Advanced Oxidation Technology for High-Purity Pantoprazole and PPI Intermediates Manufacturing
The pharmaceutical industry continuously seeks robust, scalable, and cost-efficient synthetic routes for Proton Pump Inhibitors (PPIs), a class of drugs essential for treating gastric acid-related disorders. Patent CN1839127A introduces a transformative methodology for preparing 2-[(pyridyl)methyl]sulfinyl substituted benzimidazoles, including key active pharmaceutical ingredients like Pantoprazole, Lansoprazole, Omeprazole, and Rabeprazole. This technology addresses critical bottlenecks in traditional manufacturing by replacing expensive and hazardous peracid oxidants with controlled sodium hypochlorite systems. For R&D directors and procurement specialists, this shift represents a significant opportunity to optimize the cost of goods sold (COGS) while enhancing the purity profile of the final API intermediate. The process leverages precise stoichiometric control and temperature management to achieve high selectivity, ensuring that the valuable sulfoxide moiety is preserved without degradation into inactive sulfone byproducts.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the oxidation of sulfide precursors to sulfoxides in PPI synthesis has relied heavily on peracids, most notably m-chloroperbenzoic acid (MCPBA). While chemically effective, MCPBA presents substantial commercial and technical drawbacks that hinder large-scale manufacturing efficiency. The primary concern is the tendency of peracids to over-oxidize the sulfur atom, leading to the formation of sulfone impurities, as illustrated in the reaction pathway below. These sulfone byproducts are not only pharmacologically inactive but are also structurally similar to the target molecule, making their removal via crystallization or chromatography difficult and yield-reducing. Additionally, MCPBA is a specialized reagent with a higher cost basis and stricter storage requirements due to its potential instability and shock sensitivity, creating unnecessary supply chain vulnerabilities and safety hazards in a production environment.
The Novel Approach
The methodology disclosed in CN1839127A fundamentally reengineers this oxidation step by utilizing sodium hypochlorite (NaOCl) under carefully regulated alkaline conditions. Unlike the aggressive nature of peracids, aqueous sodium hypochlorite acts as a mild yet effective oxidant when the reaction environment is strictly controlled. By employing a molar equivalent ratio of oxidant to thioether between 1.05 and 1.6, and maintaining the reaction temperature at or below 5°C, the process kinetically favors the formation of the sulfoxide while thermodynamically suppressing further oxidation to the sulfone. This approach transforms the oxidation step from a high-risk purification challenge into a robust, high-yielding unit operation. The use of commodity-grade bleach eliminates the need for expensive specialized oxidants, drastically simplifying the raw material sourcing strategy and reducing the overall environmental footprint of the synthesis through the generation of benign salt byproducts.
Mechanistic Insights into NaOCl-Catalyzed Selective Oxidation
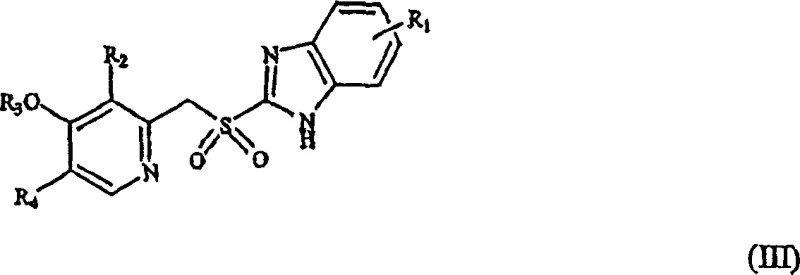
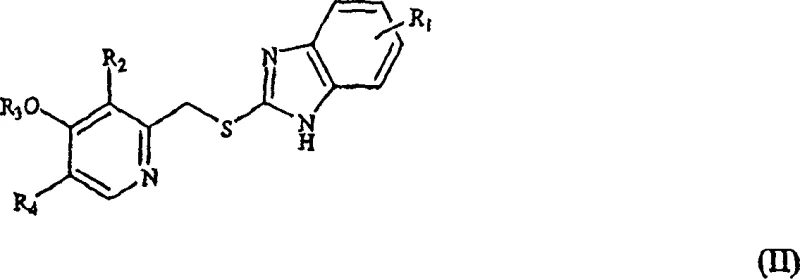
The core of this technological advancement lies in the precise manipulation of reaction kinetics to favor the sulfoxide state. The general transformation involves the conversion of a 2-[(pyridyl)methyl]thio-substituted benzimidazole (Formula II) into the corresponding sulfoxide (Formula I). In this mechanism, the active chlorine species generated from sodium hypochlorite in the presence of a strong base, such as sodium hydroxide, acts as the oxygen transfer agent. The presence of the base is critical not only for solubilizing the reactants but also for modulating the electrophilicity of the chlorine species, ensuring it reacts selectively with the sulfur lone pair without attacking other sensitive functional groups on the pyridine or benzimidazole rings. This selectivity is paramount for maintaining the integrity of complex substituents like difluoromethoxy or trifluoroethoxy groups found in Pantoprazole and Lansoprazole.
