Advanced Benzanthracene Derivatives for OLEDs: Scalable Synthesis and Commercial Viability
Advanced Benzanthracene Derivatives for OLEDs: Scalable Synthesis and Commercial Viability
The rapid evolution of the organic light-emitting diode (OLED) industry demands materials that not only exhibit superior electroluminescent efficiency but also possess manufacturability at a commercial scale. Patent CN102925139A introduces a groundbreaking class of benzanthracene-based organic luminescent materials that address the critical bottlenecks of solubility and purification often encountered in traditional anthracene derivatives. By leveraging a robust Suzuki-Miyaura coupling strategy, this technology enables the precise construction of large conjugated systems tailored for high-performance blue emission and charge transport layers. For R&D directors and procurement specialists seeking a reliable OLED material supplier, understanding the mechanistic advantages and supply chain implications of this patented route is essential for securing a competitive edge in next-generation display manufacturing.
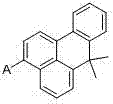
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of high-performance anthracene-based OLED materials has been plagued by significant technical hurdles that impede industrial adoption. Traditional routes often involve harsh reaction conditions, such as high-temperature cyclizations or the use of toxic reagents, which complicate safety protocols and waste management. Furthermore, the planar nature of unsubstituted anthracene cores frequently leads to strong intermolecular pi-pi stacking, resulting in poor solubility in common processing solvents and severe aggregation-caused quenching (ACQ) in the solid state. This necessitates complex and costly purification steps, such as repeated sublimation or column chromatography, to achieve the electronic-grade purity required for device longevity. These factors collectively drive up the cost of goods sold (COGS) and extend lead times, creating a fragile supply chain for high-purity electronic chemical manufacturing.
The Novel Approach
The methodology disclosed in the patent represents a paradigm shift by utilizing a modular Suzuki coupling approach centered on a 3-bromo-7,7-dimethyl-7H-benzanthracene intermediate. This strategy allows for the late-stage introduction of diverse aromatic functionalities through mild cross-coupling reactions, avoiding the need for destructive high-energy transformations. The incorporation of the gem-dimethyl group at the 7-position is a critical design element that disrupts molecular planarity, thereby drastically improving solubility and preventing detrimental aggregation without sacrificing the conjugation length necessary for efficient charge transport. This novel approach not only simplifies the synthetic workflow but also ensures that the final derivatives possess excellent film-forming properties, making them ideal candidates for both vacuum deposition and solution-processing techniques in modern OLED architectures.
Mechanistic Insights into Pd-Catalyzed Suzuki Coupling
The core of this synthesis lies in the palladium-catalyzed cross-coupling between the brominated benzanthracene core and various arylboronic acids. The catalytic cycle initiates with the oxidative addition of the palladium(0) species into the carbon-bromine bond of the 3-bromo-7,7-dimethyl-7H-benzanthracene, forming a reactive organopalladium intermediate. This step is facilitated by the electron-deficient nature of the benzanthracene ring system, which activates the C-Br bond towards metal insertion. Subsequently, the transmetallation step occurs where the aryl group from the boronic acid, activated by the base (typically sodium carbonate), transfers to the palladium center. The steric bulk of the triphenylphosphine ligands on the catalyst helps stabilize the active species while allowing sufficient access for the bulky benzanthracene substrate. Finally, reductive elimination releases the coupled product and regenerates the active Pd(0) catalyst, completing the cycle with high turnover numbers.
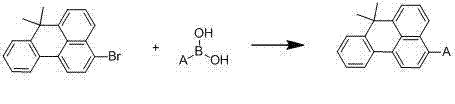
Impurity control in this process is inherently managed by the specificity of the catalytic cycle and the physical properties of the product. Unlike Friedel-Crafts alkylations which can produce multiple regioisomers, the Suzuki coupling is highly regioselective, occurring exclusively at the bromine substitution site. This minimizes the formation of structural isomers that are notoriously difficult to separate. Furthermore, the byproduct of the reaction is primarily inorganic boron salts, which are easily removed during the aqueous workup phase. The patent highlights that the final products precipitate out of the reaction mixture upon cooling, allowing for isolation via simple suction filtration. This precipitation behavior acts as a self-purification mechanism, as soluble impurities and catalyst residues remain in the mother liquor, ensuring that the crude solid already possesses high purity levels prior to any final recrystallization or washing steps.
How to Synthesize Benzanthracene Derivatives Efficiently
The practical implementation of this synthesis route is designed for operational simplicity and reproducibility, making it highly attractive for contract development and manufacturing organizations (CDMOs). The process begins by charging the key building blocks, 3-bromo-7,7-dimethyl-7H-benzanthracene and the specific substituted boronic acid, into a reaction vessel along with a biphasic solvent system of tetrahydrofuran (THF) and water. The use of a mixed solvent system is crucial for solubilizing both the organic halide and the inorganic base, facilitating the interfacial reaction. As demonstrated in the specific examples provided in the patent, such as the synthesis of Compound 001, the reaction proceeds smoothly under a nitrogen atmosphere to prevent catalyst oxidation.
