triphenylphosphine reacts with 1,3-dibromopropane to form the quaternary salt (123B1-10), which must then be isolated via centrifugation and drying before undergoing aminolysis with dimethylamine. This approach presents severe Environmental, Health, and Safety (EHS) challenges. The intermediate 123B1-10 is a potent alkylating agent with genotoxic potential, and the necessity of handling it in a solid state during centrifugation and packaging exposes operators to significant health risks, including skin allergies and respiratory irritation. Furthermore, the process typically requires a large excess of 1,3-dibromopropane to suppress the formation of the double-substituted byproduct (123B1-X2), leading to residual toxic reagents that are difficult to remove completely.
Additionally, the conventional aminolysis step often employs ethanol under severe reflux conditions (70-80°C). Since the boiling point of the dimethylamine aqueous solution is approximately 50°C, this thermal mismatch causes substantial volatilization of the amine reagent, resulting in material waste and increased burden on waste gas treatment systems. The post-treatment phase is equally problematic; removing the large volume of water introduced by the aqueous amine requires energy-intensive concentration steps. If not managed perfectly, residual water can cause the reaction mass to harden into blocks that lock stirring equipment, leading to production downtime and potential mechanical damage.
The Novel Approach
The methodology described in patent CN111548369B fundamentally reengineers this synthesis to mitigate these risks through a clever one-pot design. By switching the solvent system to n-heptane, the process leverages a non-polar medium that significantly reduces the formation of the double-connected impurity (123B1-X2) compared to protic solvents like ethanol. More critically, the novel route eliminates the isolation of the toxic intermediate 123B1-10. Instead, the reaction mixture containing the quaternary salt is directly treated with aqueous dimethylamine in the same vessel. This "telescoped" approach ensures that any residual 1,3-dibromopropane reacts immediately with the amine to form a harmless diamine salt, effectively neutralizing the genotoxic hazard before workup begins.
This innovation also optimizes the thermal profile of the aminolysis step. By conducting the reaction at a controlled temperature of 45-50°C, which aligns closely with the boiling point of the dimethylamine solution, the process minimizes reagent loss due to volatilization. The use of n-heptane further enhances safety during the final filtration; unlike toluene, n-heptane is less prone to static electricity accumulation, drastically reducing the risk of flash explosions during solid-liquid separation. This holistic redesign not only simplifies the operational workflow but also delivers a product with superior purity profiles, making it an ideal candidate for cost reduction in pharmaceutical intermediates manufacturing.
Mechanistic Insights into Quaternization and Aminolysis
The core of this synthesis relies on a nucleophilic substitution mechanism, specifically the Menshutkin reaction, where the lone pair of electrons on the phosphorus atom of triphenylphosphine attacks the terminal carbon of 1,3-dibromopropane. This initial quaternization step forms the phosphonium bromide salt (123B1-10). The choice of solvent plays a pivotal role here; in polar protic solvents, the solvation of the bromide anion can facilitate a second substitution event where another molecule of triphenylphosphine attacks the remaining bromine, forming the undesirable bis-phosphonium byproduct (123B1-X2). By utilizing n-heptane, a non-polar solvent, the ion pairing is tighter, and the reactivity of the remaining bromine is modulated, kinetically favoring the mono-substituted product.
Following the formation of 123B1-10, the system undergoes aminolysis. The dimethylamine acts as a nucleophile, displacing the bromide on the propyl chain to install the tertiary amine functionality. A critical mechanistic advantage of the one-pot method is the in-situ scavenging of impurities. Any unreacted 1,3-dibromopropane remaining from the first step reacts rapidly with the excess dimethylamine to form N1,N1,N3,N3-tetramethyl-1,3-propanediamine dihydrobromide. This byproduct, along with the double-connected phosphonium impurity (123B1-X2), exhibits high solubility in the final ethanol pulping wash. Consequently, these contaminants remain in the mother liquor while the desired product (123B2-10) crystallizes out, ensuring that the final high-purity phosphonium salt meets stringent quality specifications without requiring complex chromatographic purification.
How to Synthesize [3-(dimethylamine)propyl]triphenylphosphonium Bromide Hydrobromide Efficiently
The execution of this synthesis requires precise control over reaction stoichiometry and thermal conditions to maximize yield while maintaining safety. The patent data suggests that a molar ratio of triphenylphosphine to 1,3-dibromopropane close to 1:1 is optimal when using n-heptane, contrasting with the excess reagent typically needed in toluene. The reaction is heated to reflux for approximately 60 hours to ensure complete conversion before the addition of the amine. Following the aminolysis at 45-50°C for roughly 4 to 5 hours, the system is concentrated to remove water and solvent. The resulting crude solid is then subjected to a hot pulping process with absolute ethanol, which serves as the critical purification step to remove soluble impurities.
