Advanced Manufacturing of 1,2-Cyclopentanediimide for Global Gliclazide Supply Chains
Advanced Manufacturing of 1,2-Cyclopentanediimide for Global Gliclazide Supply Chains
The global demand for second-generation sulfonylurea oral hypoglycemic agents, particularly Gliclazide, necessitates a robust and efficient supply chain for its critical intermediates. A pivotal breakthrough in this domain is detailed in the Chinese patent CN112358436B, which discloses a superior preparation method for 1,2-cyclopentanediimide. This technical advancement addresses long-standing inefficiencies in the pharmaceutical intermediate sector by replacing harsh, high-temperature dehydration processes with a milder, acetic anhydride-mediated cyclization. For R&D directors and procurement strategists, this patent represents a significant opportunity to optimize the manufacturing of high-purity pharmaceutical intermediates. By shifting away from energy-intensive and polluting legacy methods, manufacturers can achieve yields exceeding 95% while maintaining purity levels above 99.5%. This report analyzes the technical merits of this innovation and its profound implications for cost reduction in API manufacturing and supply chain reliability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of 1,2-cyclopentanediimide has been plagued by severe operational challenges that impact both economic efficiency and environmental compliance. The most common prior art involves the direct thermal dehydration of 2-carbamyl ammonium cyclopentacate at extremely high temperatures, often reaching approximately 250°C. As illustrated in the reaction scheme below, this high-energy pathway frequently results in substantial side reactions, charring, and polymerization, limiting the overall yield to a mediocre range of 60-80%. Furthermore, alternative routes disclosed in patents such as CN102952063A rely heavily on the use of industrial phosphoric acid as a catalyst and dehydrating agent. While effective in driving the reaction, the reliance on phosphoric acid introduces significant downstream processing burdens, including complex neutralization steps and the generation of large volumes of acidic wastewater. These factors collectively increase the total cost of ownership and complicate the regulatory approval process for the final active pharmaceutical ingredient.
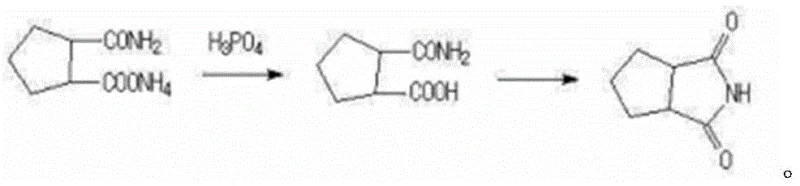
The Novel Approach
In stark contrast to these legacy methodologies, the novel approach outlined in patent CN112358436B utilizes acetic anhydride as both a reactant and a dehydrating medium, fundamentally altering the reaction landscape. This method operates under significantly milder conditions, typically involving reflux temperatures between 100°C and 130°C, which drastically reduces the thermal stress on the molecular structure of the intermediate. The reaction mechanism proceeds through the formation of an acetylated intermediate which subsequently undergoes cyclization and elimination, as depicted in the following chemical equation. This pathway not only accelerates the reaction kinetics but also suppresses the formation of thermal degradation byproducts. The result is a streamlined process that eliminates the need for corrosive phosphoric acid, thereby simplifying the workup procedure to a straightforward extraction and crystallization sequence. This transition represents a paradigm shift towards greener chemistry in the production of reliable pharmaceutical intermediate supplier networks.
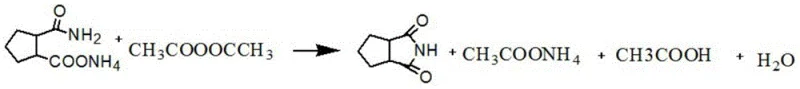
Mechanistic Insights into Acetic Anhydride-Mediated Cyclization
The core of this technological advancement lies in the dual functionality of acetic anhydride, which acts as a potent dehydrating agent while simultaneously activating the carboxylic acid moiety for nucleophilic attack. In the initial stage of the reaction, the ammonium formate salt reacts with acetic anhydride to generate an activated acyl species. This activation lowers the energy barrier required for the subsequent intramolecular nucleophilic attack by the amide nitrogen, facilitating ring closure to form the imide structure. Unlike thermal dehydration which relies solely on entropy-driven water removal at extreme heat, this chemical dehydration pathway is enthalpy-driven and highly selective. The mild thermal conditions prevent the decarboxylation or decomposition of the sensitive cyclopentane ring system, which is a common failure mode in the 250°C thermal process. Consequently, the impurity profile of the crude product is markedly cleaner, containing fewer polymeric tars and unidentified organic byproducts that are difficult to remove.
From a quality control perspective, the mechanism inherently favors the formation of the thermodynamically stable imide ring without generating significant amounts of regio-isomers or open-chain oligomers. The subsequent addition of water serves a critical hydrolytic function, converting any remaining acetic anhydride or acetylated byproducts into water-soluble acetic acid and ammonium acetate, which are easily separated into the aqueous phase. This "self-purifying" aspect of the reaction workup is crucial for achieving the reported purity of >99.5% without the need for resource-intensive distillation or multiple recrystallization steps. For R&D teams focused on impurity profiling, this mechanism offers a predictable and controllable pathway that aligns perfectly with stringent ICH guidelines for residual solvents and genotoxic impurities, ensuring that the commercial scale-up of complex pharmaceutical intermediates remains compliant with global regulatory standards.
