Advanced Manufacturing of Halauxifen-methyl Intermediates via Optimized Suzuki Coupling
Advanced Manufacturing of Halauxifen-methyl Intermediates via Optimized Suzuki Coupling
The global demand for high-efficiency herbicides continues to drive innovation in agrochemical intermediate manufacturing, particularly for synthetic auxins like Halauxifen-methyl. Patent CN113603637A, published in late 2021, introduces a transformative preparation method for fluorochloropyridine ester, chemically known as 4-amino-3-chloro-6-(2-fluoro-4-chloro-3-methoxyphenyl)pyridine-2-methyl formate. This technical disclosure addresses critical bottlenecks in existing supply chains by replacing hazardous reagents and low-yield pathways with a streamlined, three-step protocol involving amino protection, Suzuki coupling, and acidic deprotection. For R&D directors and procurement strategists, this patent represents a pivotal shift towards safer, more cost-effective production of this key active ingredient precursor. The structural integrity and functional groups of the target molecule are depicted below, highlighting the complexity that necessitates such precise synthetic control.
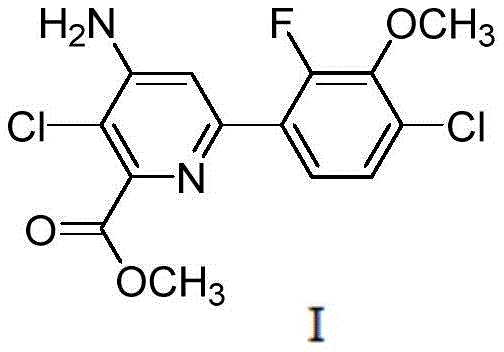
As a reliable agrochemical intermediate supplier, understanding the nuances of this new pathway is essential for maintaining competitive advantage in the herbicide market. The method leverages easily obtainable raw materials and avoids the need for specialized production equipment, which directly translates to reduced capital expenditure and operational risk. By achieving reaction yields of more than 70% at each step and final product purity exceeding 99%, this technology offers a compelling value proposition for manufacturers seeking to optimize their cost reduction in herbicide manufacturing strategies while adhering to increasingly stringent environmental and safety regulations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of fluorochloropyridine ester has been plagued by significant safety hazards and inefficiencies that hinder large-scale commercial viability. As illustrated in the prior art documentation, Route 1 (WO2007082098) relies on a sequence involving lithiation, boration, and methylation. This pathway is fraught with danger, utilizing trimethyl borate and easily exploded peroxyacetic acid in the initial hydroxylation steps. Furthermore, the methylation stage employs methyl iodide, a known carcinogen that poses severe health risks to operators and requires expensive containment and waste treatment systems. The subsequent boration steps involve dangerous chemicals like triisopropyl borate and acetyl chloride, creating a cumulative risk profile that is untenable for modern, sustainable industrial production.
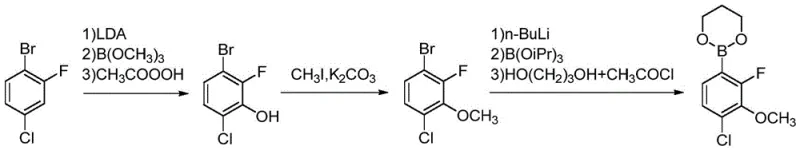
Alternative pathways have also failed to provide a robust solution for the commercial scale-up of complex agrochemical intermediates. Route 3 (WO2013102078), for instance, utilizes 4-acetamido-3,6-dichloropyridine-2-methyl formate as a starting material. While the coupling yield might appear acceptable, the starting material itself is notoriously difficult to source commercially, creating a fragile supply chain dependency. Moreover, literature data indicates that preparing this specific starting material involves refluxing with acetic anhydride for extended periods, resulting in a dismal total route yield of merely 27.20%. Such low efficiency results in massive material waste and inflated production costs, rendering these conventional methods economically unviable for high-volume manufacturing.
The Novel Approach
In stark contrast to these legacy processes, the methodology disclosed in CN113603637A introduces a sophisticated yet operationally simple strategy centered on amino protection. The core innovation lies in the conversion of 4-amino-3,6-dichloropyridine-2-methyl formate into a protected intermediate using di-tert-butyl dicarbonate. This protection step is not merely a defensive maneuver; it fundamentally alters the reactivity profile of the molecule, stabilizing the ester bond against hydrolysis during the harsh conditions of the subsequent coupling reaction. The overall process flow, as detailed in the patent, streamlines the synthesis into three distinct, high-yielding stages that eliminate the need for carcinogenic methylating agents or explosive peroxides.