Furthermore, the patent elucidates the formation and identification of novel chlorinated derivatives that may arise as trace impurities under specific conditions. Compounds such as 5-(difluoromethoxy)-2-[[(3,4-dimethoxy-2-pyridyl)-chloromethyl]sulfinyl]-1H-benzimidazole (Compound IV) and its hydroxymethyl analog (Compound V) are identified as potential side products. Understanding the mechanistic origin of these chlorinated species allows process chemists to fine-tune reaction parameters, such as pH and addition rates, to minimize their formation. The ability to quantify and identify these specific impurities using advanced chromatographic techniques ensures that the final API meets stringent regulatory standards for impurity profiles. This deep mechanistic understanding provides a solid foundation for scaling the process from laboratory benchtop to multi-ton commercial production without unexpected purity deviations.
How to Synthesize Pantoprazole Efficiently
The synthesis of Pantoprazole via this oxidative route requires strict adherence to thermal and stoichiometric parameters to ensure optimal yield and purity. The process begins with the dissolution of the sulfide precursor in a suitable organic solvent, followed by the establishment of a strongly alkaline environment. The subsequent addition of the oxidant must be performed slowly to manage the exotherm and prevent local hotspots that could trigger over-oxidation. Detailed standard operating procedures regarding solvent selection, quenching agents, and isolation techniques are critical for reproducibility. For a comprehensive guide on the specific experimental conditions and workup procedures validated in the patent examples, please refer to the standardized synthesis protocol outlined below.
- Dissolve the thioether precursor in an organic solvent such as ethyl acetate or acetonitrile and add an alkali metal base like sodium hydroxide to create alkaline conditions.
- Cool the reaction mixture to temperatures below 5°C, preferably near 0°C, and slowly add an aqueous solution of sodium hypochlorite (1.05 to 1.6 molar equivalents).
- Quench excess oxidant with sodium metabisulfite or thiosulfate, adjust pH to approximately 8.0-8.5, and recover the high-purity sulfoxide product via crystallization or phase separation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this sodium hypochlorite-based oxidation process offers compelling strategic advantages beyond mere technical feasibility. The shift from specialized peracids to commodity bleach fundamentally alters the cost structure of PPI intermediate manufacturing. Sodium hypochlorite is produced globally in massive quantities for water treatment and sanitation, ensuring a stable, abundant, and low-cost supply chain that is immune to the fluctuations often seen in the market for fine chemical oxidants. This reliability reduces the risk of production stoppages due to raw material shortages and allows for more accurate long-term budgeting and cost forecasting. Furthermore, the simplified waste stream, consisting primarily of inorganic salts rather than chlorinated organic byproducts from peracid decomposition, lowers the complexity and cost associated with effluent treatment and environmental compliance.