How to Synthesize 1,2-Cyclopentanediimide Efficiently
The practical implementation of this synthesis route is designed for scalability and operational simplicity, making it highly attractive for contract development and manufacturing organizations (CDMOs). The process begins with the precise charging of cyclopentane-1-formamide-2-ammonium formate and acetic anhydride into a standard glass-lined or stainless steel reactor. The mixture is heated to reflux, where the temperature is maintained within the optimal window of 100-130°C for a duration of 2 to 10 hours, depending on the specific batch size and agitation efficiency. Following the reaction completion, the excess acetic anhydride is removed under reduced pressure, and the residue is taken up in hot toluene. The detailed standardized operating procedures, including specific molar ratios and cooling profiles required to maximize crystal yield and morphology, are outlined in the technical guide below.
- Charge cyclopentane-1-formamide-2-ammonium formate and acetic anhydride into a reaction vessel and heat to reflux (100-130°C) for 2-10 hours.
- Concentrate the mixture under reduced pressure to remove excess acetic anhydride, then add hot toluene to dissolve the residue completely.
- Add water to the solution, maintain temperature at 40-60°C for hydrolysis, separate the water layer, cool the toluene layer to 5-10°C, and filter to obtain the product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this acetic anhydride-based route offers compelling strategic advantages that extend beyond mere technical performance. The elimination of phosphoric acid removes a major bottleneck in waste management, significantly reducing the environmental footprint and associated disposal costs of the manufacturing process. This aligns with the growing corporate mandates for sustainable sourcing and green chemistry practices. Furthermore, the use of commodity chemicals like acetic anhydride and toluene ensures a stable and resilient supply chain, mitigating the risks associated with specialized or regulated catalyst availability. The simplified purification protocol translates directly into reduced cycle times and lower utility consumption, providing a robust foundation for cost reduction in API manufacturing without compromising on product quality or safety standards.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the drastic simplification of the downstream processing units. By avoiding the use of phosphoric acid, manufacturers eliminate the need for expensive corrosion-resistant equipment and the complex neutralization steps required to treat acidic waste streams. Additionally, the high selectivity of the reaction minimizes the loss of valuable starting materials to side products, effectively improving the atom economy of the process. The ability to achieve high purity through a single crystallization step from toluene further reduces the consumption of solvents and energy typically associated with multi-step purification protocols, leading to substantial operational expenditure savings.
- Enhanced Supply Chain Reliability: The reliance on widely available bulk chemicals such as acetic anhydride and toluene ensures that production schedules are not disrupted by the scarcity of niche reagents. Unlike processes dependent on specific transition metal catalysts or proprietary acids that may face supply volatility, the reagents for this synthesis are produced on a massive global scale. This commoditization of inputs enhances the predictability of lead times and allows for more accurate inventory planning. Moreover, the robustness of the reaction conditions means that the process is less sensitive to minor fluctuations in raw material quality, ensuring consistent output even when sourcing from diverse suppliers in the global market.
- Scalability and Environmental Compliance: Scaling this process from pilot plant to commercial production is facilitated by the moderate temperature and pressure requirements, which reduce the engineering complexity of the reactor systems. The absence of high-temperature operations (250°C) lowers the risk of thermal runaway incidents, enhancing overall plant safety. From an environmental standpoint, the replacement of phosphoric acid with acetic anhydride significantly reduces the load of inorganic salts in the wastewater, simplifying effluent treatment and ensuring compliance with increasingly strict environmental regulations. This eco-friendly profile not only mitigates regulatory risk but also enhances the brand reputation of the final pharmaceutical product among environmentally conscious stakeholders.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis route. These insights are derived directly from the experimental data and comparative analysis presented in patent CN112358436B, providing clarity on yield optimization, purity specifications, and process safety. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this method into their existing manufacturing portfolios.
Q: What is the primary advantage of the acetic anhydride method over traditional phosphoric acid routes?
A: The acetic anhydride method eliminates the need for large quantities of phosphoric acid, significantly reducing environmental pollution and simplifying the purification process by avoiding complex neutralization and waste treatment steps.
Q: What purity levels can be achieved with this novel synthesis route?
A: According to patent CN112358436B, the optimized process yields 1,2-cyclopentanediimide with a content exceeding 99.5% and a reaction yield of up to 95.0%, surpassing traditional methods that often struggle with side reactions.
Q: How does the reaction temperature compare to conventional high-temperature dehydration?
A: Unlike conventional methods requiring temperatures around 250°C which promote side reactions and charring, this novel approach operates at a mild reflux temperature of 100-130°C, ensuring better thermal stability and product quality.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,2-Cyclopentanediimide Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced synthetic routes requires a partner with deep technical expertise and proven manufacturing capabilities. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our state-of-the-art facilities are equipped to handle the specific requirements of the acetic anhydride-mediated cyclization process, adhering to stringent purity specifications and rigorous QC labs to guarantee that every batch of 1,2-cyclopentanediimide meets the highest international standards. We are committed to delivering high-purity pharmaceutical intermediates that empower your drug development pipelines.
We invite you to collaborate with us to leverage this cutting-edge technology for your Gliclazide production needs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this optimized route can enhance your bottom line. Please contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments, and let us help you secure a competitive advantage in the global pharmaceutical market.