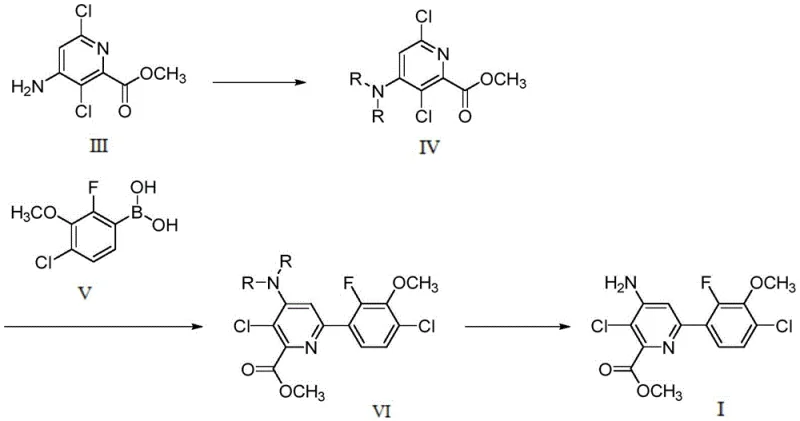
The novelty of this approach extends to its remarkable adaptability and efficiency. By employing a protection reagent sensitive to acidic conditions, the process ensures that the final deprotection step can be executed cleanly without compromising the structural integrity of the sensitive ester moiety. The reaction conditions are mild, typically operating between 50°C and 150°C for the coupling stage, and do not require exotic high-pressure reactors. This simplicity allows for a drastic simplification of post-treatment operations, as the product can be isolated through standard filtration and washing procedures rather than complex chromatographic separations. For supply chain heads, this translates to reducing lead time for high-purity agrochemical intermediates, as the simplified workflow accelerates batch turnover and minimizes downtime between production cycles.
Mechanistic Insights into Amino-Protection Assisted Suzuki Coupling
The success of this synthetic route hinges on the precise orchestration of the Suzuki-Miyaura coupling reaction facilitated by the strategic placement of the amino protecting group. In the coupling stage, the protected pyridine intermediate reacts with 4-chloro-2-fluoro-3-methoxyphenylboronic acid in the presence of a palladium catalyst, such as [1,1'-bis(diphenylphosphino)ferrocene]palladium dichloride or palladium acetate. The mechanism involves the oxidative addition of the palladium catalyst to the chloro-substituted pyridine ring, followed by transmetallation with the boronic acid species activated by a base like cesium carbonate or potassium carbonate. The presence of the bulky tert-butoxycarbonyl (Boc) groups on the amino nitrogen sterically shields the adjacent ester functionality, preventing nucleophilic attack by the base or solvent which would otherwise lead to ester hydrolysis and the formation of carboxylic acid impurities.
Furthermore, the choice of solvent system plays a critical role in maximizing yield and purity. The patent specifies the use of mixed solvents such as 1,4-dioxane and water or tetrahydrofuran and water, which facilitate the solubility of both the organic intermediates and the inorganic base. This biphasic or homogeneous environment ensures efficient mass transfer during the catalytic cycle. The deprotection mechanism subsequently utilizes acidic conditions, typically with hydrogen chloride or trifluoroacetic acid, to cleave the acid-sensitive Boc groups. This step regenerates the free amino group essential for the biological activity of the final herbicide while leaving the newly formed biaryl bond and the methyl ester intact. This orthogonal stability—stable to base during coupling, labile to acid during deprotection—is the key mechanistic feature that enables the high purity (>99%) reported in the examples.
How to Synthesize Fluorochloropyridine Ester Efficiently
The synthesis of this high-value intermediate requires strict adherence to the optimized parameters outlined in the patent to ensure reproducibility and safety. The process begins with the preparation of the starting material, methyl 4-amino-3,6-dichloropyridine-2-carboxylate, which can be efficiently synthesized from the corresponding carboxylic acid using thionyl chloride or concentrated sulfuric acid in methanol. Following this, the critical amino protection step must be conducted at controlled temperatures (-5°C to 0°C initially) to manage the exotherm associated with the addition of di-tert-butyl dicarbonate. The subsequent coupling reaction demands careful selection of the palladium catalyst loading (typically 1:0.01 to 1:0.15 molar ratio) and base to drive the reaction to completion within 8 to 10 hours. Detailed standardized synthesis steps see the guide below.