- Reflux triphenylphosphine and 1,3-dibromopropane in n-heptane for approximately 60 hours to form the quaternary phosphonium salt dispersion.
- Without isolating the intermediate, add 40% aqueous dimethylamine solution directly to the reaction mixture and heat to 45-50°C for aminolysis.
- Concentrate the system to remove water and n-heptane, then purify the crude solid via hot pulping with absolute ethanol to obtain the final high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented process translates into tangible operational improvements beyond mere chemical yield. The shift from a multi-step isolation process to a one-pot telescoped reaction significantly reduces the number of unit operations, such as centrifugation and drying of hazardous intermediates. This reduction in processing steps directly correlates to lower labor costs, decreased equipment wear and tear, and a smaller physical footprint for production. Furthermore, the elimination of toxic intermediate handling simplifies regulatory compliance and reduces the need for specialized personal protective equipment (PPE) protocols, fostering a safer working environment.
- Cost Reduction in Manufacturing: The process achieves significant cost optimization by eliminating the need for expensive solvent swaps and extensive waste treatment associated with toluene and excess amine volatilization. By preventing the loss of dimethylamine through controlled temperature reflux and avoiding the yield losses inherent in isolating the intermediate 123B1-10, the overall material efficiency is drastically improved. Patent data indicates a total yield increase from approximately 71.7% in the conventional route to over 90% in the new process, representing a substantial reduction in the cost of goods sold (COGS) per kilogram of product.
- Enhanced Supply Chain Reliability: The use of n-heptane, a commodity solvent with stable pricing and availability, mitigates supply risks associated with more regulated solvents like toluene. Additionally, the robustness of the one-pot method against impurity carryover ensures consistent batch-to-batch quality. This reliability is crucial for maintaining uninterrupted supply lines for downstream API manufacturers, reducing the risk of production delays caused by out-of-specification intermediate batches.
- Scalability and Environmental Compliance: From an EHS perspective, the replacement of toluene with n-heptane reduces the risk of static-induced fires, a critical factor when scaling up to multi-ton production volumes. The process also generates less hazardous waste, as the toxic alkylating agents are consumed in situ rather than being separated and disposed of. This alignment with green chemistry principles facilitates easier permitting and reduces the environmental liability for manufacturing sites, supporting the commercial scale-up of complex quaternary ammonium salts.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, offering clarity on how this method outperforms legacy technologies in terms of safety, purity, and efficiency.
Q: How does the new one-pot method improve operator safety compared to traditional processes?
A: The novel method eliminates the centrifugal separation and drying of the toxic intermediate (3-bromopropyl)triphenylphosphonium bromide. By reacting in a closed one-pot system, exposure to genotoxic alkylating agents and residual 1,3-dibromopropane is significantly minimized, reducing risks of allergic reactions and chemical injury.
Q: Why is n-heptane preferred over toluene or ethanol as the reaction solvent?
A: N-heptane offers superior safety characteristics regarding static electricity accumulation compared to toluene. Furthermore, unlike protic solvents like ethanol which promote double-substituted byproducts, or esters which risk transesterification, n-heptane provides an optimal balance of conversion efficiency and impurity control for this specific quaternization reaction.
Q: What is the impact of this process on the purity of the final olopatadine intermediate?
A: The process achieves exceptional purity levels, with HPLC data indicating purity greater than 99.9%. The one-pot strategy effectively manages impurities like the double-connected byproduct (123B1-X2) by keeping them in solution during the final ethanol pulping step, avoiding the carryover issues common in multi-step isolation processes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable [3-(dimethylamine)propyl]triphenylphosphonium Bromide Hydrobromide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the global pharmaceutical supply chain. Our technical team has extensively analyzed the pathway described in CN111548369B and possesses the expertise to implement this advanced one-pot synthesis at an industrial scale. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory optimization to full-scale manufacturing is seamless. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of 123B2-10 meets the exacting standards required for olopatadine synthesis.
We invite potential partners to engage with our technical procurement team to discuss how this optimized route can benefit your specific production needs. By leveraging our manufacturing capabilities, you can secure a stable supply of this critical intermediate while benefiting from the inherent cost and safety advantages of the new process. Please contact us to request a Customized Cost-Saving Analysis, along with specific COA data and route feasibility assessments tailored to your project requirements.