- Cost Reduction in Manufacturing: The replacement of m-chloroperbenzoic acid (MCPBA) with sodium hypochlorite results in a drastic reduction in raw material costs. MCPBA is a high-value specialty chemical that requires careful handling and storage, adding logistical overheads. In contrast, sodium hypochlorite is a bulk commodity available at a fraction of the cost. Additionally, the high selectivity of the new process minimizes the loss of valuable starting materials to over-oxidized sulfone byproducts, thereby improving the overall mass balance and effective yield of the process. This combination of lower input costs and higher material efficiency translates directly into a more competitive cost of goods sold for the final pharmaceutical intermediate.
- Enhanced Supply Chain Reliability: Relying on a single-source supplier for a specialized oxidant like MCPBA creates a single point of failure in the supply chain. By switching to sodium hypochlorite, manufacturers can source this critical reagent from multiple regional suppliers, significantly mitigating supply risk. The robustness of the process also means that it is less sensitive to minor variations in reagent quality, provided the active chlorine content is verified. This flexibility allows for greater agility in procurement strategies and ensures continuous production capability even during periods of market volatility or logistical disruptions affecting specialty chemical shipments.
- Scalability and Environmental Compliance: Scaling exothermic oxidation reactions is traditionally challenging due to heat management issues. However, the controlled addition of aqueous sodium hypochlorite allows for excellent thermal regulation, making the transition from pilot plant to commercial scale safer and more predictable. The process operates at near-ambient or slightly cooled temperatures, reducing the energy load required for cryogenic cooling compared to some alternative methods. From an environmental perspective, the avoidance of chlorinated organic waste associated with peracid byproducts simplifies waste disposal protocols. The resulting aqueous waste streams are easier to treat, aligning with increasingly stringent global environmental regulations and supporting the company's sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this oxidation technology. These insights are derived directly from the experimental data and claims presented in patent CN1839127A, providing a factual basis for decision-making. Understanding these nuances is essential for evaluating the feasibility of integrating this process into existing manufacturing lines or for designing new facilities dedicated to PPI production. The answers reflect the balance between chemical precision and operational practicality required for successful commercialization.
Q: Why is sodium hypochlorite preferred over MCPBA for Pantoprazole oxidation?
A: Sodium hypochlorite is a significantly cheaper commodity chemical compared to m-chloroperbenzoic acid (MCPBA). Furthermore, MCPBA poses a higher risk of over-oxidation to sulfone impurities, whereas controlled NaOCl oxidation offers superior selectivity for the sulfoxide.
Q: How does this process minimize sulfone impurities?
A: By maintaining low reaction temperatures (0°C to 5°C) and strictly controlling the stoichiometry of the oxidant (1.05 to 1.6 equivalents), the kinetic formation of the over-oxidized sulfone byproduct is effectively suppressed.
Q: What are the novel chlorinated derivatives mentioned in the patent?
A: The patent identifies specific chlorinated byproducts, such as 5-(difluoromethoxy)-2-[[(3,4-dimethoxy-2-pyridyl)-chloromethyl]sulfinyl]-1H-benzimidazole, which serve as valuable reference markers for impurity profiling and quality control.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pantoprazole Supplier
At NINGBO INNO PHARMCHEM, we recognize that the theoretical advantages of a patent must be translated into tangible commercial reality through expert process engineering. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the high purity and yield demonstrated in laboratory examples are maintained at an industrial level. We employ stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to monitor critical impurities like sulfones and chlorinated derivatives. Our commitment to quality assurance guarantees that every batch of Pantoprazole intermediate meets the exacting standards required by global regulatory authorities, providing our partners with peace of mind and supply security.
We invite pharmaceutical companies and contract manufacturers to collaborate with us to leverage this advanced oxidation technology for their PPI portfolios. By partnering with our technical procurement team, you can obtain a Customized Cost-Saving Analysis tailored to your specific volume requirements and current supply chain setup. We encourage you to reach out today to request specific COA data, route feasibility assessments, and samples for your qualification process. Let us help you optimize your manufacturing economics while securing a reliable supply of high-quality pharmaceutical intermediates for the global market.