- Perform amino protection on 4-amino-3,6-dichloropyridine-2-methyl formate using di-tert-butyl dicarbonate to stabilize the amine group.
- Execute a Suzuki coupling reaction between the protected pyridine intermediate and 4-chloro-2-fluoro-3-methoxyphenylboronic acid using a palladium catalyst.
- Conduct an acidic deprotection step using hydrogen chloride or trifluoroacetic acid to remove the protecting group and yield the final fluorochloropyridine ester.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented methodology offers substantial strategic benefits that extend beyond mere technical feasibility. The elimination of hazardous reagents such as methyl iodide and peroxyacetic acid removes the need for specialized storage facilities and expensive hazardous waste disposal protocols. This shift significantly lowers the operational overhead associated with regulatory compliance and environmental health and safety (EHS) management. By transitioning to a process that utilizes common, non-carcinogenic reagents, manufacturers can mitigate the risk of supply disruptions caused by the tightening regulations on toxic chemicals, thereby ensuring a more resilient and continuous supply of critical agrochemical intermediates.
- Cost Reduction in Manufacturing: The economic impact of this new route is driven primarily by the dramatic improvement in material efficiency and yield. With each reaction step achieving yields exceeding 70% and the protection step reaching over 90%, the overall material throughput is vastly superior to the 27.20% total yield of conventional Route 3. This higher efficiency means less raw material is required to produce the same amount of final product, directly lowering the cost of goods sold (COGS). Additionally, the avoidance of expensive purification techniques like column chromatography, replaced instead by simple crystallization and filtration, reduces labor costs and solvent consumption, leading to substantial cost savings in herbicide manufacturing.
- Enhanced Supply Chain Reliability: The reliance on easily obtainable raw materials is a critical factor for supply chain stability. Unlike Route 3, which depends on hard-to-source acetamido precursors, this method starts with readily available 4-amino-3,6-dichloropyridine-2-carboxylic acid and common boronic acids. This accessibility reduces the risk of vendor lock-in and price volatility. Furthermore, the mild reaction conditions and lack of requirement for special production equipment mean that the process can be easily transferred to multiple manufacturing sites or scaled up rapidly to meet surging market demand without significant capital investment in new infrastructure.
- Scalability and Environmental Compliance: From an environmental perspective, this process aligns perfectly with green chemistry principles. The absence of heavy metal contaminants (beyond the catalytic palladium which is recoverable) and carcinogenic byproducts simplifies the wastewater treatment process. The high purity of the crude product (>99% after simple workup) minimizes the generation of organic waste streams associated with extensive purification. This environmental friendliness not only reduces disposal costs but also enhances the corporate sustainability profile of the manufacturer, a key metric for modern agricultural clients who are increasingly demanding eco-friendly supply chains for their crop protection products.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on yield expectations, safety profiles, and scalability potential for industrial partners considering this route for their production portfolios.
Q: Why is amino protection critical in this synthesis route?
A: Amino protection using reagents sensitive to acidic conditions, such as di-tert-butyl dicarbonate, significantly improves the stability of ester bonds during the subsequent coupling and deprotection stages. This strategy minimizes side reactions like ester hydrolysis, thereby simplifying post-treatment and ensuring final product purity exceeds 99%.
Q: How does this method compare to prior art routes regarding safety?
A: Unlike conventional Route 1 which utilizes carcinogenic methyl iodide and explosive peroxyacetic acid, or Route 3 which suffers from low yields and difficult-to-obtain raw materials, this novel approach operates under mild conditions without special production equipment. It eliminates the need for highly toxic reagents, making it far more suitable for industrial scale-up.
Q: What yields can be expected from this preparation method?
A: The patent data indicates that the reaction yield for each individual step exceeds 70%, with the amino protection step specifically reaching over 90%. The final product achieves a chemical purity of greater than 99%, demonstrating high efficiency and robustness for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fluorochloropyridine Ester Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory patent to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising yields and purity levels described in CN113603637A can be reliably replicated on an industrial scale. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch of Halauxifen-methyl intermediate meets the exacting standards required by global agrochemical formulators.
We invite you to collaborate with us to leverage this advanced synthesis technology for your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized manufacturing processes can enhance your operational efficiency and reduce your overall procurement costs.