- Charge 3-bromo-7,7-dimethyl-7H-benzanthracene and substituted boronic acid into a reactor with THF/water solvent and sodium carbonate base.
- Degas the system with nitrogen, add tetrakis(triphenylphosphine)palladium catalyst, and heat to 70-100°C for 10-30 hours.
- Cool the reaction, filter the precipitated solid, wash thoroughly with water, ethanol, and ether, and dry to obtain the final product.
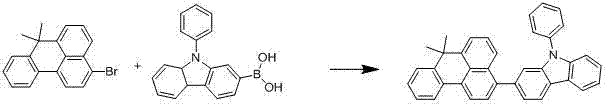
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this benzanthracene synthesis platform offers tangible strategic benefits that extend beyond mere technical performance. The reliance on commodity chemicals such as sodium carbonate and widely available palladium catalysts reduces dependency on exotic or single-source reagents, thereby mitigating supply risk. The moderate reaction temperatures ranging from 70°C to 100°C mean that standard glass-lined or stainless steel reactors can be utilized without the need for specialized high-pressure or cryogenic equipment. This compatibility with existing infrastructure significantly lowers the barrier to entry for scale-up, allowing manufacturers to ramp up production capacity rapidly in response to market demand fluctuations without substantial capital expenditure on new plant assets.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the elimination of expensive and time-consuming purification steps. Since the product precipitates directly from the reaction mixture, the need for large-scale column chromatography is obviated, leading to substantial savings in silica gel, solvents, and labor costs. Additionally, the high yields reported in the patent examples, consistently exceeding 90%, maximize the throughput of raw materials, reducing the effective cost per kilogram of the final API intermediate. The ability to recycle the aqueous phase containing the base further contributes to waste reduction and overall process economy, aligning with green chemistry principles that are increasingly mandated by global regulatory bodies.
- Enhanced Supply Chain Reliability: The modular nature of the Suzuki coupling allows for flexible manufacturing strategies. Different derivatives can be produced on the same production line simply by swapping the boronic acid feedstock, which are generally stable and commercially accessible. This flexibility ensures that a reliable OLED material supplier can maintain continuity of supply even if specific downstream device requirements change. The robustness of the reaction conditions also means that batch-to-batch variability is minimized, ensuring consistent quality that meets the stringent specifications of display panel manufacturers. This reliability is critical for maintaining just-in-time inventory models and avoiding production stoppages in the highly competitive consumer electronics sector.
- Scalability and Environmental Compliance: Scaling this chemistry from gram to tonnage scales is straightforward due to the absence of hazardous reagents or extreme conditions. The primary waste streams are aqueous salts and organic solvents, both of which are amenable to standard treatment and recovery processes. The use of water as a co-solvent reduces the overall volume of volatile organic compounds (VOCs) emitted, simplifying compliance with environmental regulations. Furthermore, the high atom economy of the coupling reaction ensures that the majority of the reactant mass is incorporated into the final product, minimizing the generation of heavy organic waste. This environmental profile makes the process sustainable for long-term commercial scale-up of complex electronic chemicals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these benzanthracene derivatives. The answers are derived directly from the experimental data and technical disclosures within the patent documentation, providing a factual basis for decision-making. Understanding these details is vital for stakeholders evaluating the feasibility of integrating these materials into their existing supply chains or device architectures.
Q: What are the key purity specifications for these benzanthracene derivatives?
A: According to the patent data, the synthesized materials achieve HPLC purity greater than 98% through simple filtration and washing steps, eliminating the need for complex chromatographic purification which is often required for traditional OLED intermediates.
Q: How does the 7,7-dimethyl substitution affect material performance?
A: The introduction of the 7,7-dimethyl group significantly enhances the solubility of the benzanthracene core in common organic solvents and improves film-forming properties, which is critical for solution-processable OLED device fabrication.
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, the process utilizes standard Suzuki coupling conditions with readily available reagents like sodium carbonate and palladium catalysts, operating at moderate temperatures (70-100°C), making it highly scalable for commercial manufacturing without specialized high-pressure equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzanthracene Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-performance organic semiconductors play in the future of display technology. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. We are equipped with rigorous QC labs capable of verifying stringent purity specifications, including trace metal analysis to ensure palladium levels meet the demanding requirements of OLED fabrication. Our commitment to quality assurance guarantees that every batch of benzanthracene derivative delivered meets the exacting standards necessary for high-efficiency, long-lifetime devices.
We invite potential partners to engage with our technical procurement team to discuss how this advanced synthesis route can be adapted to your specific material needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how optimizing the supply of these key intermediates can impact your overall manufacturing budget. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your next-generation OLED projects are built on a foundation of reliable, high-quality chemical supply.
